Clear Sky Science · en
Multimodal imaging evaluation of hypoxic bone marrow microenvironment and type H vascular injury in diabetes
Why bone health matters in diabetes
People often associate type 1 diabetes with blood sugar and insulin, but the disease quietly reshapes the skeleton as well. Many patients develop weaker bones and face a higher risk of fractures, even when their bone density tests look nearly normal. This study explores what is happening deep inside the bone marrow of diabetes-model rabbits, using advanced medical imaging to watch tiny blood vessels and oxygen levels in action. Understanding this hidden damage could point the way to earlier diagnosis and new treatments that protect bone strength in people living with diabetes.
The bone’s hidden blood highways
Inside long bones like the tibia lies a rich forest of blood vessels that not only feed the bone, but also guide its growth and repair. Among these vessels is a special subtype called “type H” vessels, clustered near the ends of bones where growth plates and active bone remodeling live. These vessels carry more oxygen than others and are closely linked to the formation of new bone. In type 1 diabetes, however, long-term high blood sugar triggers chemical byproducts and oxidative stress that can choke off this finely tuned system. The researchers set out to visualize, in living animals, how these changes unfold in different regions of the bone.
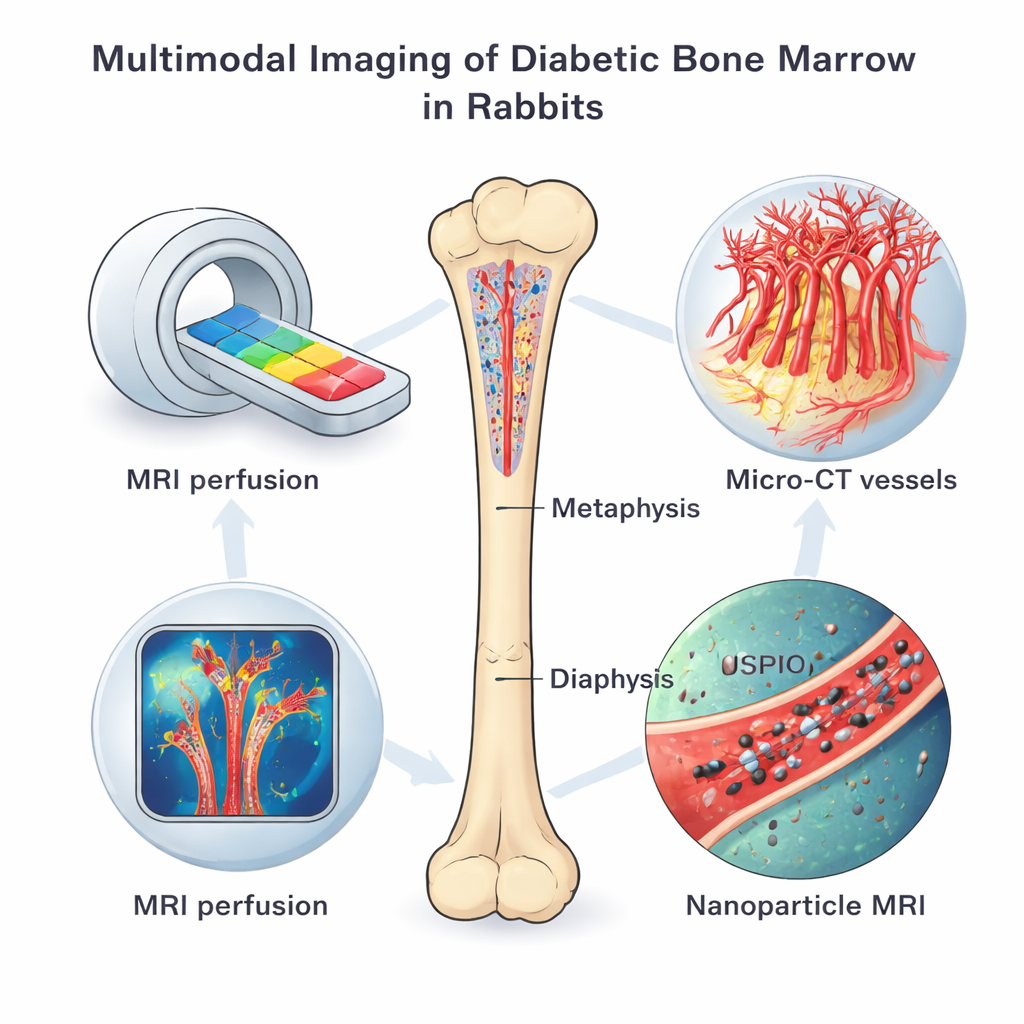
Scanning living bone from multiple angles
The team used a battery of imaging tools on rabbits with chemically induced type 1 diabetes and on healthy controls, all studied four months after diabetes was established. Dynamic contrast-enhanced MRI tracked how quickly a dye moved in and out of bone marrow, revealing how well tiny vessels perfused the tissue and how leaky they were. Another MRI technique, enhanced with special iron-based nanoparticles, showed how actively the vessel lining cells took up particles, a sign of altered metabolism and vessel behavior. After the animals were sacrificed, high-resolution micro-CT scans mapped the three-dimensional network of microvessels, while fluorescent staining, gene tests, and electron microscopy examined vessel types, oxygen-sensing proteins, and cellular damage in fine detail.
