Clear Sky Science · en
Senescence-associated LncRNAs TRMP and TRMP-S promote gastric cancer by activating IGFL4
Why this research matters
Stomach (gastric) cancer is still one of the world’s deadliest cancers, largely because it is often found late and can be hard to treat. This study uncovers how two little-known genetic molecules, called TRMP and TRMP-S, help stomach tumors grow and spread. By revealing the chain of events they trigger inside cancer cells and in the tumor’s immune environment, the work points to new ways doctors might one day diagnose patients earlier and design more effective, targeted treatments.
Hidden messages in our DNA
Most people think of genes as blueprints for proteins, but our DNA also produces long noncoding RNAs (lncRNAs) that do not make proteins yet strongly influence how cells behave. TRMP and its shorter splice variant TRMP-S are two such lncRNAs. They were previously linked to aging and cell cycle control in lung and colon cells, but their role in stomach cancer was unknown. Because lncRNAs can act as markers for diagnosis and targets for therapy, the authors set out to discover whether TRMP and TRMP-S push gastric cells toward cancer and how they might interact with other molecules to do so.
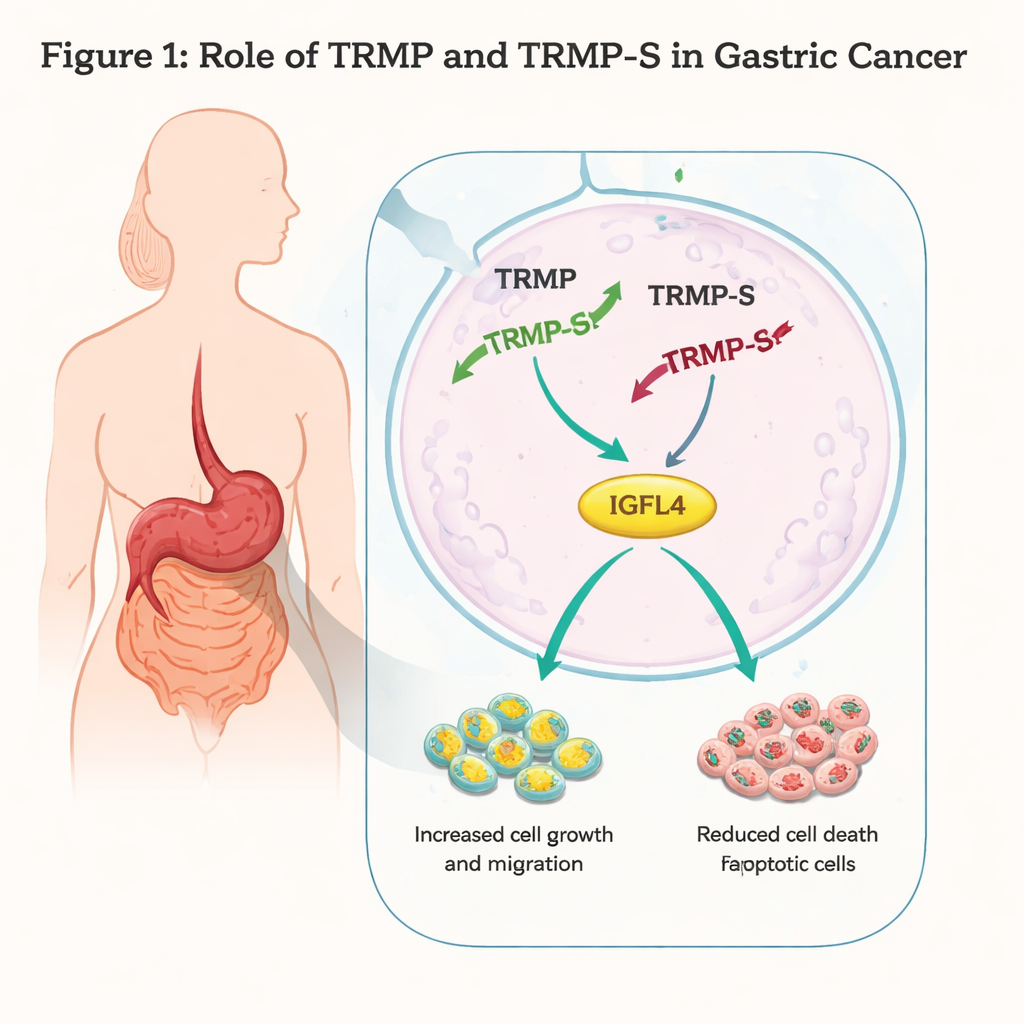
Turning off a cancer accelerator
The researchers worked with two human gastric cancer cell lines, AGS and MKN45, and used genetic tools to reduce the levels of TRMP-S and TRMP. When they silenced TRMP-S, cancer cells grew more slowly, formed fewer colonies, and were less able to migrate through artificial barriers or close wounds in a dish. Flow cytometry and protein analyses showed that more cells were undergoing programmed cell death (apoptosis) and that the cell cycle was being halted in the G1 phase, a key “checkpoint” before cells copy their DNA. Levels of p73, a member of the p53 tumor-suppressor family, rose when TRMP-S was lost, consistent with stronger cell-cycle control and more cell death. These results together indicate that TRMP-S normally acts like an accelerator pedal for gastric cancer cells.
A key partner: the IGFL4 signal
