Clear Sky Science · en
Hippocampo–supramammillary coupling across sleep and wake
Why this matters for everyday memory
Every night, while we sleep and even while we quietly rest, our brains replay experiences and reshape memories. This study zooms in on how a key memory center, the hippocampus, chats with deeper brain hubs that control arousal, movement, and mood. By watching these regions talk during different sleep and wake states in rats, the researchers uncover how the brain flexibly routes information—sometimes sending strong top-down signals from memory circuits, other times letting deeper regions briefly take the lead. Understanding this back-and-forth helps explain how sleep supports memory while keeping basic bodily functions stable.
Two-way talks between memory and arousal centers
The team recorded tiny electrical signals from the hippocampus and two connected regions, the supramammillary nucleus and the lateral septum, in freely moving rats. They followed the animals across quiet rest, deep non-rapid eye movement (non-REM) sleep, dream-rich rapid eye movement (REM) sleep, and active wakefulness. Using these recordings, they tracked brief bursts of synchronized hippocampal activity, called ripples, and related events in a nearby region called the dentate gyrus. At the same time, they measured how often neurons fired in the supramammillary nucleus, which influences arousal and sleep stages, and the lateral septum, which links memory circuits to motivation and movement systems.
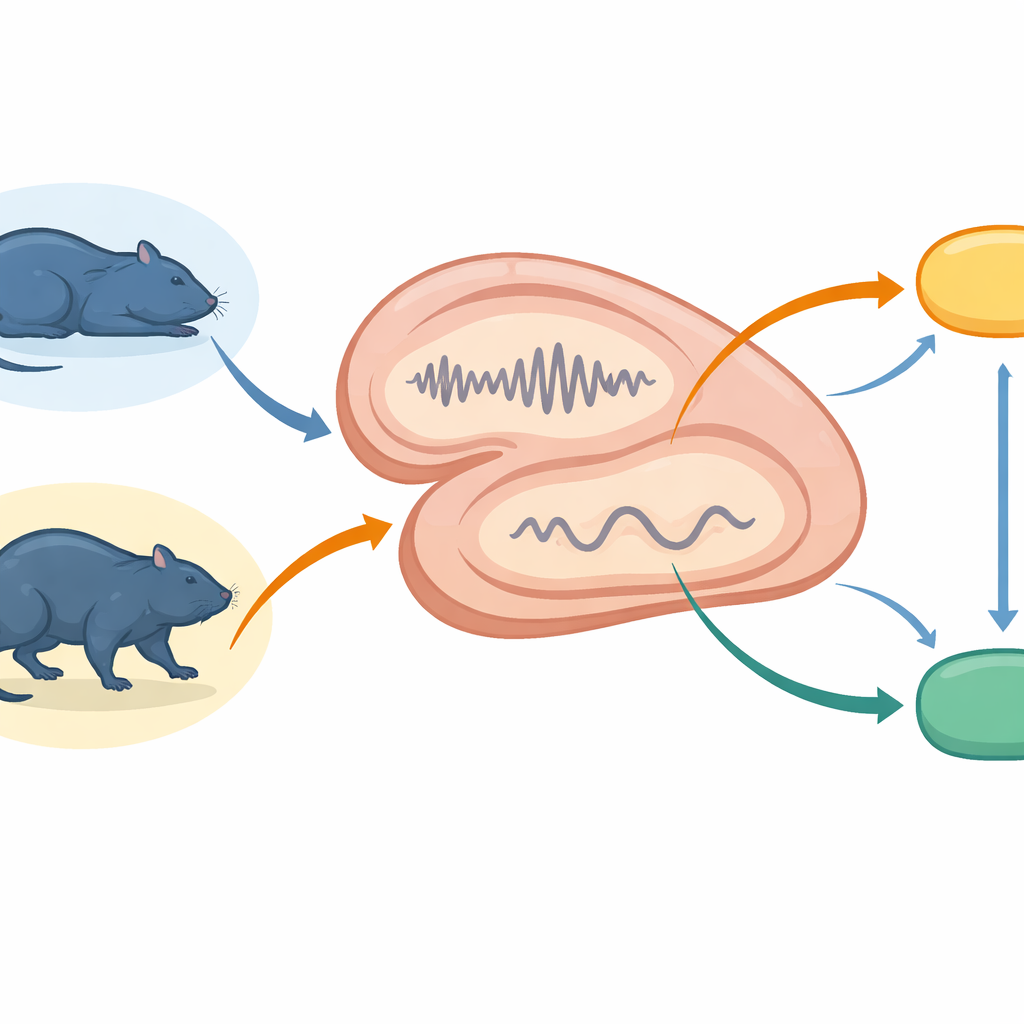
Top-down bursts during quiet moments
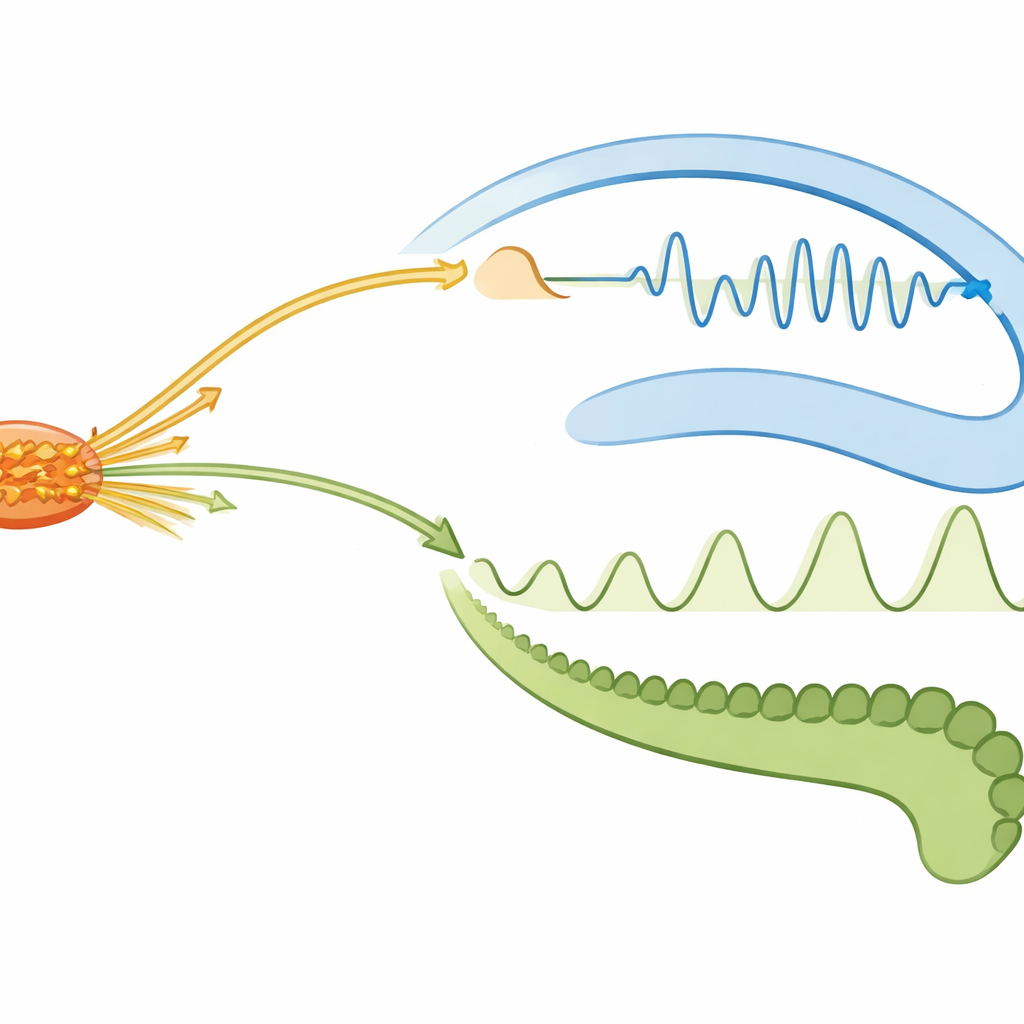
During quiet wakefulness and non-REM sleep—states thought to favor memory replay—the hippocampus frequently produced sharp, fast ripple events. These ripples triggered large firing surges locally within the hippocampus and smaller, but reliable, activation in the supramammillary nucleus and lateral septum. In other words, the memory system broadcast brief top-down bursts to deeper hubs, but with reduced strength compared with its internal activity. Dentate spikes, another kind of fast event in a nearby hippocampal subregion, caused weaker and more uniform responses that barely reached subcortical areas. This contrast suggests that ripples are the main vehicle for rapid, widespread coordination, whereas dentate spikes fine-tune local processing without strongly driving downstream centers.
Bottom-up nudges from deep brain bursts
