Clear Sky Science · en
Identification of RBX1 as a regulator of LIPT1 transcription and its role in copper-induced cell death in GBM cells
Why copper and brain tumors matter
Glioblastoma multiforme is one of the deadliest forms of brain cancer, with most patients surviving only a short time after diagnosis despite aggressive surgery, radiation, and chemotherapy. This study explores an unexpected ally against these tumors: the metal copper. The researchers examine a newly recognized form of cell death triggered by copper and identify a molecular pathway that may help doctors harness this process to weaken tumors and potentially boost the immune system’s attack on cancer cells.
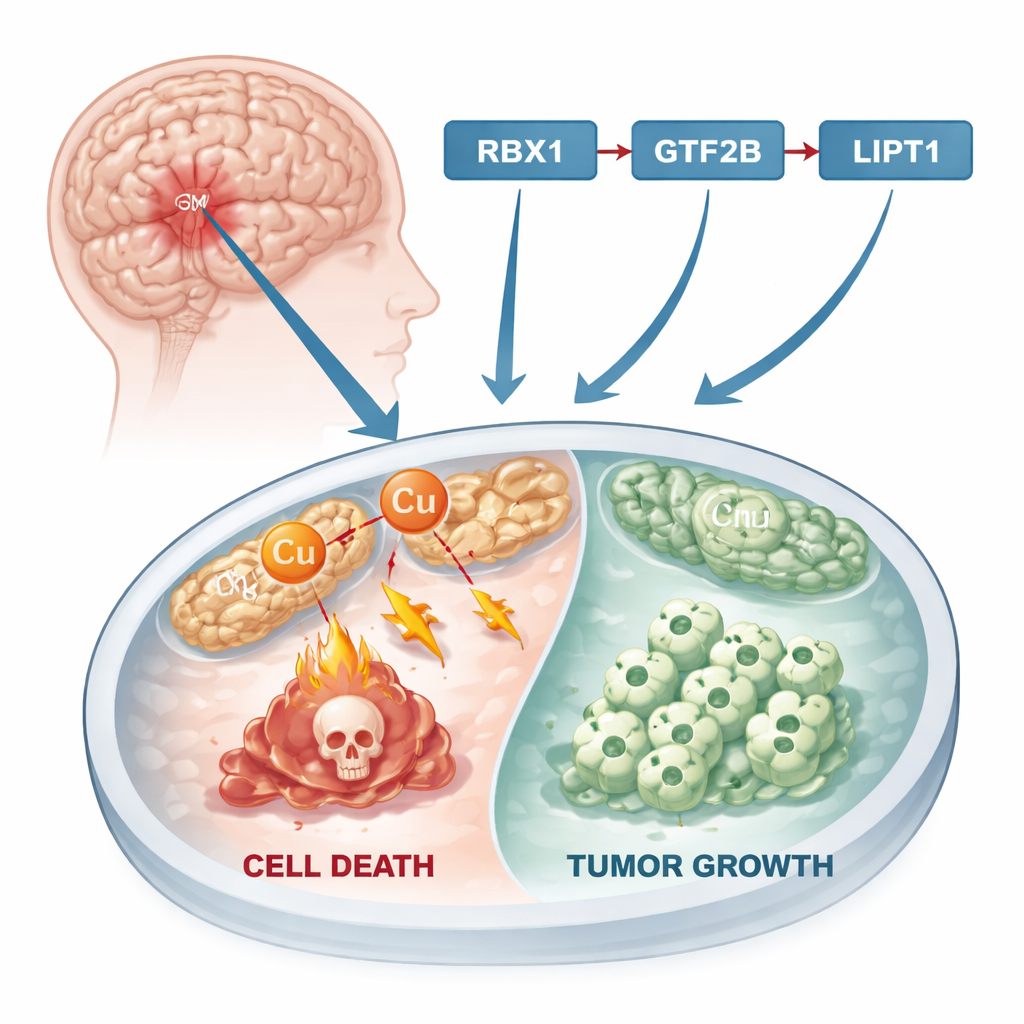
A new way for cells to die
For decades, cancer research has focused on familiar forms of cell death such as apoptosis, in which damaged cells quietly shut themselves down. Recently, scientists discovered a different route called copper-induced cell death, or “cuproptosis.” In this process, excessive copper builds up inside a cell’s energy factories, the mitochondria. There it disrupts key proteins, causing toxic clumps to form and ultimately killing the cell. Because many tumors have altered metal metabolism and higher copper levels than normal tissues, cuproptosis offers a potential pressure point: if researchers can push tumor cells toward this copper-driven demise, they might slow or stop cancer growth.
Spotlighting a key gene in glioblastoma
The authors began by surveying a group of genes already known to be involved in cuproptosis and asked how they behave in glioblastoma samples versus normal brain tissue. One gene in particular, called LIPT1, stood out. It was more active in glioblastoma tissue and in several glioblastoma cell lines than in normal brain cells. Importantly, patients whose tumors had higher LIPT1 activity tended to go longer without the cancer coming back after treatment. High LIPT1 levels were also linked with greater presence of CD8 T cells, the immune system’s frontline cancer killers, suggesting that this gene might help make the tumor environment more vulnerable to immune attack.
What happens when the copper death switch is turned down
To test whether LIPT1 truly affects copper-driven cell death, the team exposed glioblastoma cells to a copper-carrying drug combination that reliably triggers cuproptosis. They then used genetic tools to reduce LIPT1 levels. When LIPT1 was suppressed, tumor cells became more resistant to copper-induced killing, survived better, and showed stronger abilities to migrate and invade—behaviors associated with more aggressive cancer. In mixed cultures of tumor cells and human CD8 T cells, lowering LIPT1 also reduced the release of immune signaling molecules and made the cancer cells harder for T cells to destroy. Together, these experiments indicate that LIPT1 heightens sensitivity to copper-induced death and supports anti-tumor immune activity.
