Clear Sky Science · en
Integrating machine learning and physics-based modeling for predictive design of gemcitabine-loaded nanocomposites
Smarter Cancer Medicines by Design
Cancer drugs like gemcitabine can save lives, but they often behave more like blunt tools than precision instruments: they break down quickly, miss their targets, and cause harsh side effects. This study explores how computers can help scientists "pre-test" new tiny drug carriers on a screen, instead of only in the lab, to make gemcitabine treatments more effective, longer lasting, and potentially safer for patients.
Why Tiny Carriers Matter
Traditional chemotherapy floods the body with drug molecules that circulate everywhere, not just in tumors. Nanocomposites—engineered particles thousands of times smaller than a grain of sand—offer a way to package gemcitabine so it is protected in the bloodstream and delivered more directly to cancer cells. Two numbers largely decide whether a design is useful. Loading efficiency tells us how much drug ends up inside each particle, while encapsulation efficiency measures what fraction of the starting drug is successfully trapped rather than wasted. High values for both mean fewer injections, less carrier material, and a better chance of hitting the tumor hard without overwhelming the rest of the body.
From Trial-and-Error to Data-Guided Design
Designing these nanocarriers by hand is like trying to tune a radio with hundreds of knobs—particle size, surface charge, materials, coatings, and preparation methods all interact in complex ways. Until now, researchers mostly changed one or two settings at a time and measured the result, a slow and expensive cycle that reveals only a small slice of the full picture. In this work, the authors gathered 59 carefully vetted gemcitabine nanocarrier recipes from the scientific literature and enriched them with additional, physics-based examples generated on the computer. This combined dataset let them test several machine learning approaches—computer programs that learn patterns from data—to predict loading and encapsulation efficiencies from the design choices alone.
Letting Physics Steer the Algorithms
Most machine learning systems are powerful curve-fitters: they can spot patterns but do not know when an answer breaks the laws of nature. To avoid unrealistic predictions, the researchers built a hybrid method that blends data with basic physical rules about how drug molecules move, bind, and stay balanced in a closed system. In their framework, any prediction that would imply, for example, creating more drug than was originally added, or ignoring how molecules diffuse through a material, is gently punished during training. This "physics-informed" approach nudges the model toward answers that make chemical and physical sense, especially in regions of design space where real measurements are scarce.
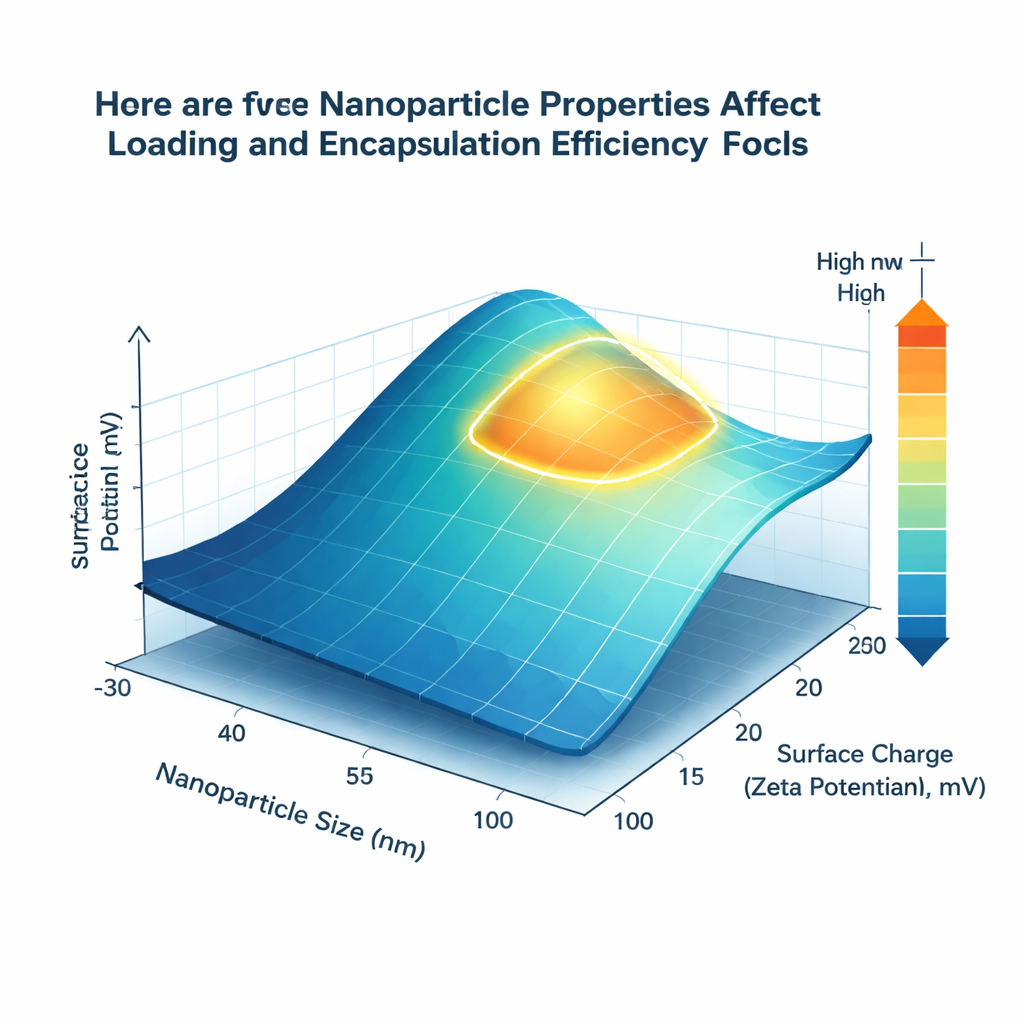
What the Models Discovered
Among the tested techniques, an advanced ensemble algorithm called XGBoost gave the most accurate forecasts, closely matching reported experimental results for both key efficiencies. But beyond accuracy, the team wanted understandable design rules. Using a method called SHAP, which ranks how strongly each factor pushes the prediction up or down, they found that particle size and surface charge consistently dominate performance. Nanocarriers between about 80 and 150 nanometers in diameter offered the best balance between surface area and internal volume, boosting both loading and encapsulation. A mildly positive surface charge, in the range of +15 to +25 millivolts, was linked to better trapping of gemcitabine, likely because the positively charged carrier surfaces interact favorably with the drug’s negatively charged groups.
A Digital Map for Future Cancer Treatments
The end result is not a finished medicine but a powerful planning tool. The study delivers a kind of "design map" that points researchers toward nanocarrier recipes most likely to hold and protect gemcitabine well, while avoiding millions of unpromising combinations. The authors stress that their predictions still need to be confirmed in fresh lab and animal studies, and that their dataset—though carefully assembled—is modest in size. Even so, this physics-aware machine learning framework shows how computers can help narrow the search for better cancer drug carriers, cutting costs and speeding the journey from ideas to more precise, patient-friendly therapies.
Citation: Rahdar, A., Fathi-karkan, S. & Shirzad, M. Integrating machine learning and physics-based modeling for predictive design of gemcitabine-loaded nanocomposites. Sci Rep 16, 6268 (2026). https://doi.org/10.1038/s41598-026-37098-6
Keywords: nanomedicine, gemcitabine, drug delivery, machine learning, nanoparticles