Clear Sky Science · en
Integrative machine learning and molecular docking reveal Narirutin targets ABCC1/ABCG2 for immune microenvironment remodeling in colorectal cancer
Fruit Compound With Big Cancer Hopes
Colorectal cancer is one of the world’s deadliest cancers, and many patients still face harsh treatments and drug resistance. This study looks at narirutin, a natural substance found in citrus fruits, to see whether it might help the body fight colon tumors more gently—by changing how cancer cells handle drugs and how immune cells behave around the tumor.
From Citrus Fruit to Cancer Target
Researchers started by asking a simple question: if narirutin were a medicine, which parts of our cells would it latch onto? Using large public cancer databases and computer prediction tools, they scanned thousands of genes from colon tumors and healthy tissue. They then overlapped these cancer‑linked genes with those that narirutin is most likely to affect. Out of more than 3,000 altered genes in colon cancer, narirutin was predicted to meaningfully interact with just nine, and especially with five “hub” genes that sit at key control points inside tumor cells.
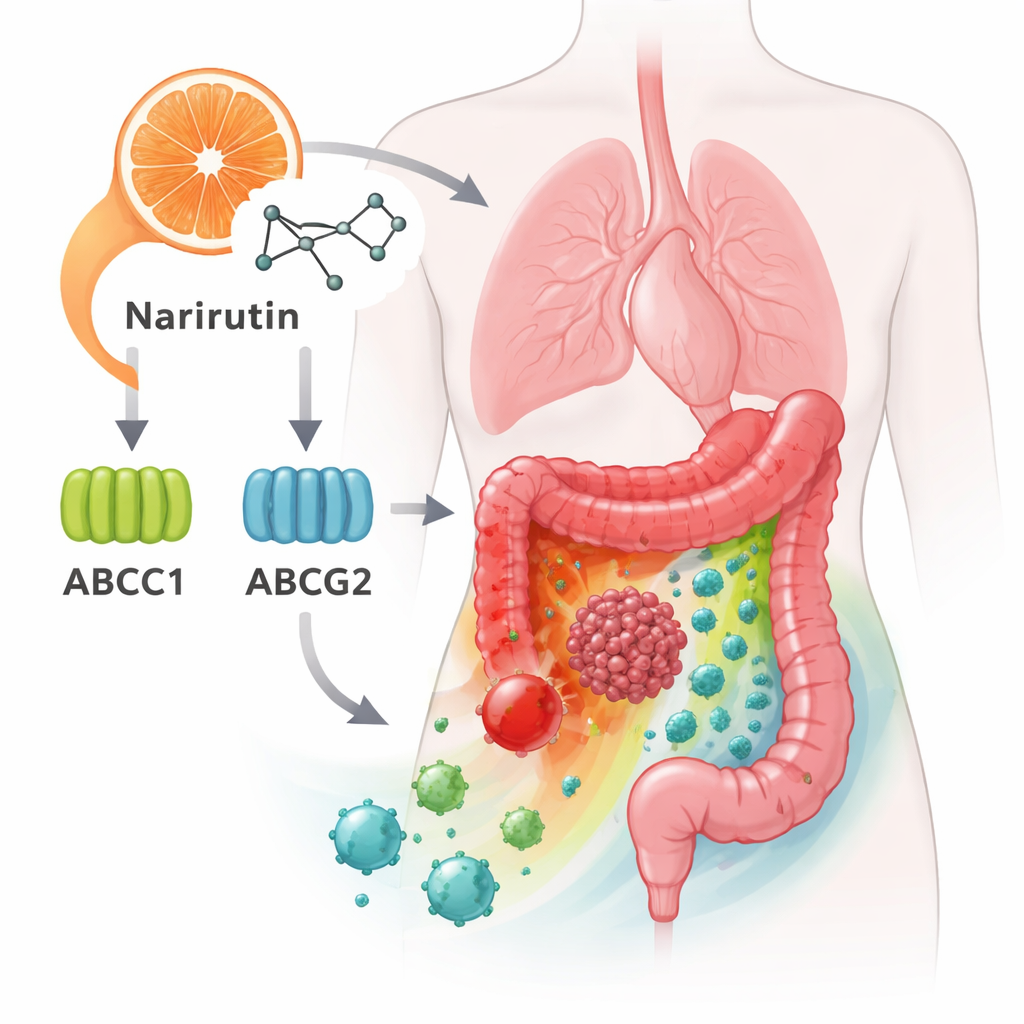
Key Gatekeepers on Cancer Cell Surfaces
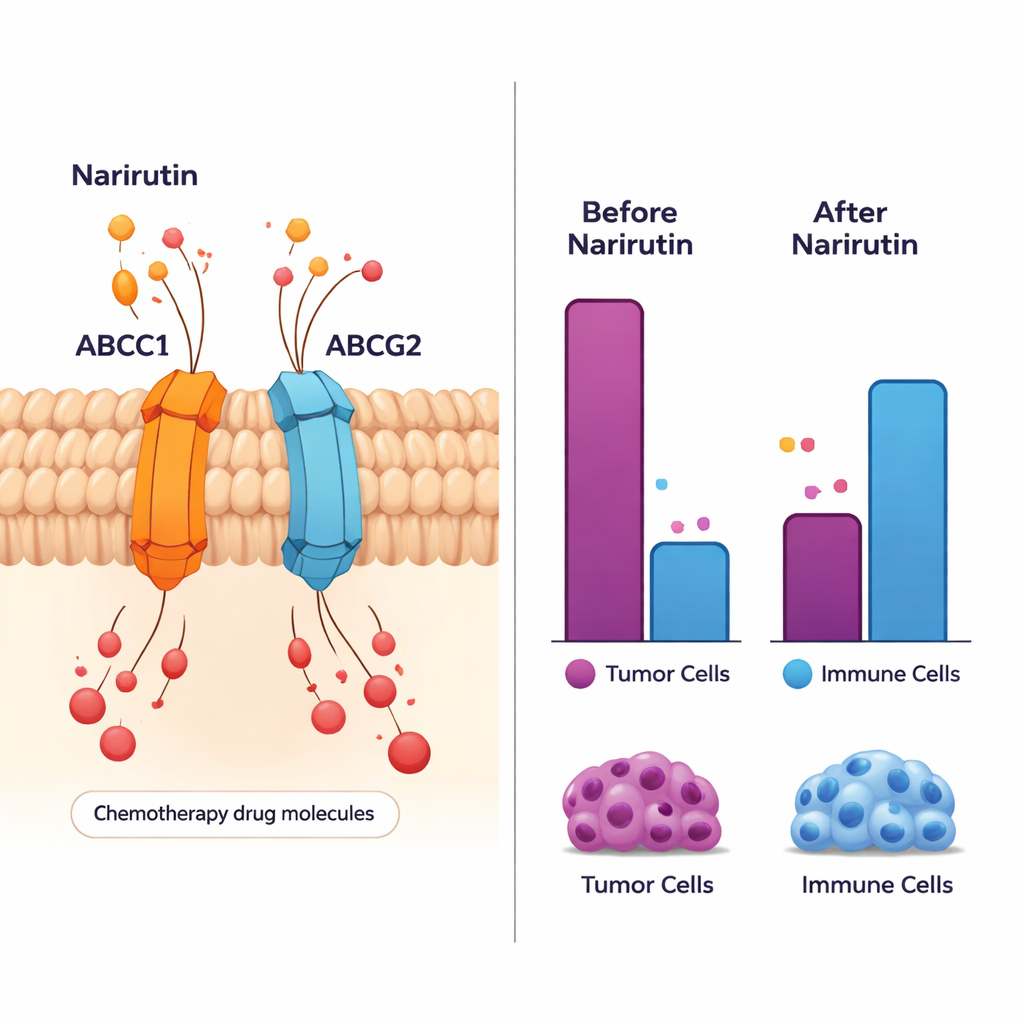
Among these hub genes, two stood out: ABCC1 and ABCG2. They make pump‑like proteins that sit in the outer membrane of cells and push drugs back out—one reason some cancers become resistant to chemotherapy. The new analysis suggests narirutin can bind to these pumps and possibly influence how they work. Other hub genes, including CA12, EPHX2, and PTGS1, are involved in how cells manage acids, fats, and inflammatory signals. Together, these genes help determine whether a tumor grows unchecked, how it responds to medicines, and how it interacts with nearby normal tissue.
Simulated Binding and Real‑World Outcomes