To understand how TRMP and TRMP-S exert their effects, the team mined large patient datasets from The Cancer Genome Atlas and a Korean gastric cancer cohort. They looked for genes that were both abnormally high in tumors and positively linked to TRMP levels, then built a six-gene “risk model” that predicted patient survival—higher scores were tied to worse outcomes. Among these genes, one stood out: IGFL4, a member of a family related to insulin-like growth factors, which are known to drive growth and survival in many cancers. In gastric cancer cells, knocking down TRMP-S sharply lowered IGFL4’s RNA and protein levels, while an RNA immunoprecipitation assay showed that both TRMP and TRMP-S physically bind IGFL4 protein. Directly reducing TRMP had the same dampening effect on IGFL4, suggesting that these lncRNAs help stabilize or support the production of this growth-promoting signal.
Slowing spread and reshaping the immune scene
When the researchers used small interfering RNAs to silence IGFL4 itself, gastric cancer cells slowed their growth, migrated less, and showed weaker ability to invade through membranes and close wounds. Analysis of patient tumor data revealed that IGFL4 levels were significantly higher in gastric tumors than in normal stomach tissue, and elevated across many other cancer types as well. Importantly, tumors with high IGFL4 expression showed distinct patterns of immune-cell infiltration: more undifferentiated macrophages (M0) and fewer plasma cells, monocytes, eosinophils, and neutrophils. Computational immune scores suggested that high-IGFL4 tumors might respond better to modern immunotherapies that block immune checkpoints, indicating IGFL4 could help identify patients who are good candidates for these treatments.
A small RNA that pushes back
The study also uncovered a counterbalance to this cancer-promoting pathway. By correlating microRNA and gene expression in patient data, the authors pinpointed miR-129-5p as a small RNA that is negatively linked to both TRMP and IGFL4. In gastric cancer cells, adding miR-129-5p reduced IGFL4 levels and slowed growth, whereas blocking miR-129-5p increased IGFL4 and sped up proliferation. Another candidate microRNA, miR-4739, did not measurably affect IGFL4 and was ruled out. These results support a regulatory chain in which TRMP and TRMP-S, miR-129-5p, and IGFL4 form a network that fine-tunes how aggressively gastric cancer cells grow and how they interact with their surroundings.
