Clear Sky Science · en
Isotopic and defect analysis of enriched molybdenum oxide using EPR spectroscopy and DFT simulation
Why tiny differences in atoms matter for medicine
Molybdenum might sound like an obscure metal, but some of its atomic varieties (isotopes) sit at the heart of modern medical imaging. A key hospital tracer, technetium‑99m, is made from molybdenum isotopes, and global demand for it is huge. Safely tracking and verifying these valuable isotopes is difficult, because today’s leading analytical tools often destroy the sample. This study explores a non‑destructive way to tell molybdenum isotopes apart and to see subtle flaws in their crystal form, potentially helping future nuclear medicine, advanced alloys, and materials research.
Special versions of a useful metal
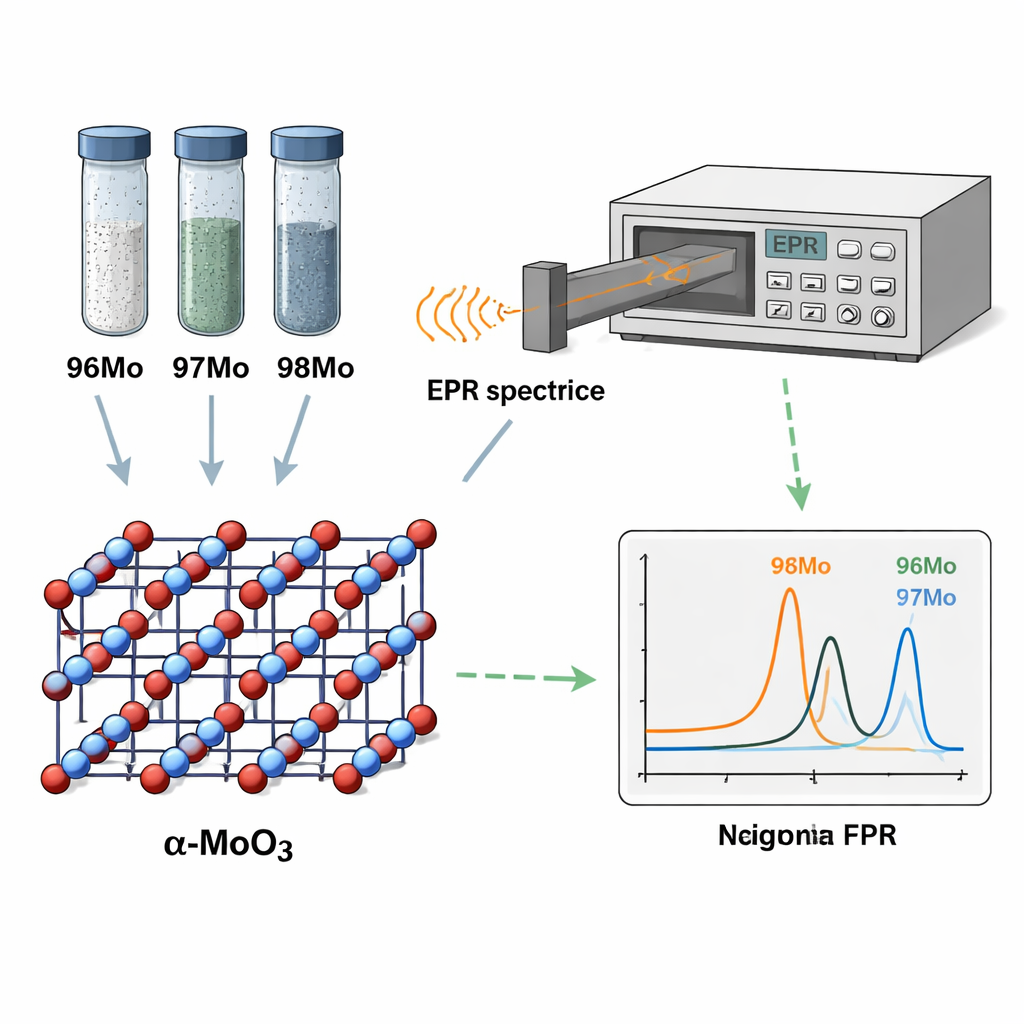
Molybdenum occurs in several stable isotopes that differ by just a few neutrons in the nucleus. Three of them—96Mo, 97Mo, and 98Mo—are especially important because they are precursors to technetium‑99m, used to scan organs such as the heart, lungs, and thyroid. Industry typically enriches these isotopes by electromagnetic separation, which yields powders that are both precious and difficult to waste. Standard mass‑spectrometry methods can measure their ratios precisely but require dissolving the sample, extensive chemistry, and expensive instrumentation. The authors instead turned to electron paramagnetic resonance (EPR), a technique that detects unpaired electrons in a magnetic field, to see whether tiny isotope‑dependent shifts in electronic structure could reveal which molybdenum atom is which—without damaging the material.