To test whether narirutin could truly stick to these target proteins, the team used molecular docking—high‑resolution computer models that predict how a small molecule fits into a larger protein. These simulations showed that narirutin fits snugly into pockets on its target proteins and forms stable contacts, meaning binding is physically plausible. When the researchers looked at patient data, they found that the hub genes strongly distinguished cancer tissue from normal tissue and were tied to how long people lived. For example, higher levels of ABCC1 and PTGS1 tended to go along with poorer outcomes, suggesting that turning these genes down—potentially with compounds like narirutin—could be beneficial.
Re‑Shaping the Tumor’s Immune Neighborhood
Cancer does not grow in isolation; it lives within a busy “neighborhood” of immune cells, blood vessels, and supporting tissue. The study examined 28 different immune cell types around colon tumors and found broad shifts compared with healthy tissue. Certain helpful immune cells, such as activated B cells and specific T cells, were more abundant or behaved differently in tumors. Narirutin’s main targets, especially ABCC1, ABCG2, and PTGS1, showed strong links to these immune changes. This suggests that by acting on these genes, narirutin might not just affect cancer cells directly, but also tune the local immune response—possibly making the environment less friendly to tumor growth.
Promise, With Work Still To Do
In everyday terms, this research proposes that a natural flavonoid from citrus could “jam the pumps” cancer cells use to escape chemotherapy and help reset the immune landscape around colon tumors. The work is based on powerful computer analyses and large genetic datasets, rather than lab or clinical trials, so it represents an early but important step. If future experiments in cells, animals, and patients confirm these predictions, narirutin—or drugs inspired by it—could one day become part of combination therapies that make standard treatments for colorectal cancer more effective and less prone to resistance.
Citation: Bian, R., Wang, H., Zhang, H. et al. Integrative machine learning and molecular docking reveal Narirutin targets ABCC1/ABCG2 for immune microenvironment remodeling in colorectal cancer. Sci Rep 16, 7649 (2026). https://doi.org/10.1038/s41598-026-37181-y
Keywords: colorectal cancer, narirutin, drug resistance, tumor microenvironment, immune modulation