Clear Sky Science · en
Probing into the binding mechanism of phosphorus(V)-corrole with hemoglobin using photophysical and computational approach
Light-Activated Medicines Riding in Your Blood
Many next‑generation cancer treatments rely on special dyes that light up or become toxic only when shone with a specific color of light. One such dye family, called corroles, shows promise for killing tumor cells with fewer side effects. But before any such agent can be used safely in people, scientists must know how it travels through the body, especially how it interacts with the main oxygen‑carrying protein in our blood: hemoglobin. This study explores exactly how a new phosphorus‑based corrole latches onto human hemoglobin, and what that means for turning blood proteins into natural drug carriers. 
A New Partner for the Blood’s Oxygen Carrier
Hemoglobin, packed inside red blood cells, ferries oxygen from the lungs to every organ and brings back carbon dioxide for removal. Because it is abundant and has many pockets and surfaces where small molecules can nestle, hemoglobin can also bind drugs and influence how long they circulate. Corroles are ring‑shaped pigment molecules related to the heme group inside hemoglobin, but they can be chemically tuned for medical tasks such as imaging, fighting infections, or destroying cancer cells using light. The researchers focused on one specially designed phosphorus(V)‑corrole, called 1P, chosen for its stability, strong light absorption, and ability to generate reactive oxygen species used in photodynamic therapy.
Watching Molecules Talk with Light
To see whether 1P truly binds to hemoglobin, the team first used light‑based techniques. By shining ultraviolet and visible light through solutions of hemoglobin with increasing amounts of 1P, they tracked subtle shifts in the protein’s characteristic absorption peaks. These changes revealed that 1P and hemoglobin form a stable complex in their ground state rather than merely bumping into each other. Fluorescence experiments, which measure the natural glow of specific amino acids in hemoglobin, showed that this glow dims in a way best explained by 1P forming a tight complex close to these glowing residues. From the degree of dimming at different temperatures, the scientists calculated a sizeable binding strength and a negative Gibbs free energy, meaning the interaction occurs spontaneously and is energetically favorable under body‑like conditions.
How Binding Nudges Protein Shape
