Clear Sky Science · en
Mechanistic insights into modification of clay minerals by detergent-derived surfactants and their impact on petroleum hydrocarbon uptake
Why Detergents and Clays Matter for Oil Pollution
Spills of diesel, kerosene, and other petroleum products can linger in soil and water for years, harming ecosystems and human health. One promising clean-up strategy is to use naturally abundant clays as sponges that trap these stubborn hydrocarbons. This study asks a deceptively simple question with big practical consequences: when everyday detergent ingredients stick to clays, do they just coat the outside, or do they actually work their way between the clay layers—and how does that choice of "parking spot" change how well clays can soak up spilled fuel?
Two Very Different Natural Sponges
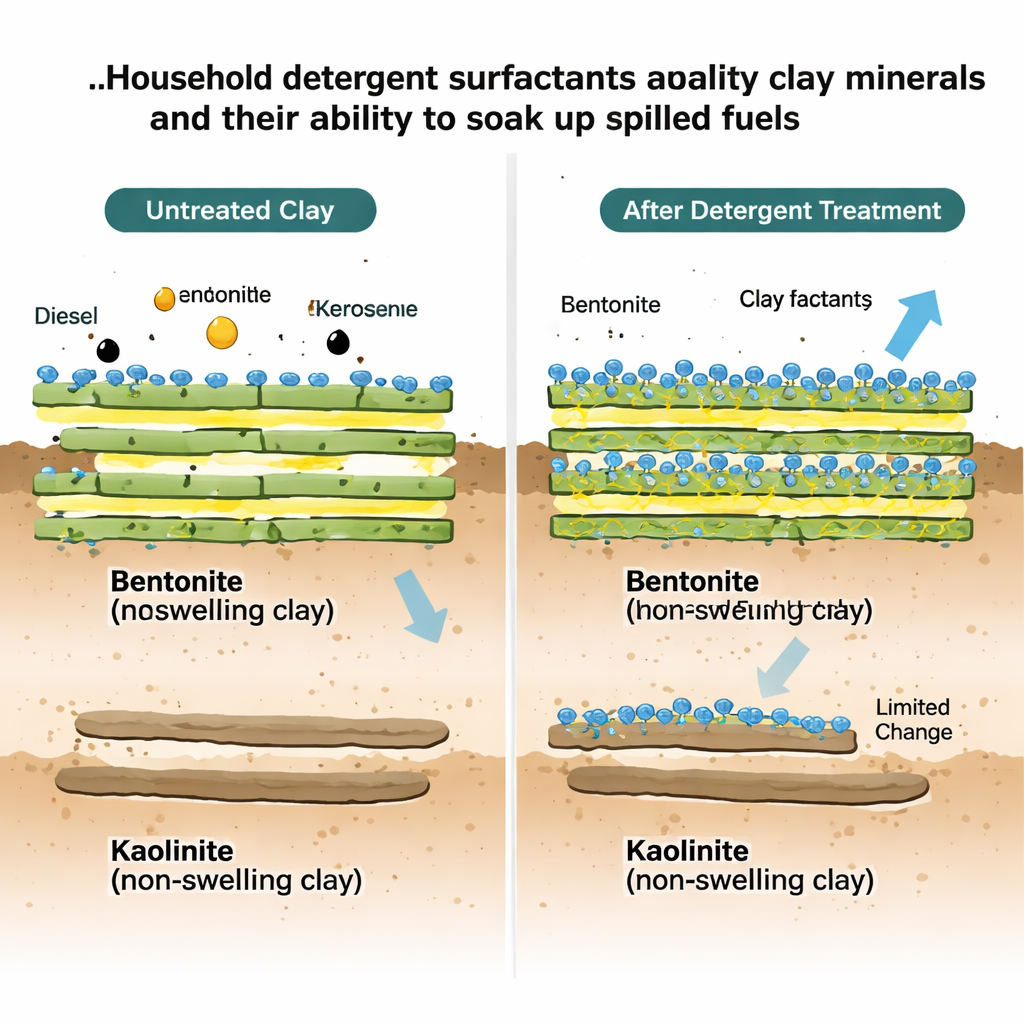
The researchers focused on two common clays, bentonite and kaolinite, which behave very differently at the microscopic level. Bentonite is a “swelling” clay made of stacks of sheets that can slide apart, creating internal galleries where liquids and molecules can slip in. Kaolinite, by contrast, has tighter, non-expandable layers and mainly offers its outer surfaces and edges for interactions. Because of these structural contrasts, bentonite is naturally better at tucking guest molecules between its layers, while kaolinite mostly hosts them on the outside. The team wanted to see how this plays out when clays encounter the surfactants—the active cleaning molecules—found in household detergents.
Turning Laundry Soap into a Clay Modifier
Instead of using pure laboratory surfactants, the study uses three real-world commercial detergents representing kitchen, bathroom, and laundry products. Their surfactant content was first characterized by how strongly they lower water’s surface tension and by measuring a key point called the critical micelle concentration, where surfactant molecules start to cluster. The clays were then treated with very dilute detergent solutions just below this threshold, a regime in which individual surfactant molecules dominate. By comparing the detergent concentration before and after contact with the clay, the authors calculated how much surfactant each gram of clay had captured. Bentonite consistently took up more (about 2.8–3.1 milligrams per gram) than kaolinite (about 2.5–2.7 milligrams per gram), hinting that its internal galleries were playing an active role.
Watching Fluids Creep into Clay
Measuring total surfactant on a clay is one thing; figuring out exactly where those molecules end up is another. To solve this, the team combined two clever but relatively simple laboratory approaches. In a capillary rise experiment, they tracked how fast and how much liquid (water, detergents, diesel, or kerosene) was drawn into a carefully packed clay bed over time, which reveals how readily fluid can access tiny spaces, including interlayers. In a companion immersion setup, they measured changes in buoyant force when clay was submerged, which carries information about how much fluid ends up inside the particles versus outside. By running these tests before and after detergent treatment, and with different probe liquids, they built a “four-stage” diagnostic picture of whether the surfactants mainly coat external faces or truly move into the galleries between clay sheets.