Looking at light and magnetism in crystal powders
The team produced enriched samples of 96Mo, 97Mo, and 98Mo, purified them chemically, and confirmed their crystal form as α‑MoO₃ using standard tools such as X‑ray diffraction and electron microscopy. They then shone ultraviolet light on the powders and recorded the emitted light with photoluminescence (PL) spectroscopy. These PL spectra showed bright features near the band edge of pristine α‑MoO₃ and additional peaks caused by defects—small disruptions such as extra or missing oxygen atoms or missing molybdenum atoms. However, the PL peaks from different defects largely overlapped, making it impossible to say exactly which defects were present or to extract isotope information from light alone. This limitation motivated a closer look with EPR, which directly probes how unpaired electrons respond to a magnetic field and can sense much finer differences in energy levels.
Defects in the crystal and what they reveal
Using X‑band EPR (around 10 GHz), the researchers observed distinct resonance patterns for the three enriched powders: the 96Mo and 98Mo samples each showed a single main peak, while the 97Mo sample showed a more complex, multi‑peak signal. To interpret these patterns, they performed first‑principles (ab initio) calculations based on density functional theory (DFT) and molecular dynamics (MD). These simulations mapped the electronic band structure of α‑MoO₃, calculated how various native defects form under oxygen‑rich conditions, and predicted how each defect would modify the EPR signal. The work identified several likely defects—different kinds of extra oxygen, missing molybdenum, and their combinations—as stable in a positively charged state. These defects create energy levels that explain the visible‑range PL emission and host unpaired electrons that produce characteristic EPR fingerprints.
Subtle isotope fingerprints in the magnetic signal
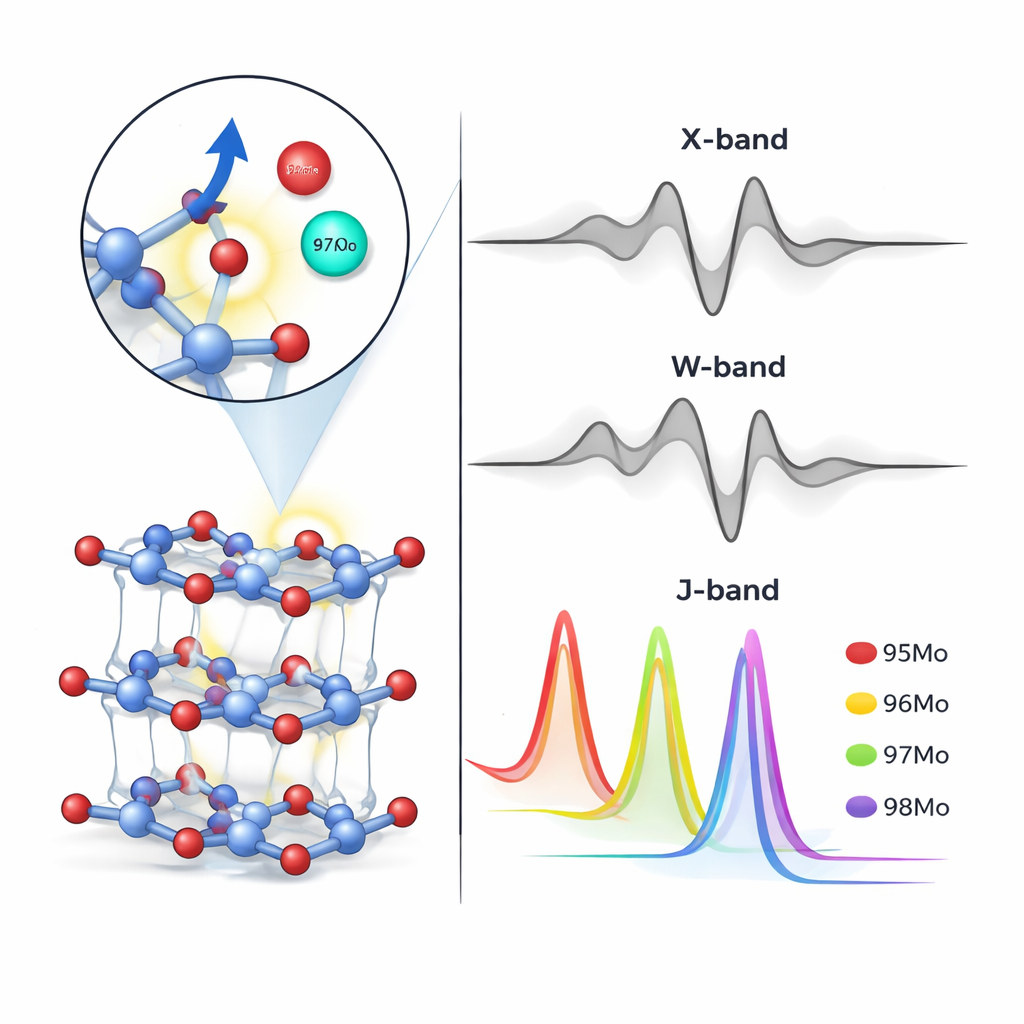
Beyond defects, the study examined how different molybdenum isotopes slightly change the EPR response through their nuclear mass and spin. Isotopes with nuclear spin, such as 95Mo and 97Mo, cause extra splitting of the EPR lines, while spin‑zero isotopes like 96Mo and 98Mo do not. By combining experiment and theory, the authors matched specific resonance fields to particular defect–isotope combinations: for example, certain oxygen‑related defects dominated in 