Clear Sky Science · en
Optimizing immersion time and frequency in a twin-bottle temporary immersion system for mass production of Zingiber officinale Roscoe
Why ginger growers need a lab helper
Ginger is more than a kitchen spice; it is a global medicinal crop used for everything from easing nausea to supporting heart and metabolic health. Yet farmers still mostly rely on chunks of rhizome to plant new fields, a slow method that spreads soil‑borne diseases and limits supply. This study explores how a simple, automated glass‑bottle system can mass‑produce clean, robust ginger plants in the lab, offering a way to support growing demand while protecting crops and soil.
A bottleneck in ginger planting material
Because ginger rarely makes good seed, nearly all new plants come from dividing underground rhizomes. That approach ties production to field space, invites pathogens that live in soil, and usually allows only one crop per year on the same land. Tissue culture—growing tiny plant pieces in sterile flasks—can generate many disease‑free clones, but traditional methods use small jars and a lot of hands‑on labor. The authors focused on a "temporary immersion" system, where ginger shoots are periodically bathed in liquid nutrients and then exposed to air. Their goal was to tune this system so it would grow large numbers of strong, transplant‑ready plants at low cost.
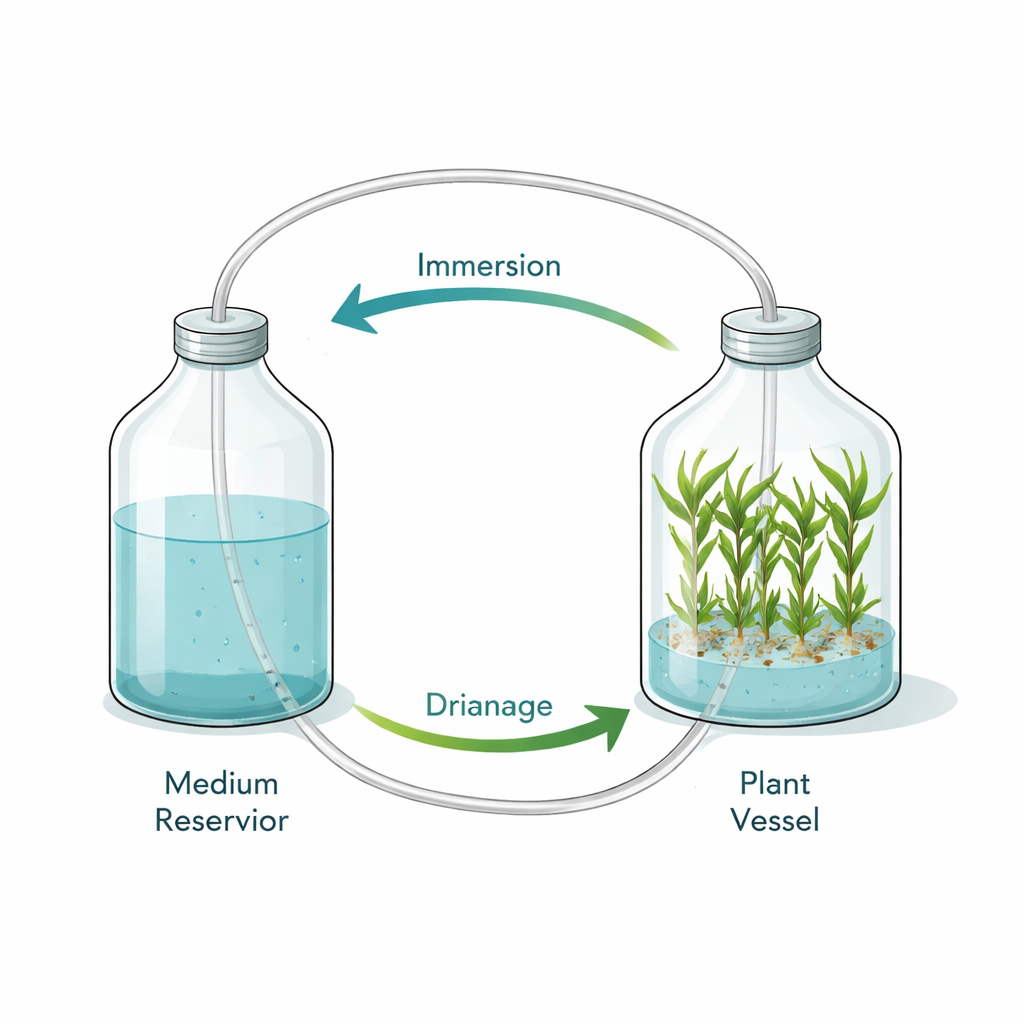
Finding the sweet spot for dipping plants
In the twin‑bottle setup, one bottle holds the nutrient solution and the other holds the ginger shoots. Air pressure moves the liquid in and out, briefly immersing the shoots several times a day. The team tested nine schedules, changing how often and how long the shoots were immersed. Too much immersion—especially many times a day for long periods—stunted plants, shortened leaves, and sharply reduced survival after they were moved to soil. In contrast, a moderate schedule of six immersions a day, each lasting only five minutes, produced the tallest shoots, the heaviest clumps, and the longest leaves, along with the highest survival in the greenhouse: nearly 9 out of 10 plantlets lived.
