Clear Sky Science · en
Structural characterization of metal binding in human tyrosylprotein sulfotransferase 2, TPST2
Why tiny metal ions matter for cell signaling
Inside our cells, countless molecular machines quietly tune how tissues grow, hormones act, and immune cells communicate. One such machine, an enzyme called TPST2, adds a small sulfate tag to certain proteins as they pass through the cell’s shipping center, the Golgi apparatus. This study reveals how simple metal ions like sodium and manganese help TPST2 work more efficiently by subtly stiffening parts of its structure—an insight that may connect mineral balance in cells to cancer and hormone-related diseases. 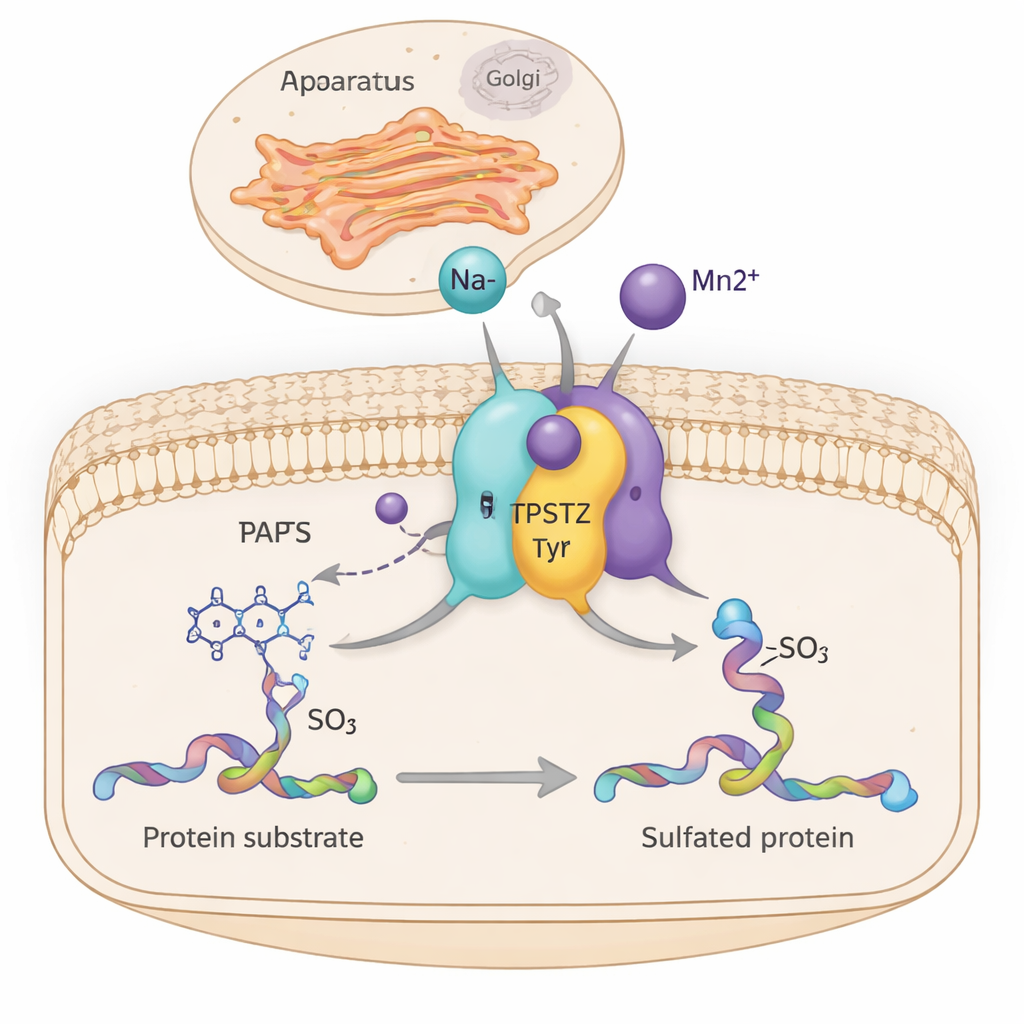
A protein that decorates other proteins
TPST2 belongs to a family of enzymes that perform “tyrosine sulfation,” a chemical modification added to specific tyrosine building blocks on proteins. These sulfated proteins play key roles outside cells, influencing blood clotting, white blood cell movement, hormone–receptor binding, and even how viruses enter cells. In humans there are two main sulfating enzymes, TPST1 and TPST2, which sit in the membrane of the Golgi and modify proteins as they are prepared for export. Mouse studies show that losing TPST2 disrupts thyroid hormone production, stunts growth, and causes male infertility, highlighting its importance for normal physiology.
Links to cancer and immune escape
Recent research has tied TPST2 directly to disease. In melanoma, TPST2 modifies a component of the interferon receptor in a way that weakens immune signaling, and blocking TPST2 boosts the impact of immune checkpoint therapies that target PD-1. In pancreatic cancer, a TPST2-dependent pathway helps stabilize a protein (integrin β4) that promotes tumor growth and spread. Inhibiting TPST2 in these models slows cancer cell division and invasion. Because TPST2 acts on proteins at the cell surface, it effectively rewires how cells sense and respond to their surroundings, making it an attractive target for new therapies.
Finding the metal “on switches”
Biochemists have long known that TPST2 becomes much more active when divalent metals such as manganese (Mn²⁺) are present, but the structural reason was missing. In this study, the authors produced the catalytic core of human TPST2 and crystallized it together with a reaction by-product and either sodium (Na⁺) or manganese. Using high-resolution X-ray crystallography, they discovered two small metal-binding pockets within the enzyme, each with a neat, six-sided (octahedral) arrangement of surrounding atoms. By tuning the X-ray wavelength to the unique signatures of different metals, they confirmed that Mn²⁺ selectively occupies these sites, while metals like zinc and copper either bind weakly at other locations or not at all.