96Mo and 98Mo samples, while a missing‑molybdenum defect was linked to the 97Mo sample. Static calculations alone were not accurate enough, so the team used MD snapshots to capture thermal motion and refine the predicted hyperfine interactions. The comparison between simulated and measured spectra showed that the enriched powders were indeed highly separated in isotope content, confirming electromagnetic enrichment and showcasing the sensitivity of EPR to isotopic makeup.
Why higher frequencies could unlock a new tool
At the commonly used X‑band frequency, the small shifts between isotopes cause EPR peaks to overlap, which limits how precisely one can read out isotope ratios from a single spectrum. The researchers therefore simulated what would happen at much higher microwave frequencies—the W‑band and J‑band—using the defect and isotope parameters extracted at X‑band. In these simulations, the resonance lines for molybdenum isotopes spread out and become clearly separated, suggesting that high‑frequency EPR could, in principle, resolve all isotopes and even quantify their amounts from peak intensities. While the team did not have access to such high‑frequency instruments, their results outline how a future non‑destructive, calibration‑based EPR method could complement or partially replace destructive mass‑spectrometry for analyzing precious isotope‑enriched materials.
What this means for future applications
For a non‑specialist, the key takeaway is that the way a crystal’s unpaired electrons respond to a magnetic field subtly remembers which version of an atom sits nearby. By pairing careful experiments with advanced simulations, this study shows that electron paramagnetic resonance can not only pinpoint specific defects in molybdenum oxide but also sense which molybdenum isotope is present. With access to higher‑frequency EPR spectrometers and proper calibration, this approach could evolve into a practical, non‑destructive tool for monitoring medical‑grade isotopes and studying complex materials where every atom—and every defect—matters.
Citation: Hosseini, R., Karimi-sabet, J., Janbazi, M. et al. Isotopic and defect analysis of enriched molybdenum oxide using EPR spectroscopy and DFT simulation. Sci Rep 16, 6128 (2026). https://doi.org/10.1038/s41598-026-37195-6
Keywords: molybdenum isotopes, electron paramagnetic resonance, defects in crystals, medical imaging tracers, high frequency spectroscopy