Clear Sky Science · en
Optimization of parameters for electro Fenton degradation of anion resin by response surface methodology
Cleaning Up Nuclear Wastewater
The nuclear power and research industries rely on special “sponges” called ion exchange resins to strip contaminants from water. When these resins are used up, they themselves become hazardous waste loaded with concentrated pollutants. This study explores a faster, cleaner way to destroy one common type of waste resin, turning it into harmless end products and making the treatment of radioactive wastewater safer and more efficient.
Why Old Filter Beads Are a Big Problem
In nuclear facilities, tiny plastic beads known as anion exchange resins pull unwanted chemicals out of water. Over time, these beads fill up with organic compounds and radioactive elements and must be taken out of service. Traditional treatment options—such as burning, landfilling, or simple chemical neutralization—can leave behind hard-to-handle residues, risk releasing radioactivity, or require long processing times. Wet oxidation, which uses hot, oxygen-rich water to break down the beads, is safer but slow, often needing 8–10 hours and wasting much of the added oxidizing chemicals.
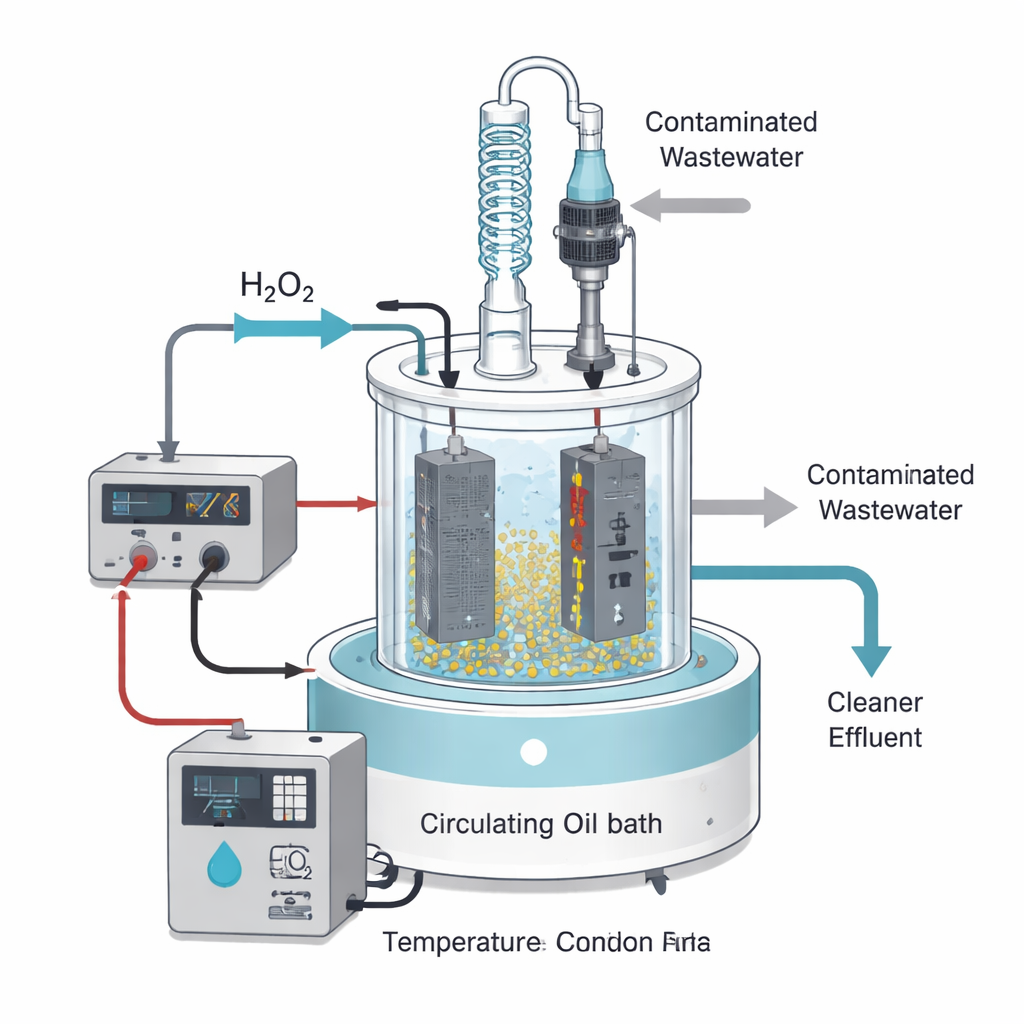
A Power-Assisted Chemical Clean-Up
The researchers focused on an advanced method called the electro-Fenton process, which marries electricity with classic chemical oxidation. In the Fenton reaction, hydrogen peroxide works together with iron salts to generate extremely reactive hydroxyl radicals—short-lived chemical “bulldozers” that tear apart organic molecules. The electro-Fenton version keeps this reaction going more efficiently: a special titanium electrode coated with lead dioxide helps generate radicals and regenerate the active iron form, while a mesh cathode helps recycle iron in the solution. The team treated a real nuclear waste anion resin (ZG CNR170) in a lab-scale reactor equipped with heating, stirring, and a controlled feed of hydrogen peroxide.
Finding the Sweet Spot in the Settings
To turn this promising setup into a practical tool, the scientists systematically varied four key knobs: the acidity (pH) of the mixture, the electric current, the dose of iron salt (FeSO₄), and the rate at which hydrogen peroxide was added. They tracked treatment success by measuring the chemical oxygen demand (COD) of the liquid after the beads dissolved—a standard measure of how much organic pollution remains. First, they changed one factor at a time to see broad trends: moderate electric current sped up degradation but very high current actually hurt performance; adding more iron catalyst helped only up to a point; and feeding hydrogen peroxide too slowly starved the reaction, while overfeeding risked waste and foaming. Acidity mattered as well: the process worked best in strongly acidic conditions, but not at the very lowest pH levels.