Stiffening key moving parts instead of reshaping the whole
Surprisingly, TPST2 with sodium and TPST2 with manganese looked almost identical overall; there was no dramatic opening or closing of the protein. Instead, the metals mainly affected two flexible regions near the entrance to the active site: a helix called α3 and a nearby loop known as the α12–α13 loop. In metal-free or loosely bound states these segments wiggle and are partly disordered. When Na⁺ binds, they become more ordered; Mn²⁺ tightens them further, as shown by reduced thermal motion in the X-ray data and by computer-based flexibility analyses. This extra rigidity modestly raises the enzyme’s melting temperature and is thought to lower the “entropic” cost of organizing the active site—making it easier and faster for TPST2 to guide substrates into the right position for sulfation. 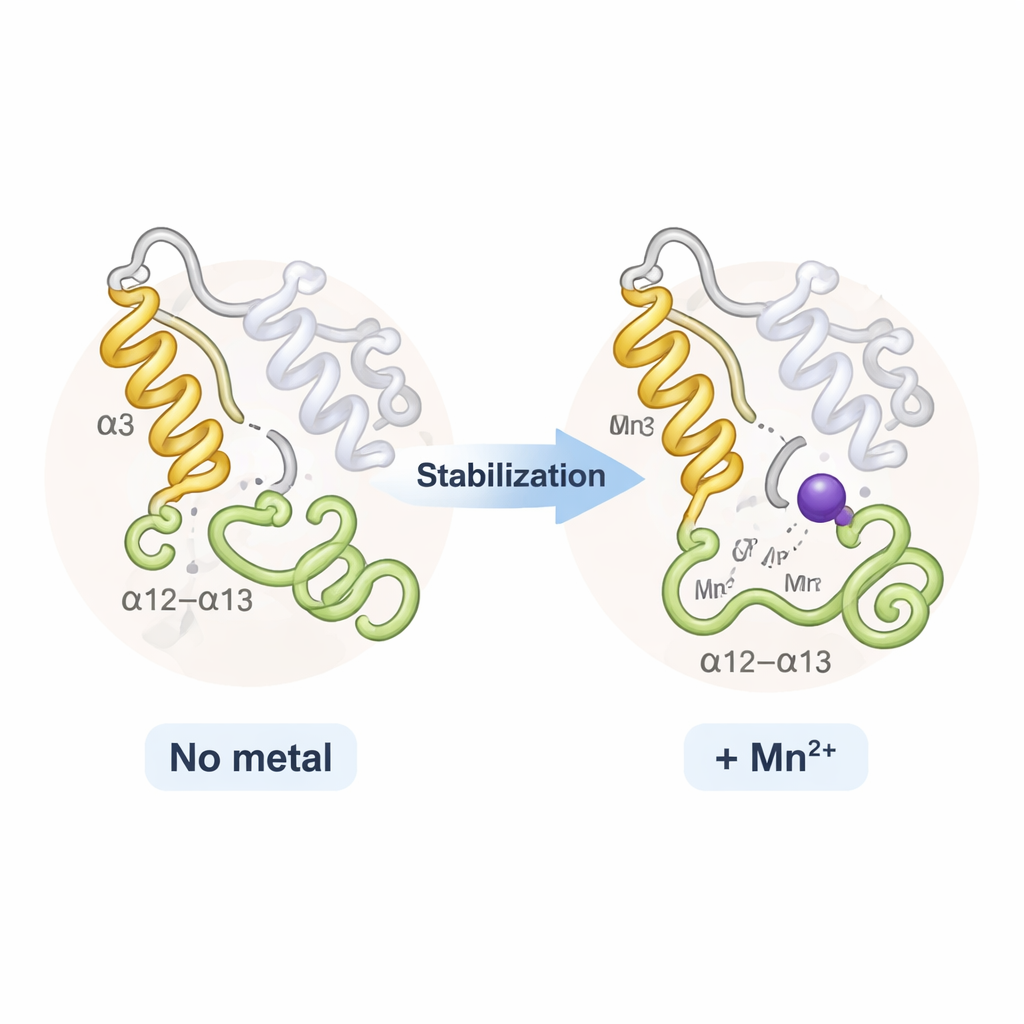
From cell minerals to signaling control
The authors propose an “activation-by-ordering” model. In the absence of bound metal, TPST2 is floppy around its active-site entrance and works inefficiently. Sodium, abundant in cells, can bind the same sites and create a partially ordered, modestly active state. Manganese, present in carefully controlled micromolar amounts within the Golgi, binds more strongly and locks the key regions into an optimally arranged state, maximizing catalytic efficiency. This suggests that subtle shifts in Golgi manganese levels could tune how strongly TPST2 modifies its targets. Because TPST2 influences hormone production and cancer signaling, the work highlights a potential link between metal ion balance, protein “decorations,” and disease—and provides a structural framework for designing drugs that modulate TPST2 activity by mimicking or disrupting metal binding.
Citation: Jin, M., Noh, C., Yang, J. et al. Structural characterization of metal binding in human tyrosylprotein sulfotransferase 2, TPST2. Sci Rep 16, 6066 (2026). https://doi.org/10.1038/s41598-026-37189-4
Keywords: tyrosine sulfation, TPST2, manganese, Golgi apparatus, protein modification