Following the chain of command up to RBX1
The next challenge was to understand why LIPT1 is turned up in glioblastoma. By combining several large gene and protein databases, the researchers identified a transcription factor—a kind of master switch for gene activity—called GTF2B that binds near the LIPT1 gene and likely boosts its expression. They then asked what controls GTF2B itself. A second protein, RBX1, emerged as a strong candidate. RBX1 is part of the cell’s tagging-and-disposal system that marks other proteins for breakdown. In glioblastoma cells, RBX1 levels were lower than in normal brain cells, while GTF2B and LIPT1 were higher. Laboratory assays showed that RBX1 can attach small “destroy me” tags to GTF2B, leading to its degradation; when RBX1 was increased, GTF2B levels and LIPT1 activity fell, and blocking the cell’s degradation machinery reversed this effect.
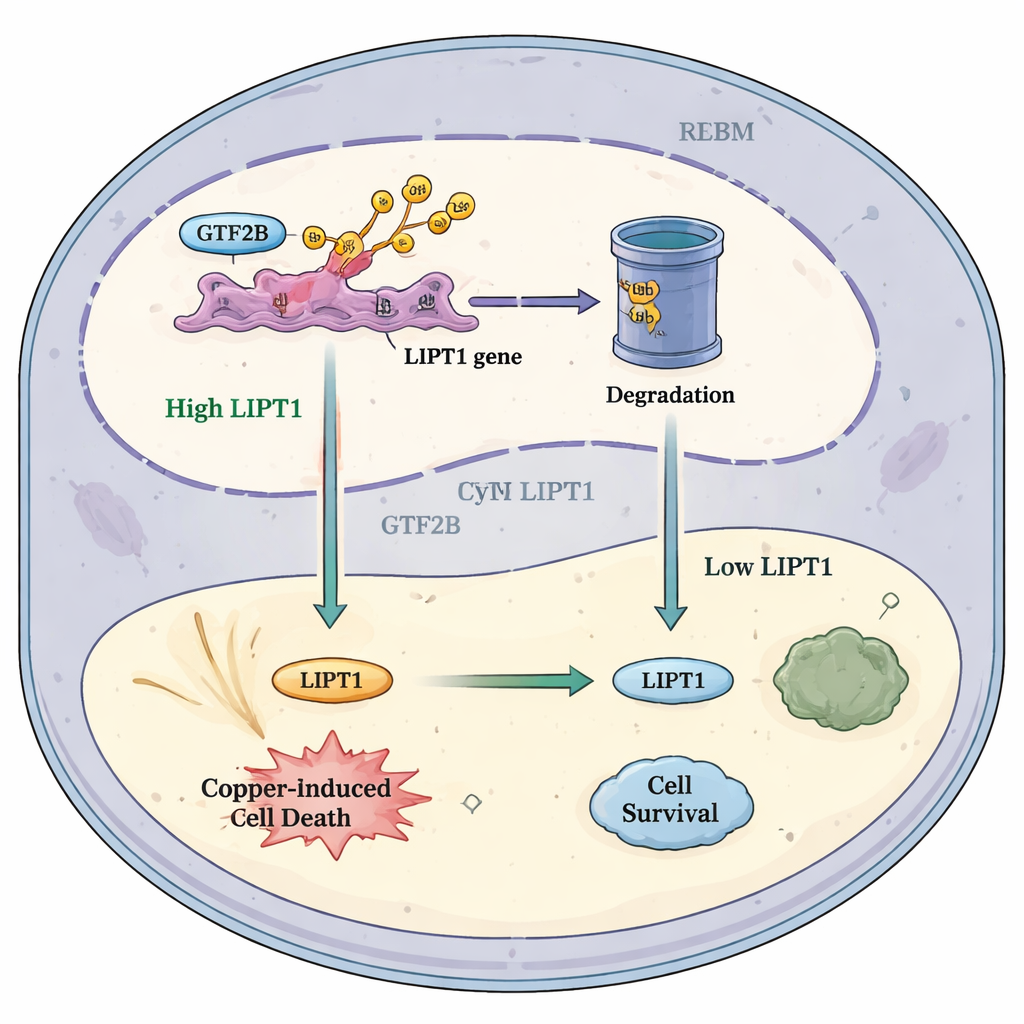
How this pathway could help future treatments
Putting these findings together, the authors propose a simple model: in healthy conditions, RBX1 keeps GTF2B in check, limiting how strongly it activates the LIPT1 gene. In glioblastoma, reduced RBX1 means less GTF2B is broken down. Extra GTF2B then boosts LIPT1, increasing the tumor cells’ sensitivity to copper-induced death and drawing in more cancer-fighting immune cells. This model suggests that carefully tuning this RBX1–GTF2B–LIPT1 pathway, possibly alongside copper-targeting drugs and immunotherapies, might tip the balance inside brain tumors toward self-destruction. While much work remains before this idea can reach patients, the study highlights a promising intersection of metal biology, gene regulation, and cancer immunology that could open new therapeutic avenues for one of the most stubborn cancers.
Citation: Zeng, J., Liu, J., Hua, S. et al. Identification of RBX1 as a regulator of LIPT1 transcription and its role in copper-induced cell death in GBM cells. Sci Rep 16, 6837 (2026). https://doi.org/10.1038/s41598-026-37105-w
Keywords: glioblastoma, copper-induced cell death, LIPT1, tumor immunology, RBX1 pathway