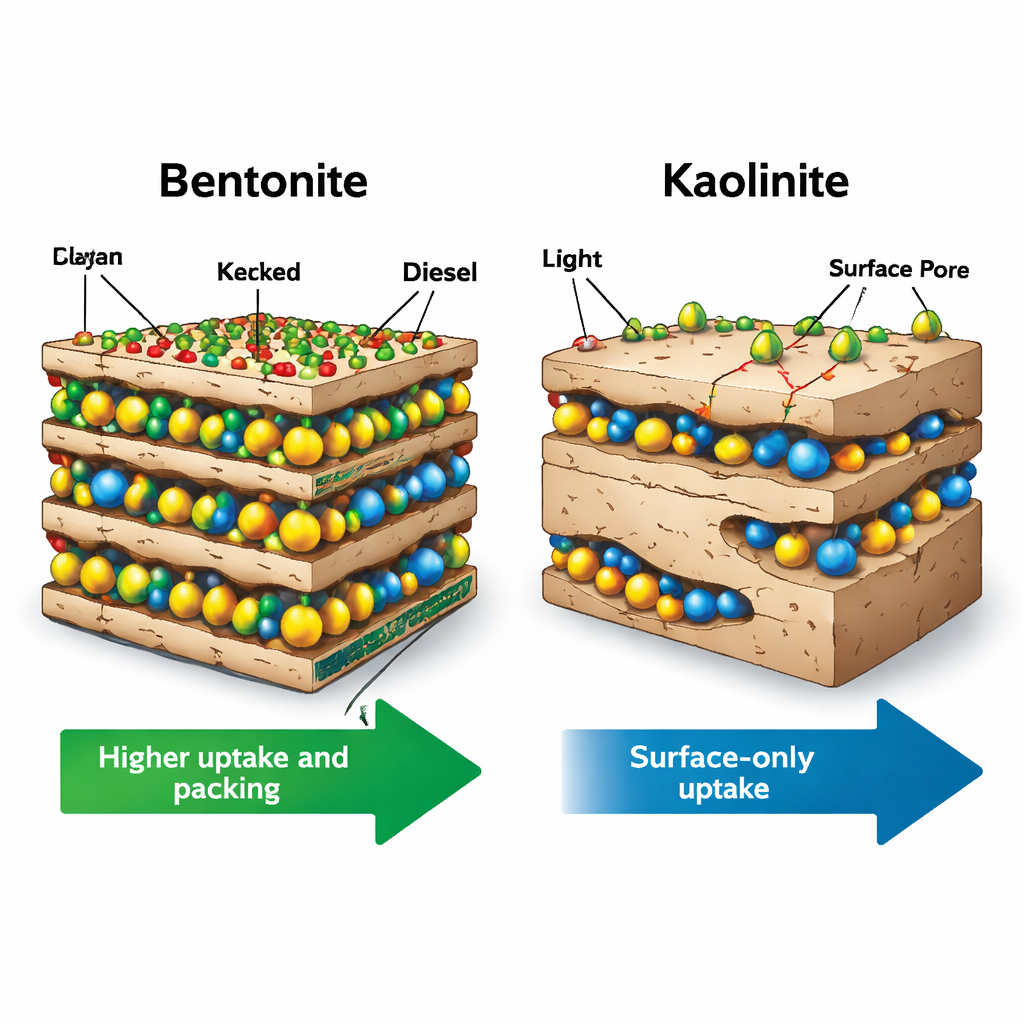
Where the Surfactant Sits Changes What the Clay Can Do
The contrast between the two clays turned out to be stark. In bentonite, detergent surfactants clearly migrated into the interlayer spaces and stayed there, even after washing. Water uptake and buoyancy signals indicated that these confined surfactant molecules flipped their hydrophilic heads toward the clay and their oily tails toward the gallery, turning the interior from water-loving to oil-loving. As a result, bentonite’s ability to hold diesel and kerosene increased by roughly 13–33 percent, and the hydrocarbons packed more densely between the layers. Kaolinite told the opposite story: most surfactant ended up stuck on outer surfaces and edges, with only modest and reversible signs of deeper penetration. This surface coating actually reduced the persistent uptake of heavier diesel components by about 10–30 percent, likely by partially blocking access to already limited internal sites, while the uptake of lighter kerosene molecules barely changed.
What This Means for Cleaning Up Oil Spills
For non-specialists, the core message is straightforward: it is not just how much detergent material a clay captures that matters, but exactly where inside the clay those molecules park. When surfactants from common detergents burrow into the expandable bentonite layers, they act like tiny oil-friendly linings that help pull and pack petroleum hydrocarbons into the clay, boosting its performance as a clean-up material. When similar surfactants merely coat the outside of dense kaolinite particles, they can even make things worse for heavier fuels by clogging the limited entrances. The combined testing method developed here offers a practical way to tell these scenarios apart, helping engineers and environmental scientists screen and tune clay–surfactant combinations for more effective, low-cost remediation of fuel-contaminated soils and waters.
Citation: Khalaj, A., Bahramian, Y., Bahramian, A. et al. Mechanistic insights into modification of clay minerals by detergent-derived surfactants and their impact on petroleum hydrocarbon uptake. Sci Rep 16, 7058 (2026). https://doi.org/10.1038/s41598-026-37171-0
Keywords: oil spill cleanup, clay adsorbents, household detergents, bentonite and kaolinite, petroleum hydrocarbons