Clear Sky Science · en
Sustainable activated carbon from palm waste for aqueous nickel II adsorption
Turning Farm Waste into a Powerful Water Filter
Across the globe, communities struggle with rivers and wells polluted by industrial waste. One especially troubling contaminant is nickel, a metal that can harm lungs, kidneys, and even trigger cancer when it builds up in our bodies. This study explores an unexpectedly simple idea: using discarded palm fronds—agricultural waste that is often burned—to make an inexpensive filter material that can strip nickel from water with near‑perfect efficiency.
Why Nickel in Water Is a Hidden Threat
Nickel shows up in wastewater from electroplating, battery factories, metal finishing, and many other industries. Because it dissolves easily and moves freely through soil and groundwater, it can travel far from the original pollution source. Low doses are part of normal biology, but higher levels are linked to breathing problems, weakened immunity, kidney damage, allergic reactions, and an increased risk of lung cancer. Existing treatment technologies—such as chemical precipitation, membrane filtration, and ion exchange—can work well, but they tend to be expensive, energy‑intensive, and generate large volumes of sludge, which is a burden for smaller and less wealthy communities.
From Palm Fronds to High‑Performance Carbon
Egypt grows more date palms than any other country, and each tree produces over 25 kilograms of dry leaves and fronds every year. These leftovers are usually treated as trash. In this work, the researchers cut palm fronds into small pieces, washed and dried them, then soaked the material in phosphoric acid and heated it in a furnace. This process turns the plant matter into activated carbon—a very porous form of carbon with a huge internal surface area. Careful measurements showed that the resulting material, called PFTAC, has a mesoporous structure with slit‑like pores, a large internal surface area (over 350 square meters per gram), and many chemical groups on its surface that can grab metal ions from water.
How the New Filter Cleans Nickel from Water
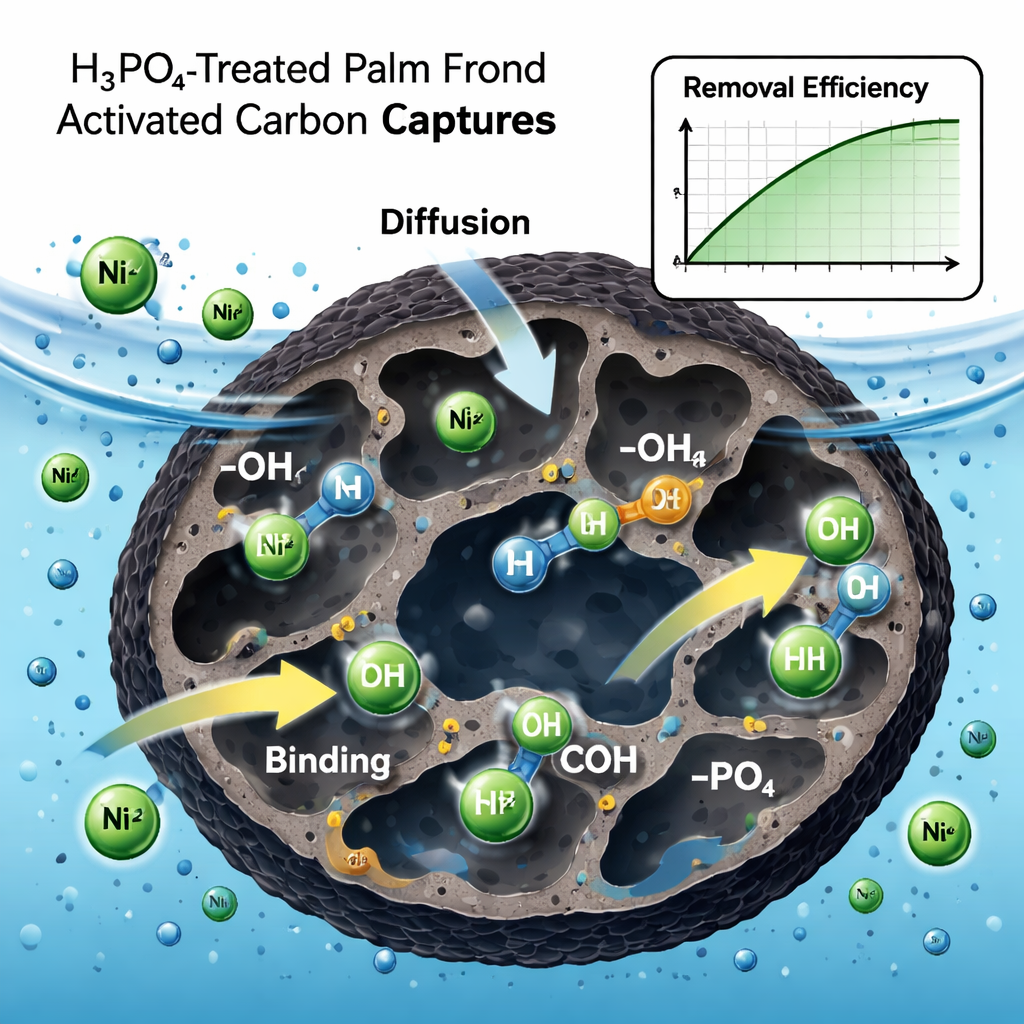
To test PFTAC, the team added it to water containing nickel and stirred the mixture under different conditions, changing the contact time, temperature, acidity (pH), and initial nickel concentration. Under optimized settings—moderate temperature, slightly acidic to neutral pH, and realistic nickel levels—the material removed up to 99.65 percent of dissolved nickel within 90 minutes. The data showed that nickel sticks mainly as a single, uniform layer on the carbon surface, consistent with a so‑called Langmuir isotherm. Kinetic analysis indicated that the process is controlled by chemical‑style interactions, rather than simple physical trapping alone. Nickel ions diffuse into the pores and bind to oxygen‑rich sites such as hydroxyl, carboxyl, and phosphate groups created during the phosphoric‑acid treatment.
Tuning Conditions for Maximum Cleanup
The researchers also studied how practical operating conditions affect performance. They found that very low pH (strongly acidic water) makes nickel and hydrogen ions compete for the same binding spots, reducing removal. As the pH rises toward about 3, nickel uptake improves, but at higher pH nickel starts to form solid hydroxide particles, changing how it behaves. Increasing the amount of palm‑based carbon gives more active sites and raises removal efficiency, while higher temperatures make it easier for nickel ions to move into the pores, confirming that the process absorbs heat and proceeds more readily when warm. Using a statistical approach called response surface methodology, the team mapped how time, temperature, and starting nickel level interact, and showed that their mathematical model can reliably predict removal efficiency across a wide range of conditions.
What This Means for Cleaner, Cheaper Water
For non‑specialists, the main takeaway is straightforward: a common agricultural waste, palm fronds, can be transformed into a low‑cost, reusable filter that strips nickel from water almost completely. Because it is made from locally available biomass, requires modest equipment, and can be regenerated and used again, this material could help factories and municipalities cut pollution without resorting to complex, high‑priced technologies. With further testing in continuous‑flow systems and real industrial effluents, palm‑waste activated carbon may offer a practical route to safer drinking water and cleaner rivers, especially in regions where both water and money are scarce.
Citation: Hammad, W.A., Abdel-latif, M.S., Hawash, S.A. et al. Sustainable activated carbon from palm waste for aqueous nickel II adsorption. Sci Rep 16, 6523 (2026). https://doi.org/10.1038/s41598-026-37088-8
Keywords: nickel removal, activated carbon, palm waste, heavy metals, wastewater treatment