The communication was not one-way. When the supramammillary nucleus entered short, high-activity epochs, it sent bottom-up signals back to the hippocampus. These had very different consequences depending on brain state. During non-REM sleep, such bursts briefly suppressed firing in a key hippocampal output area (CA1), while dentate gyrus neurons ramped up more slowly. In quiet wakefulness, the same supramammillary bursts produced a weaker dip in CA1 but a clearer net increase in overall hippocampal firing. These patterns indicate that deep brain bursts can either dampen or boost hippocampal output, tuning how strongly memory circuits influence the rest of the brain without completely turning them off.
Rhythmic timing during active and dreaming states
When the brain shifted into theta-rich states—active exploration during wake and vivid dreaming during REM sleep—the dialogue changed again. Theta waves are slower rhythms that help coordinate firing across regions. In active wakefulness, theta organized a repeating firing order: dentate gyrus neurons tended to fire first within each cycle, followed by lateral septum, and then CA1 near the trough of the wave, while the supramammillary nucleus showed little consistent timing. Supramammillary bursts in this state produced quick, brief inhibition in CA1 but left dentate gyrus activity largely intact, hinting at a targeted gating of certain hippocampal inputs. In REM sleep, the pattern flipped: supramammillary neurons now locked tightly to the theta rhythm, firing near the peak of each cycle, while hippocampal and septal cells showed weaker phase preferences. This shift suggests that during dreaming, deep arousal centers take on a more rhythmic, coordinating role.
What this means for sleep, memory, and brain balance
Taken together, the results reveal a flexible, state-dependent partnership between the hippocampus and its subcortical partners. During quiet states, sharp ripples let the hippocampus send powerful but carefully limited bursts outward, likely supporting memory replay without triggering large changes in arousal or behavior. During more active, theta-dominated states, brief supramammillary bursts can selectively curb or time hippocampal output, especially in CA1, while REM sleep allows these deep neurons to rhythmically synchronize with memory circuits. For a lay reader, the key idea is that memory regions and arousal centers are in constant two-way negotiation, adjusting who leads and who follows as we move from wakefulness to different stages of sleep, helping preserve memories while keeping the brain’s overall state stable.
Citation: Caneo, M., Espinosa, N., Lazcano, G. et al. Hippocampo–supramammillary coupling across sleep and wake. Sci Rep 16, 10465 (2026). https://doi.org/10.1038/s41598-026-37066-0
Keywords: hippocampus, sleep and memory, brain rhythms, supramammillary nucleus, neural communication