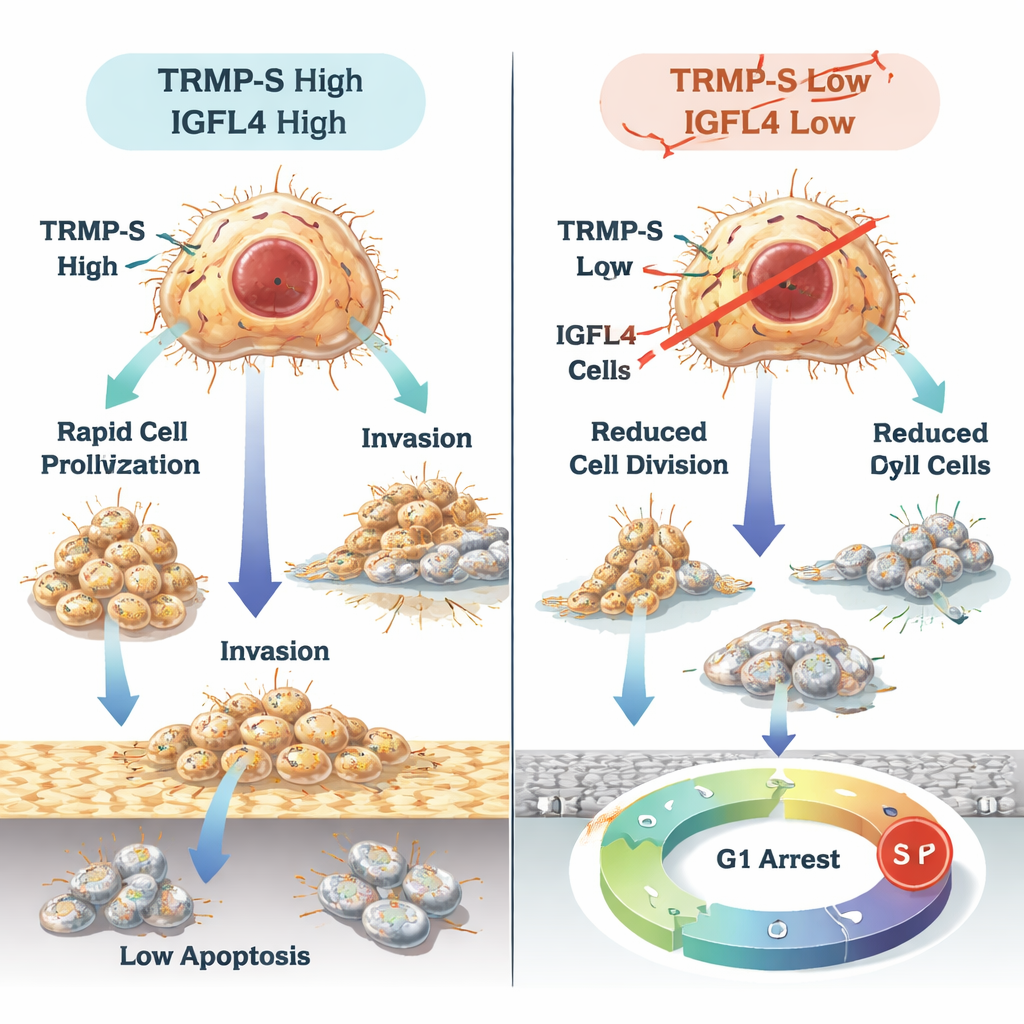
What this could mean for patients
In plain terms, this work shows that TRMP and TRMP-S act as behind-the-scenes directors that switch on IGFL4 and help stomach cancer cells grow, spread, and evade natural braking mechanisms. At the same time, the small RNA miR-129-5p acts as a brake on IGFL4, and IGFL4 itself helps shape how immune cells gather around tumors and how well therapies that awaken the immune system might work. Although more research and clinical testing are needed, TRMP, TRMP-S, IGFL4, and miR-129-5p together represent a promising set of markers for predicting prognosis and potential new targets for drugs aimed at slowing or stopping gastric cancer.
Citation: Zhang, M., Mi, Y., Li, F. et al. Senescence-associated LncRNAs TRMP and TRMP-S promote gastric cancer by activating IGFL4. Sci Rep 16, 6740 (2026). https://doi.org/10.1038/s41598-026-37106-9
Keywords: gastric cancer, long noncoding RNA, TRMP, IGFL4, tumor microenvironment