Because drug binding can gently reshape a protein, the researchers next examined hemoglobin’s structure using circular dichroism, a method that reads out how protein helices and coils absorb swirling light. Adding more 1P slightly reduced the signal linked to hemoglobin’s helical content, indicating a modest loosening of its local structure rather than a full collapse. When they heated hemoglobin with and without 1P, the complex began to unfold a few degrees earlier, again pointing to mild destabilization. These results suggest that 1P lodges near key structural regions—enough to tweak the protein’s stability and environment around the heme groups, but not enough to destroy hemoglobin’s overall architecture or function. 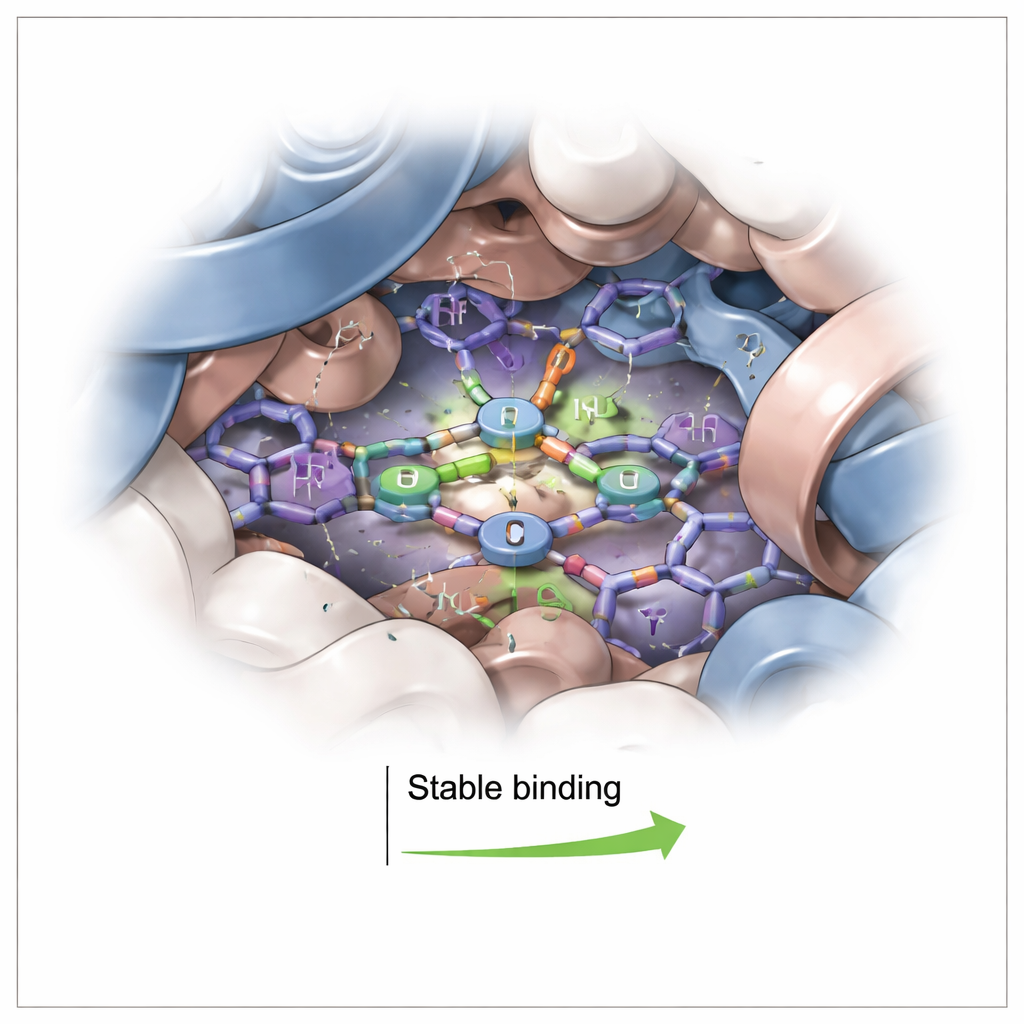
Computer Simulations Reveal the Seating Plan
To visualize exactly where 1P sits, the team turned to computer modeling. They docked 1P onto a high‑resolution structure of human hemoglobin, then simulated the complex in water for 100 billionths of a second. The simulations showed 1P settling into an aromatic pocket within about one billionth of a meter from the heme, but not directly binding the iron center. Instead, the corrole’s flat, ring‑like surface stacked against nearby aromatic amino acids, supported by occasional hydrogen bonds. Throughout the simulation, both hemoglobin’s overall shape and the position of 1P remained remarkably stable. Energy calculations confirmed that the binding is strongly favored, driven mainly by close‑packing and “oily” (hydrophobic) contacts rather than by strong electrical attraction alone.
What This Means for Future Light‑Driven Drugs
Taken together, these experiments and simulations show that phosphorus(V)‑corrole 1P binds tightly and specifically to human hemoglobin, forming a stable complex that only modestly alters the protein’s structure. In plain terms, 1P finds a comfortable seat on hemoglobin without kicking out its vital heme group. This makes hemoglobin a promising natural shuttle for delivering corrole‑based medicines through the bloodstream, potentially improving how long they circulate and how effectively they reach diseased tissues. By clarifying where and how 1P binds, this work lays groundwork for designing safer light‑activated drugs that harness our own blood proteins as built‑in carriers.
Citation: Kritika, Kubba, R., Kumar, L. et al. Probing into the binding mechanism of phosphorus(V)-corrole with hemoglobin using photophysical and computational approach. Sci Rep 16, 6097 (2026). https://doi.org/10.1038/s41598-026-37177-8
Keywords: hemoglobin drug transport, photodynamic therapy, corrole photosensitizer, protein ligand binding, molecular docking