Clear Sky Science · en
Biogenic synthesis of iron nanoparticles using Laurencia papillosa: characterization, optimization, and dual applications in heavy metal removal and potential cancer treatment
Seaweed, Tiny Iron Particles, and Big Problems
Dirty water and cancer are two of the world’s most serious health threats, and both are notoriously hard to tackle without creating new problems in the process. This study explores an inventive idea: using a common red seaweed from the Red Sea, Laurencia papillosa, to make extremely small iron particles that can both clean heavy metals from fish-farm wastewater and show promise against cancer cells in the lab. It is a story of turning marine plants into a kind of mini factory for eco-friendly technology.
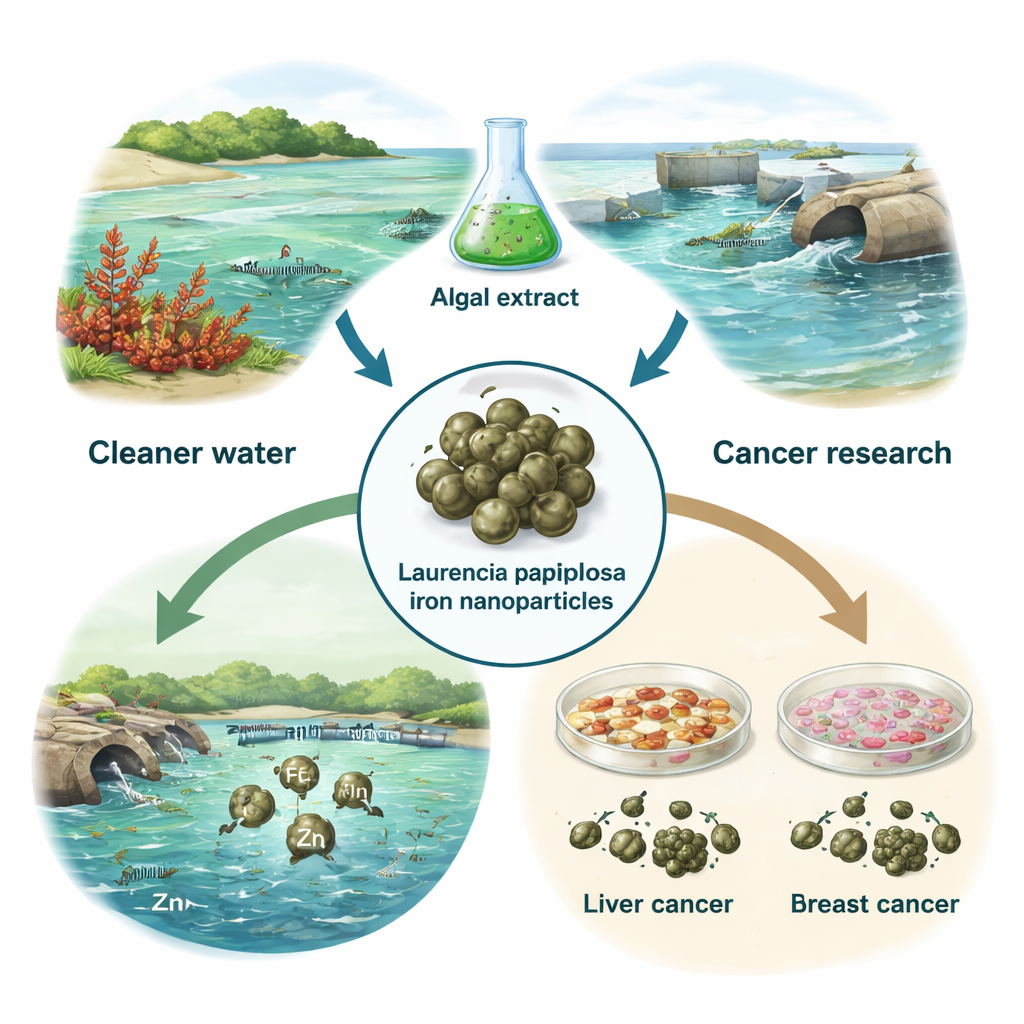
From Red Seaweed to Green Technology
The researchers began by harvesting Laurencia papillosa from Egypt’s Red Sea coast, drying and grinding the seaweed, and soaking it in water to make an extract rich in natural chemicals. These molecules, such as sugars, phenolics, and other plant compounds, act like tiny helpers that can transform dissolved iron salts into solid iron nanoparticles. By simply mixing the algal extract with an iron solution at room temperature, the team produced a brown suspension of iron nanoparticles without using harsh chemicals, high heat, or expensive equipment. This “green synthesis” approach makes the process safer for people and the environment, and potentially cheaper to scale up.
Shaping and Tuning the Nanoparticles
To understand what they had made, the scientists used a suite of imaging and analytical tools. Electron microscopes showed that the particles were nearly spherical and extremely small—about 10 to 20 billionths of a meter across—while other measurements suggested they had a moderately stable surface charge. The particles were not perfectly crystalline, which actually increases their chemical reactivity. The team then fine-tuned the recipe using a statistical method that varied three key factors: acidity (pH), the amount of seaweed extract, and reaction time. They found that a neutral pH of 7, a relatively high algal concentration, and a one-day reaction produced the strongest signal for nanoparticle formation, pointing to conditions that maximize yield and consistency.
