Clear Sky Science · en
MCT1 as a critical regulator of insulin signaling, energy homeostasis and podocyte function
Why kidney cells’ fuel choices matter
Our kidneys quietly filter hundreds of liters of blood every day, and much of that work depends on tiny cells called podocytes that wrap around the kidney’s filters. These cells must constantly reshape themselves to keep protein in the blood and waste in the urine. This study explores how podocytes get the energy they need, what happens when one of their major “fuel doors” is blocked, and why that might matter for common conditions like diabetes and kidney disease. 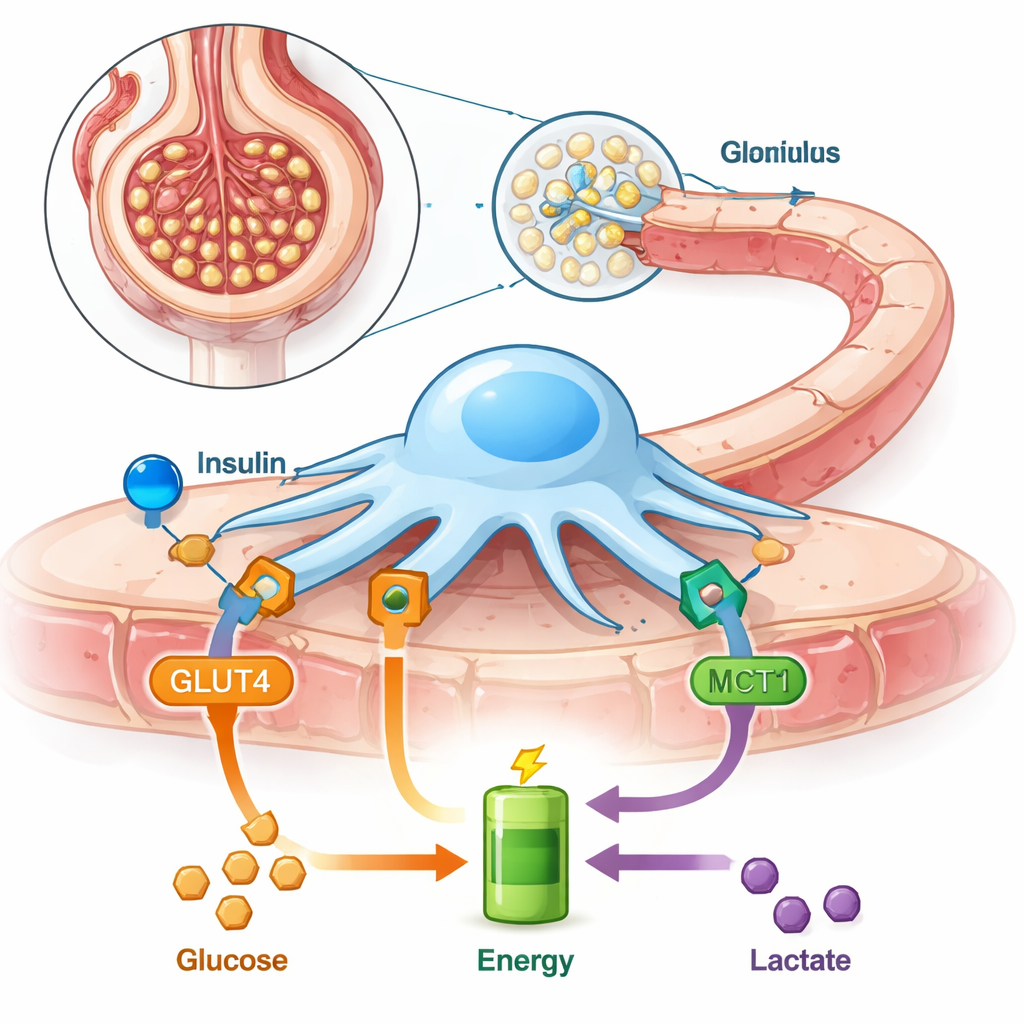
Gatekeeper cells at the filter
Podocytes sit on the outer side of each kidney filter, sending out delicate foot-like extensions that interlock to form a final barrier before urine is produced. Because they are always adjusting their shape, they burn a lot of energy. Unlike many other cells, podocytes rely heavily on breaking down sugar without oxygen (a pathway called glycolysis) and they are especially responsive to insulin, which tells them to pull more sugar in from the blood through a transporter called GLUT4. The authors had previously shown that podocytes can also run on lactate, a small molecule often dismissed as “waste,” hinting that these cells are more flexible than once thought.
The lactate doorway: MCT1
Lactate moves into and out of cells through special transport proteins. One of the most important is monocarboxylate transporter 1 (MCT1), which can carry lactate into cells to be burned for energy. In this study, researchers used rat podocytes grown in dishes and blocked MCT1 with a chemical inhibitor. They then asked how this affected insulin’s ability to drive sugar uptake, how much energy the cells could make, how their internal scaffolding looked, and how “leaky” the filter became to a large blood protein called albumin. They also tested whole rat kidney filters (isolated glomeruli) to see how lactate and MCT1 blocking changed protein leakage in a more intact system.