Where diabetes hits hardest inside the bone
Across all imaging methods, diabetic rabbits showed bone marrow vessels that were more permeable and abnormal than those of healthy animals. Contrast dye moved faster and lingered longer, and iron nanoparticles were taken up more intensely, especially near the metaphysis—the end region of the bone rich in type H vessels. Micro-CT revealed that diabetic bones had far fewer small vessels and a lower overall vessel volume, to the point that the usual advantage of the metaphysis over the shaft region disappeared. Flow cytometry and fluorescent labeling confirmed that the proportion of type H vessels themselves dropped sharply in diabetes, while new sprouting vessels along the growth plate became scarce. In short, the very vascular niche that normally supports robust bone formation was selectively thinned and damaged.
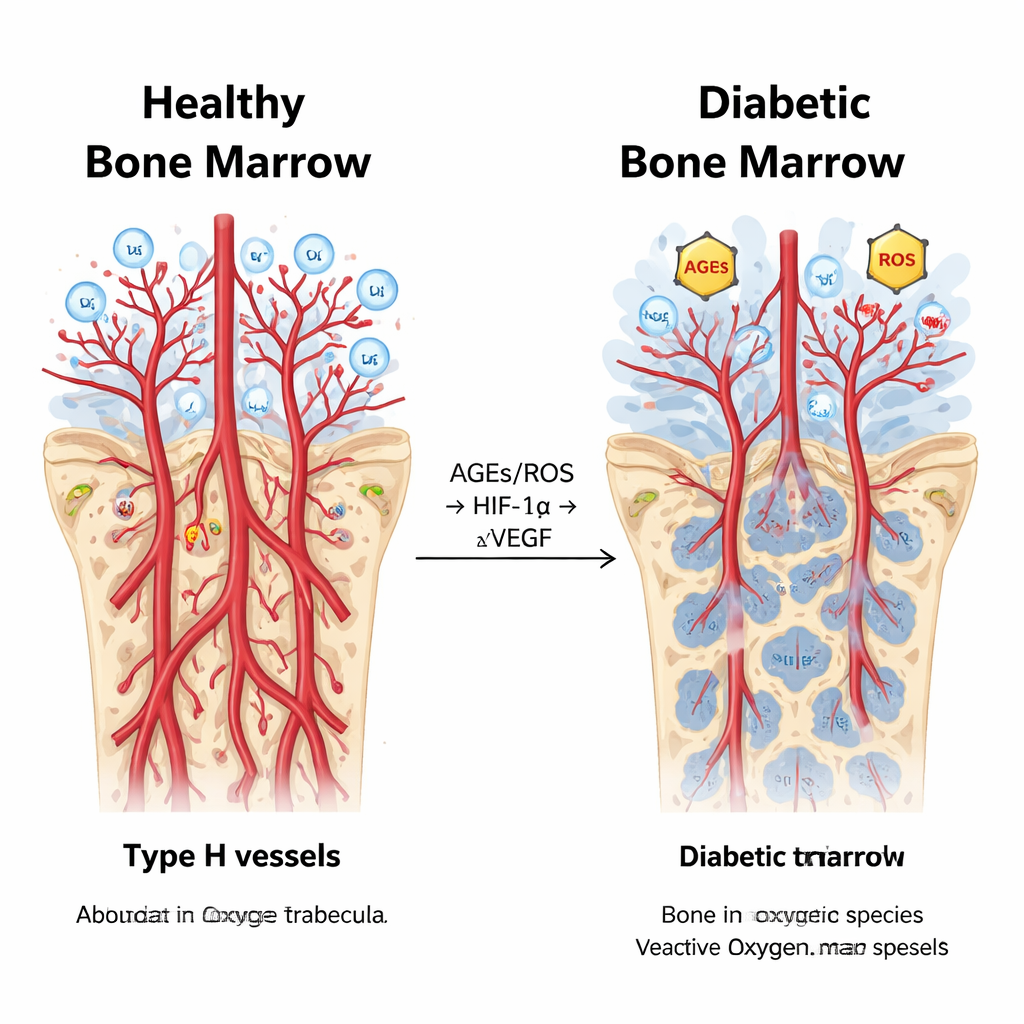
A tangled chemical cascade inside diabetic marrow
Digging deeper, the scientists measured molecules involved in hypoxia—the lack of sufficient oxygen—and in the oxidative stress driven by high blood sugar. Diabetic rabbits had higher levels of advanced glycation end products (AGEs) and reactive oxygen species (ROS), along with stronger activity of the oxygen-sensing protein HIF-1α and its receptor partner AGER, particularly in the metaphysis. Surprisingly, even though HIF-1α usually promotes vessel growth, the key growth signal VEGF was actually suppressed in diabetic bone marrow, and vessel sprouting fell. Statistical analyses tied these threads together: markers of oxidative stress and abnormal perfusion rose and fell in step with HIF-1α, while VEGF and measures of vessel density moved in the opposite direction. This pattern suggests that in chronic diabetes, the normal “oxygen alarm” system is hijacked, driving inflammation and leakiness instead of healthy new vessel and bone formation.
What this means for people with diabetes
Put simply, the study shows that diabetes does not just thin bones in a uniform way; it creates patchy “hot spots” of vessel injury and low oxygen, especially where bone should be strongest and most regenerative. By combining several imaging methods, the researchers could see both the functional leakiness and structural loss of type H vessels, and tie these changes to a specific chemical pathway involving AGEs, ROS, HIF-1α, and VEGF. For patients, this work suggests that future scans might one day detect early bone damage before fractures occur, and that therapies aimed at restoring healthy oxygen signaling and protecting type H vessels could become a new front in preventing diabetic bone disease.
Citation: Lei, H., Wang, K., Li, L. et al. Multimodal imaging evaluation of hypoxic bone marrow microenvironment and type H vascular injury in diabetes. Sci Rep 16, 7206 (2026). https://doi.org/10.1038/s41598-026-37164-z
Keywords: type 1 diabetes, bone marrow, microvascular injury, hypoxia, bone fragility