Using Statistics to Tune the Process
Next, the team used a statistical tool known as response surface methodology to explore how all four knobs interact at once. They ran 30 carefully planned experiments and built a mathematical model that predicts how much COD remains after 150 minutes under different conditions. This analysis showed that the iron salt dose had the strongest impact on cleanup, followed by the hydrogen peroxide feeding rate, then pH, with current playing a smaller but still meaningful role. Importantly, it highlighted that the ratio between iron and hydrogen peroxide is crucial: too little of either slows the reaction, while too much iron can actually consume the useful radicals instead of letting them attack the resin.
From Beads to Harmless Molecules
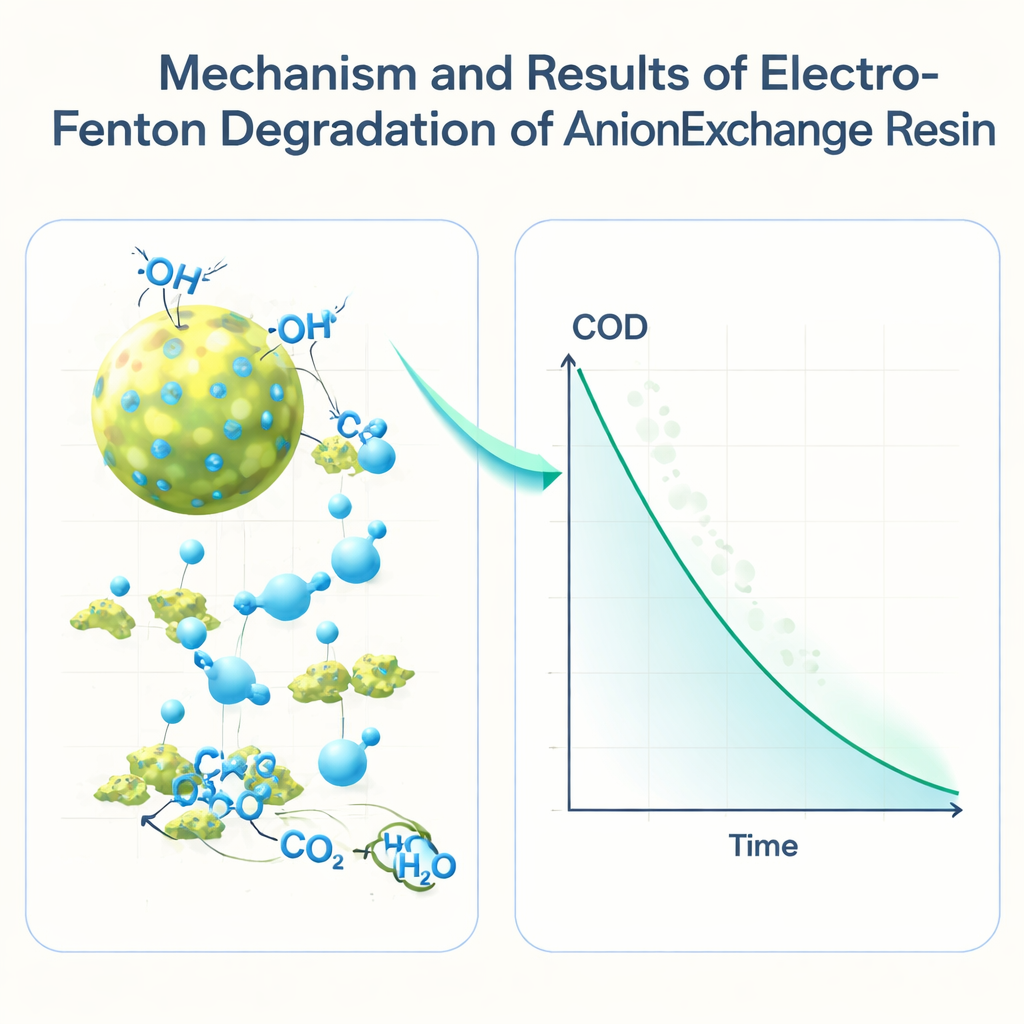
Chemically, the process works by stripping off functional groups from the resin’s structure and then chopping the backbone into ever smaller fragments. The aggressive radicals attack nitrogen-containing groups on the resin surface, then continue to break down the remaining plastic-like skeleton into small organic acids, alcohols, and finally carbon dioxide and water. Under optimized conditions—roughly pH 1.5, a current of 7 amperes, a carefully chosen iron dose, and a steady hydrogen peroxide feed—the resin completely dissolved within 150 minutes, and the remaining COD in the liquid dropped to levels that indicate almost total destruction of organic matter.
What This Means for Nuclear Waste Treatment
For non-specialists, the key message is that the study demonstrates a faster and more efficient way to “chemically burn up” used nuclear filter beads in water, without open flames or extreme conditions. By carefully balancing acidity, electric power, iron catalyst, and hydrogen peroxide, the electro-Fenton process can safely convert these stubborn waste materials into simple, non-toxic molecules in about two and a half hours. This offers a promising route to cleaner, more economical treatment of radioactive wastewater, and the statistical model developed here can help engineers design full-scale systems that minimize chemicals, energy use, and secondary waste.
Citation: Xiang, Q., Hailong, X., Xiliang, G. et al. Optimization of parameters for electro Fenton degradation of anion resin by response surface methodology. Sci Rep 16, 6633 (2026). https://doi.org/10.1038/s41598-026-37155-0
Keywords: electro-Fenton, radioactive wastewater, ion exchange resin, advanced oxidation, waste treatment optimization