Clear Sky Science · en
Pressure enhanced dual-solid-surface ultra-rapid cooling improves post-thaw recovery in hepatocytes and precision cut liver slices
Why freezing liver tissue matters
Keeping cells and tissues alive outside the body is one of the quiet workhorses of modern medicine. Frozen liver cells help researchers test new drugs, model disease, and may one day repair failing organs. But freezing living material is tricky: if ice crystals grow inside or between cells, they rip delicate structures apart. This study explores a new way to freeze liver cells and thin liver slices so quickly, and under such controlled conditions, that damaging ice crystals barely have time to form.
The problem with ice and chemical protectors
Conventional cryopreservation relies on two imperfect tools: cold and chemicals. As samples are cooled, water tends to form ice, which can puncture membranes and disrupt tissues. To fight this, scientists add cryoprotective agents, such as dimethyl sulfoxide (DMSO), that help water solidify into a glass-like state instead of crystalline ice. However, at the high concentrations typically used, these agents can themselves poison cells or cause damaging swelling and shrinking as they move in and out of the tissue. The authors’ goal was to cut back on the amount of DMSO needed while still avoiding harmful ice, making liver cell and tissue banking safer and more practical.
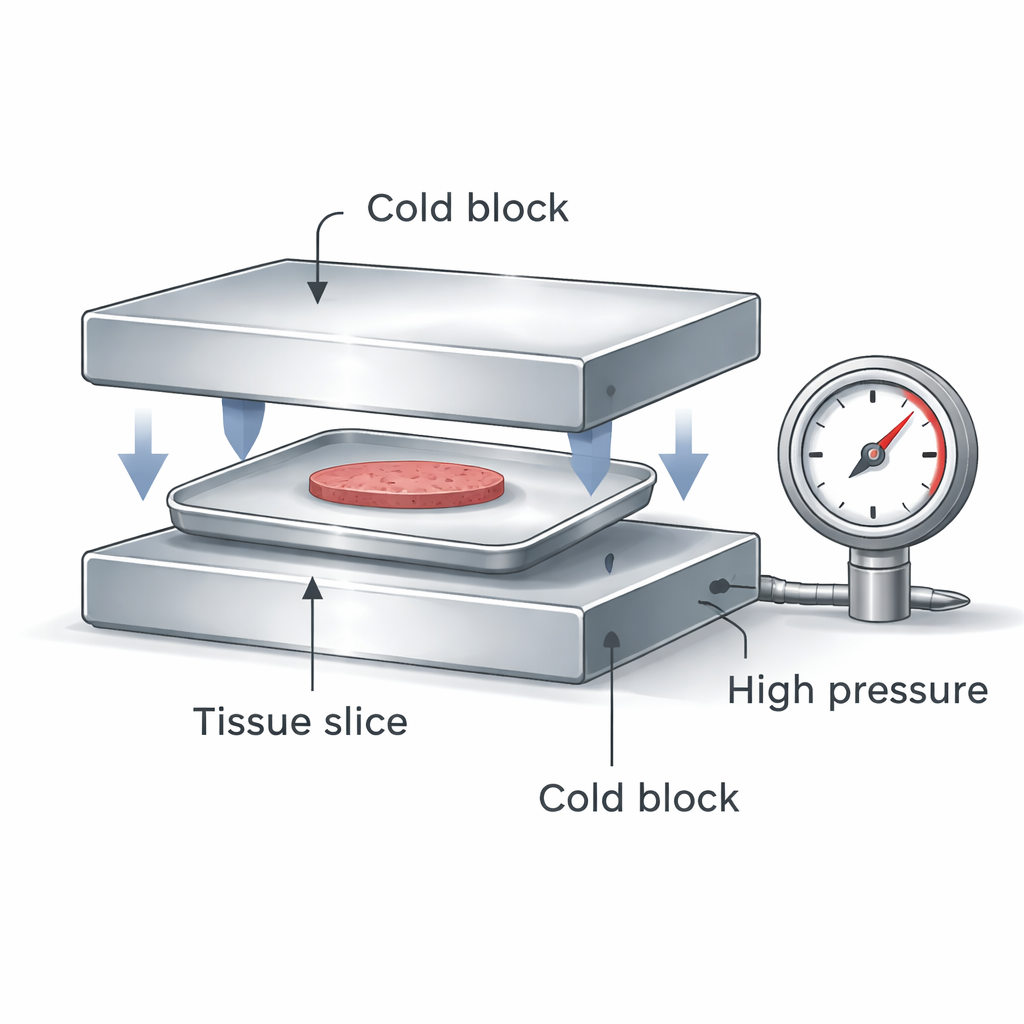
A new way to press and cool at the same time
The team designed a compact device that sandwiches a sealed, flat aluminum container holding liver cells or precision-cut liver slices between two extremely cold metal blocks. When the blocks press together, they do two important things at once: they create high pressure inside the sealed container and they pull heat out of the sample from both the top and the bottom. High pressure shifts the behavior of water so that ice forms at lower temperatures and the solution becomes glassy more easily. Double-sided contact gives a much faster and more uniform cooling rate than simply plunging a plastic vial into liquid nitrogen, and the sealed container keeps the sample isolated from contamination.