Cleaning Heavy Metals from Fish-Farm Water
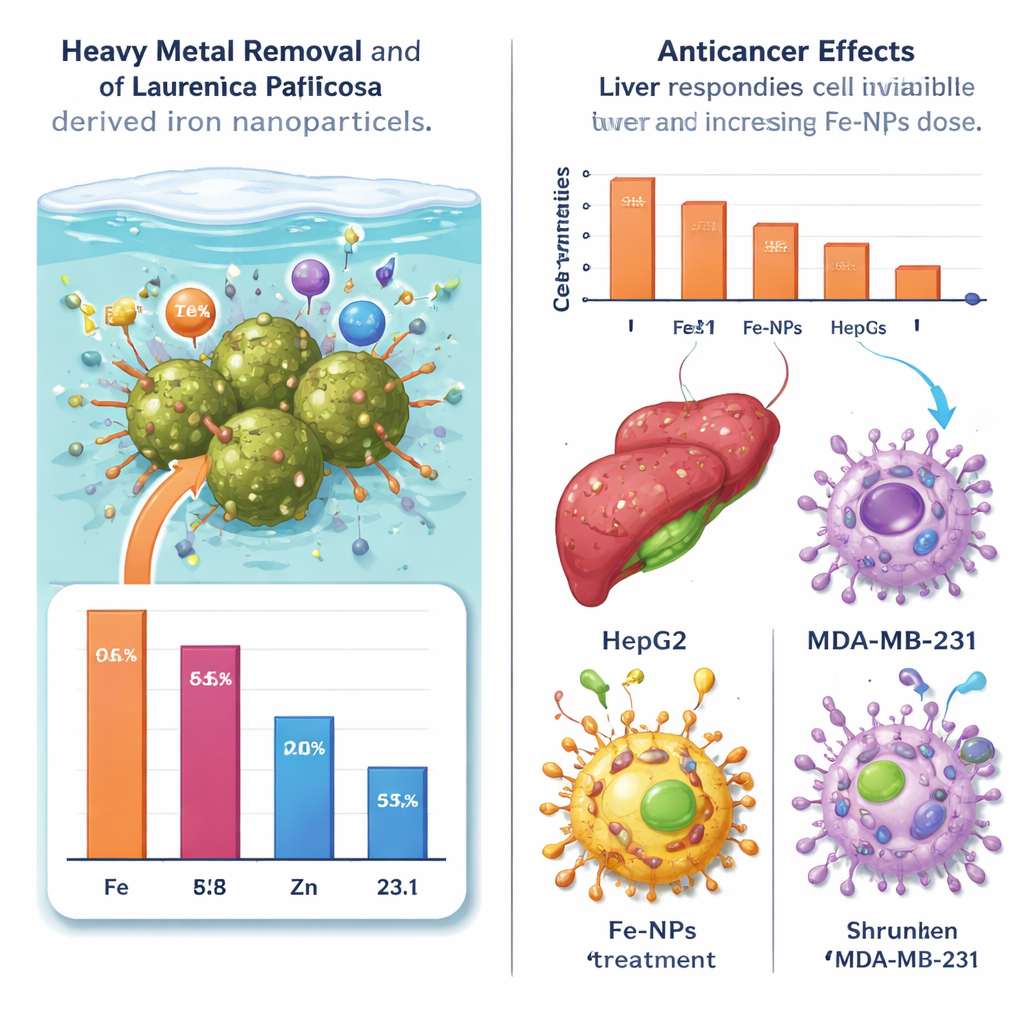
Next, the researchers tested whether these seaweed-derived iron nanoparticles could clean real-world wastewater. They collected effluent from a fish aquaculture facility containing iron (Fe), manganese (Mn), and zinc (Zn) and stirred it with a small amount of nanoparticles. After 90 minutes, they spun out the particles and measured the remaining metals. The results were striking: iron levels dropped by about 96%, manganese by roughly 58%, and zinc by about 23%. These differences reflect how strongly each metal interacts with the nanoparticle surface. Even so, the very high removal of iron—and substantial reduction of manganese—suggests that such particles could help fish farms and other facilities reduce metal pollution with a relatively simple, low-energy treatment step.
Testing Cancer Cells in the Lab
The same particles were then tested against human liver (HepG2) and breast (MDA-MB-231) cancer cell lines grown in dishes. When the researchers exposed these cells to increasing concentrations of nanoparticles for 24 hours, cell survival declined gradually in a dose-dependent way. At low doses, most cells remained alive, but at higher doses, both liver and breast cancer cells showed clear signs of stress: fewer living cells, more rounding and shrinking, and disrupted layers under the microscope. The breast cancer cells appeared slightly more sensitive than the liver cells. Importantly, the overall toxicity was relatively low compared with many conventional chemotherapy drugs, hinting that these particles might be tuned for safer future therapies or used as carriers to deliver drugs more precisely.
What This Could Mean for Health and the Environment
In simple terms, this study shows that a common red seaweed can be turned into a natural factory for iron nanoparticles that do two difficult jobs at once: cleaning heavy metals from polluted water and damaging cancer cells in laboratory tests. The work is still at an early stage—so far only in test tubes and cell cultures—and more research is needed to understand exactly how the particles interact with living tissues and how they behave in real-world water systems. But the findings point toward a future where low-cost, plant-based nanomaterials help provide cleaner water and gentler cancer treatments, showing how solutions to major health and environmental challenges can grow from the sea.
Citation: El Shehawy, A.S., Elsayed, A. & Ali, E.M. Biogenic synthesis of iron nanoparticles using Laurencia papillosa: characterization, optimization, and dual applications in heavy metal removal and potential cancer treatment. Sci Rep 16, 7191 (2026). https://doi.org/10.1038/s41598-026-37139-0
Keywords: green nanotechnology, iron nanoparticles, marine algae, wastewater treatment, cancer nanomedicine