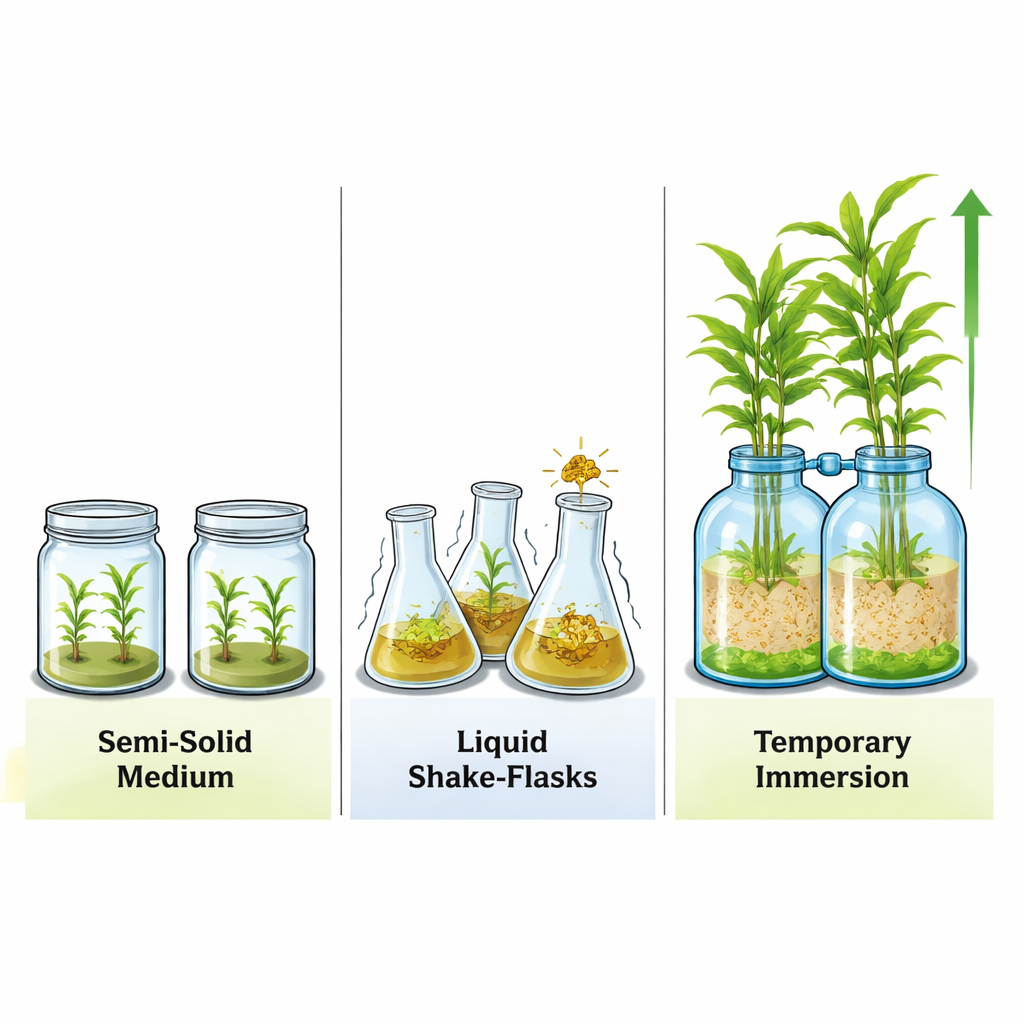
Comparing three ways to grow ginger in glass
Next, the researchers asked how this temporary dipping system stacked up against two familiar approaches: solid gel medium in jars and constant shaking in liquid flasks. They also added different doses of a growth‑shortening chemical, chlormequat chloride, which in field crops can direct more energy into storage organs. Across all treatments, the temporary immersion bottles clearly produced the strongest vegetative growth—taller shoots, longer leaves, and greater fresh weight—especially when no chemical or only a low dose was used. The shake flasks could generate many shoots but often yielded weaker plants, while the solid medium gave fewer new plants overall.
From glassware to greenhouse
Ultimately, any lab method for ginger must prove itself when plants leave the controlled environment and face real-world conditions. Plantlets raised in the temporary immersion system were more likely to survive this transition than those from the other two systems, with survival rates around 80 percent in the best treatments. This suggests that the intermittent wet‑and‑dry cycles in the bioreactor help the young plants develop leaves and roots that function more normally once exposed to sunlight, drier air, and soil microbes.
What this means for future ginger fields
The study shows that a relatively simple twin‑bottle immersion setup, run on a carefully chosen schedule of brief, frequent dips, can act as a plant "factory" for ginger. While the researchers did not yet succeed in triggering the formation of small storage rhizomes in vitro—a separate challenge that will require adjusting sugar levels and hormones—the optimized protocol already delivers large numbers of healthy, uniform ginger plants at low cost. For growers and nurseries, this approach offers a scalable way to secure clean planting stock, stabilize supply chains, and support sustainable commercial ginger production.
Citation: Kongbangkerd, A., Tubtimsri, K. & Kunakhonnuruk, B. Optimizing immersion time and frequency in a twin-bottle temporary immersion system for mass production of Zingiber officinale Roscoe. Sci Rep 16, 6291 (2026). https://doi.org/10.1038/s41598-026-37182-x
Keywords: ginger micropropagation, temporary immersion system, plant bioreactor, tissue culture, disease-free planting material