Finding the sweet spot for safety and survival
To see whether this approach was gentle enough for living material, the researchers first checked that the aluminum container itself did not harm liver-derived HepG2 cells during the short handling times used. Cell survival stayed essentially unchanged. They then exposed these cells to different DMSO levels before freezing and confirmed a familiar trade-off: more DMSO better suppresses ice but becomes clearly toxic at 30%, while 20% remains largely tolerable. For mouse liver slices, they tested how much pressure the tissue could withstand in the absence of freezing. Brief bursts up to 150 megapascals had little effect on viability, but 200 megapascals caused about a 30% drop, marking an upper limit for safe operation.
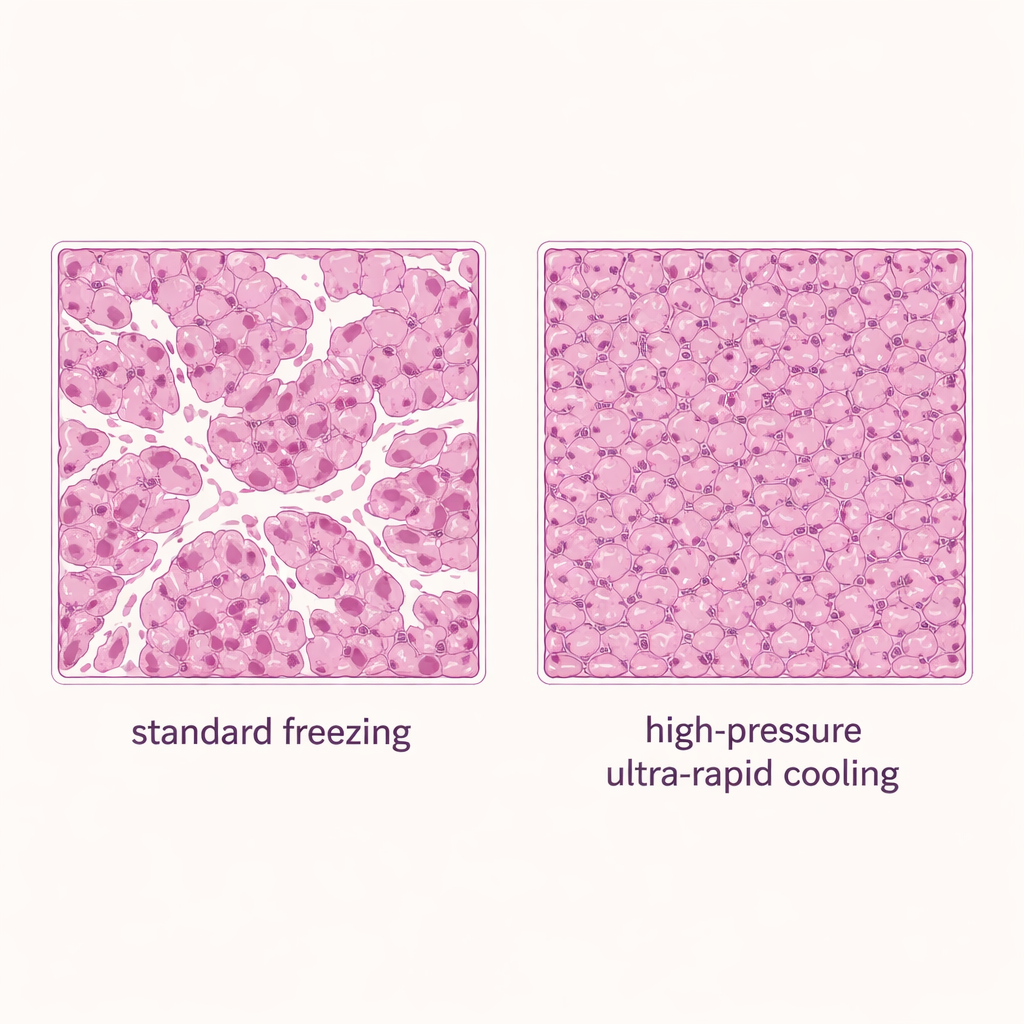
Better-preserved liver slices after thawing
With safe ranges established, the authors compared three freezing methods for liver slices: standard immersion of the sealed container into liquid nitrogen (a convection, or fluid-based, approach), solid-surface cooling with no added pressure, and solid-surface cooling with high pressure at 150 megapascals. All groups used 20% DMSO. The pressure-assisted method delivered the best results: about 80% of the tissue’s original viability was retained, outperforming both standard freezing and solid-surface cooling alone. Microscopy of stained tissue slices supported these findings. Samples frozen by conventional immersion showed many open white spaces—signatures of ice damage—whereas pressure-enhanced solid-surface cooling produced denser, more intact tissue with far fewer gaps.
What this means for future tissue banking
Overall, the study shows that squeezing a sealed, aluminum-encased liver slice between two ultra-cold blocks under carefully controlled high pressure can greatly improve how well the tissue survives thawing. By combining faster, more even cooling with pressure-altered water behavior, the method limits the growth of damaging ice while still using a moderate, clinically familiar concentration of DMSO. For a lay reader, the takeaway is that smarter engineering—how we package and cool tissue—can be just as important as the chemicals we add. This dual-sided, high-pressure approach could become a stepping stone toward safer, more reliable banking of liver tissue and, eventually, other organs for research and therapy.
Citation: Amini, M., Benson, J.D. Pressure enhanced dual-solid-surface ultra-rapid cooling improves post-thaw recovery in hepatocytes and precision cut liver slices. Sci Rep 16, 5994 (2026). https://doi.org/10.1038/s41598-026-37136-3
Keywords: cryopreservation, liver tissue, vitrification, high pressure cooling, ice-free freezing