When the fuel door closes
Blocking MCT1 had several striking effects. First, it reduced how much glucose the podocytes took up, both under resting conditions and after insulin stimulation, and it weakened a key switch in the insulin pathway (a protein called Akt) without disturbing the insulin receptor itself. At the same time, the normal rearrangement of the GLUT4 transporter toward the cell surface was blunted. Measurements of cellular metabolism showed that total energy production fell when MCT1 was blocked, and the cells shifted away from glycolysis toward more oxygen-based burning in mitochondria. Even when insulin or lactate were added, this energy shortfall was not fully corrected, suggesting that MCT1 is central to how podocytes balance their energy sources. 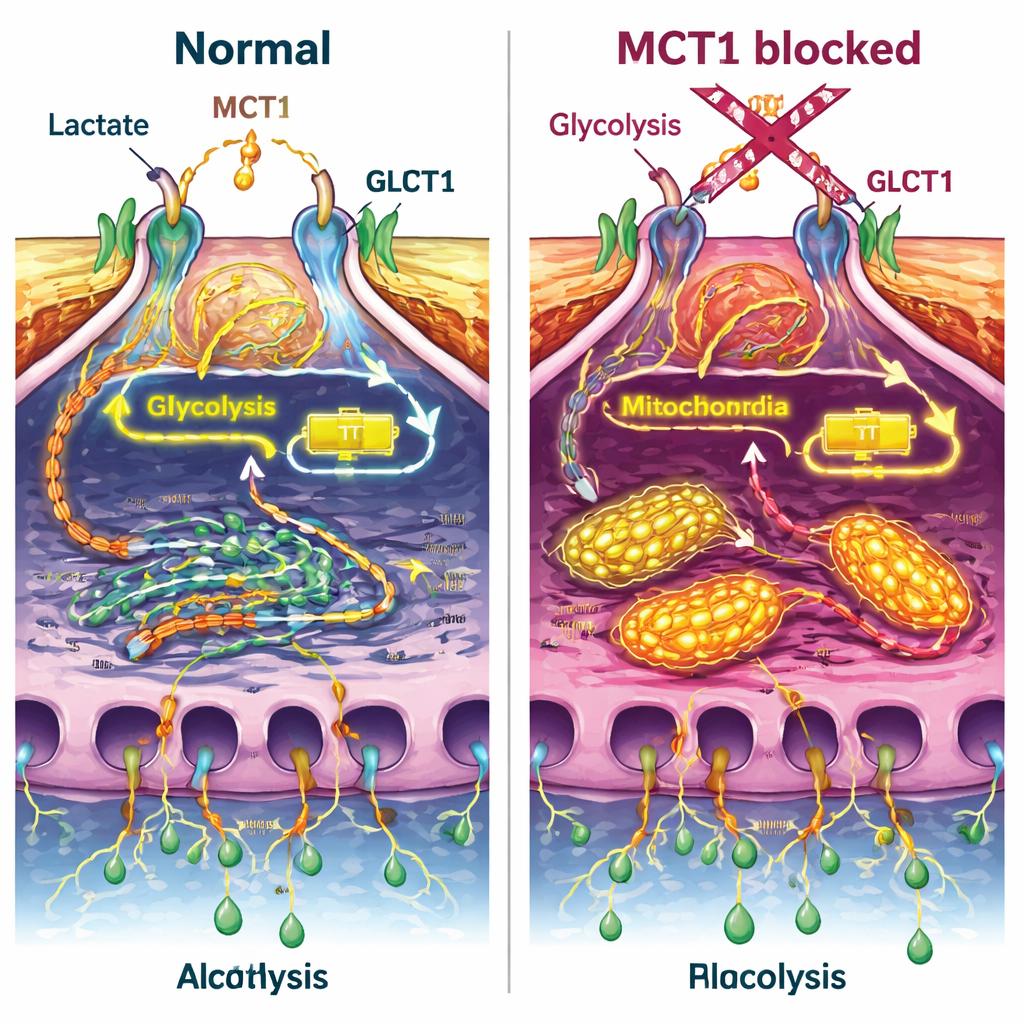
Leaky filters and stressed scaffolds
Energy changes were closely linked to physical damage. When MCT1 was blocked, podocyte layers in culture let more albumin pass through, similar to the effect of insulin alone. Lactate itself also made the barrier leakier, and combining lactate or insulin with MCT1 blockade kept permeability high. Inside the cells, the actin fibers that form their shape became more bundled and disorganized, a pattern associated with loss of the fine foot processes that make up the filter. Another key protein, nephrin, which helps tie the filter together and also supports insulin’s actions, was reduced or mislocalized when MCT1 was inhibited. In isolated whole filters from rat kidneys, adding lactate quickly increased albumin leakage, and MCT1 blockade produced a similar rise, supporting the idea that disturbed lactate handling directly weakens the filter.
Implications for diabetes and kidney health
The authors propose that proper movement of lactate through MCT1 is essential for podocytes to run their preferred energy program, respond to insulin, and maintain a tight filtration barrier. When this lactate “doorway” is disturbed—by blocking MCT1 or by chronic high blood sugar that alters lactate balance—podocytes lose metabolic flexibility, burn less sugar, over-rely on mitochondria, and become structurally unstable and leaky. For people, this work suggests that subtle changes in how kidney cells handle lactate may contribute to insulin resistance and early kidney damage long before overall kidney function appears abnormal. Understanding and eventually targeting this lactate transport system could open new ways to protect the kidney filters in diabetes and other metabolic diseases.
Citation: Szrejder, M., Audzeyenka, I., Rachubik, P. et al. MCT1 as a critical regulator of insulin signaling, energy homeostasis and podocyte function. Sci Rep 16, 5906 (2026). https://doi.org/10.1038/s41598-026-37093-x
Keywords: kidney podocytes, lactate metabolism, insulin resistance, glomerular filtration barrier, MCT1 transporter