Clear Sky Science · en
Research on the molecular mechanism of celastrol targeting CTNNB1/STAT3 to inhibit uveal melanoma based on network pharmacology and multi-omics analysis
Ancient medicine meets eye cancer
Uveal melanoma is a rare but deadly cancer that arises inside the eye. Once it spreads, today’s treatments do little to change the outcome. This study explores whether celastrol—a molecule extracted from a traditional Chinese medicinal plant—could be repurposed to fight this cancer. By combining big-data biology, computer simulations and lab experiments, the researchers uncover how celastrol may shut down key molecular switches that help uveal melanoma grow and spread.
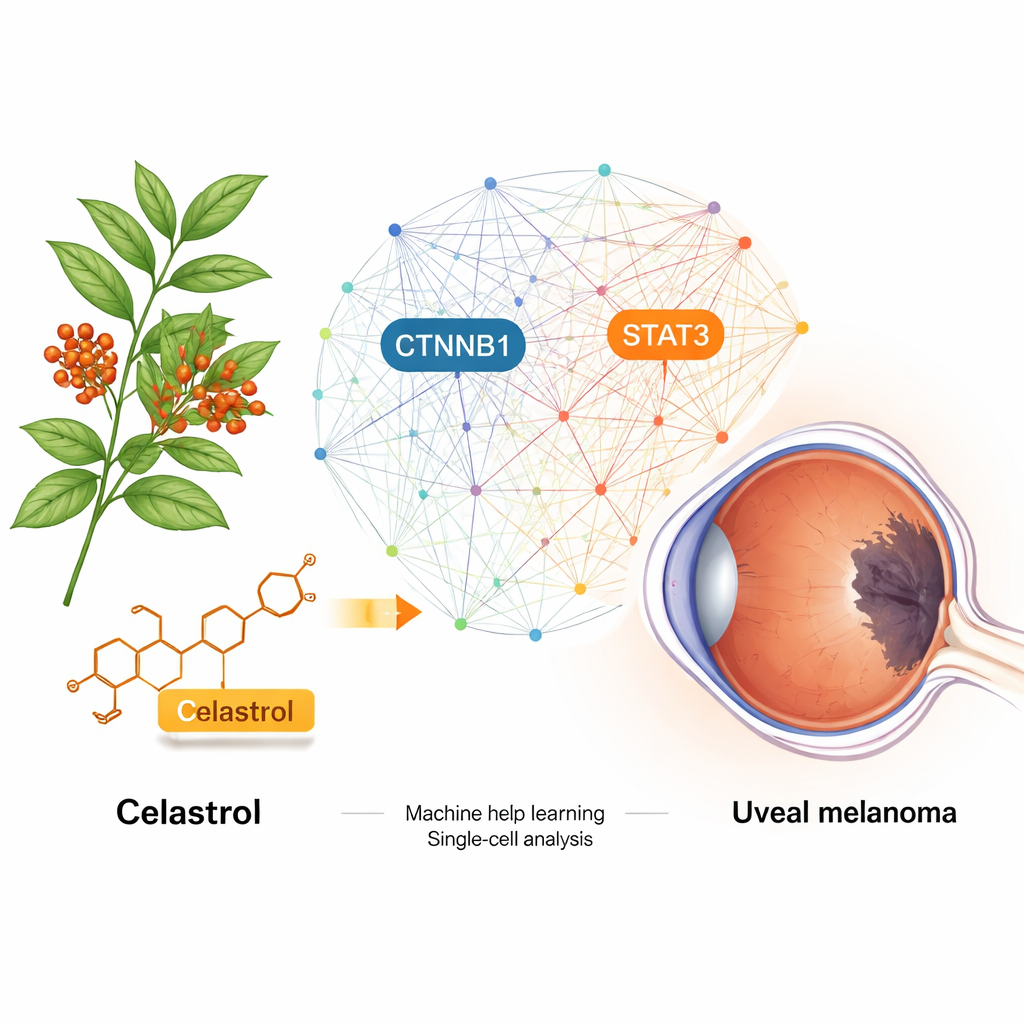
What makes this eye tumor so dangerous
Uveal melanoma is the most common primary eye cancer in adults, and about half of patients eventually develop metastases, usually in the liver. At that stage, survival is often measured in months. Standard treatments such as surgery, radiation and local therapies can control the main eye tumor, but drugs that circulate through the body have had limited success and can cause serious side effects. Scientists therefore are searching for medicines that can both curb tumor growth and be better tolerated—an area where natural products like celastrol are drawing intense interest.
A natural compound under the microscope
Celastrol comes from Tripterygium wilfordii, a plant long used in traditional Chinese medicine. Previous work showed that it can slow the growth of several cancers, but how it might act in uveal melanoma was unclear. The team began by mining numerous biomedical databases to predict which proteins in human cells celastrol might bind to, and which proteins are strongly linked to uveal melanoma. When they compared these lists, they found 46 overlapping candidates. Using computer tools that map how proteins interact with each other, they narrowed this down to a handful of “hub” molecules that sit at the control points of major growth and survival pathways in tumor cells.
Zeroing in on two master switches
To pinpoint the most critical players, the researchers combined patient tumor gene data from The Cancer Genome Atlas with three different machine-learning methods. All three approaches converged on two genes: CTNNB1, a core part of the Wnt signaling pathway that drives cell growth, and STAT3, a key regulator of inflammation, survival and immune evasion. Further analyses of bulk tumor samples and single-cell RNA sequencing showed that these genes are highly active in uveal melanoma cells, particularly in more aggressive cell subgroups, and are linked to changes in the surrounding immune cells. In short, CTNNB1 and STAT3 looked like master switches that help the cancer thrive while keeping the immune system at bay.
From computer models to living cells
The team then used molecular docking and long molecular dynamics simulations to test whether celastrol could physically latch onto CTNNB1 and STAT3. The virtual experiments suggested strong and stable binding, supported by multiple hydrogen bonds and tight packing around the drug. Next, they moved into the lab, treating human uveal melanoma cells and a related mouse melanoma cell line with celastrol. In both, celastrol sharply reduced cell survival and colony formation, slowed migration in wound-healing assays, and triggered programmed cell death. It also caused the cells to stall at specific points in the cell cycle, preventing them from dividing. When the researchers measured gene activity and protein levels, they found that celastrol markedly lowered CTNNB1 and STAT3, confirming that it hits its predicted targets inside living cells.
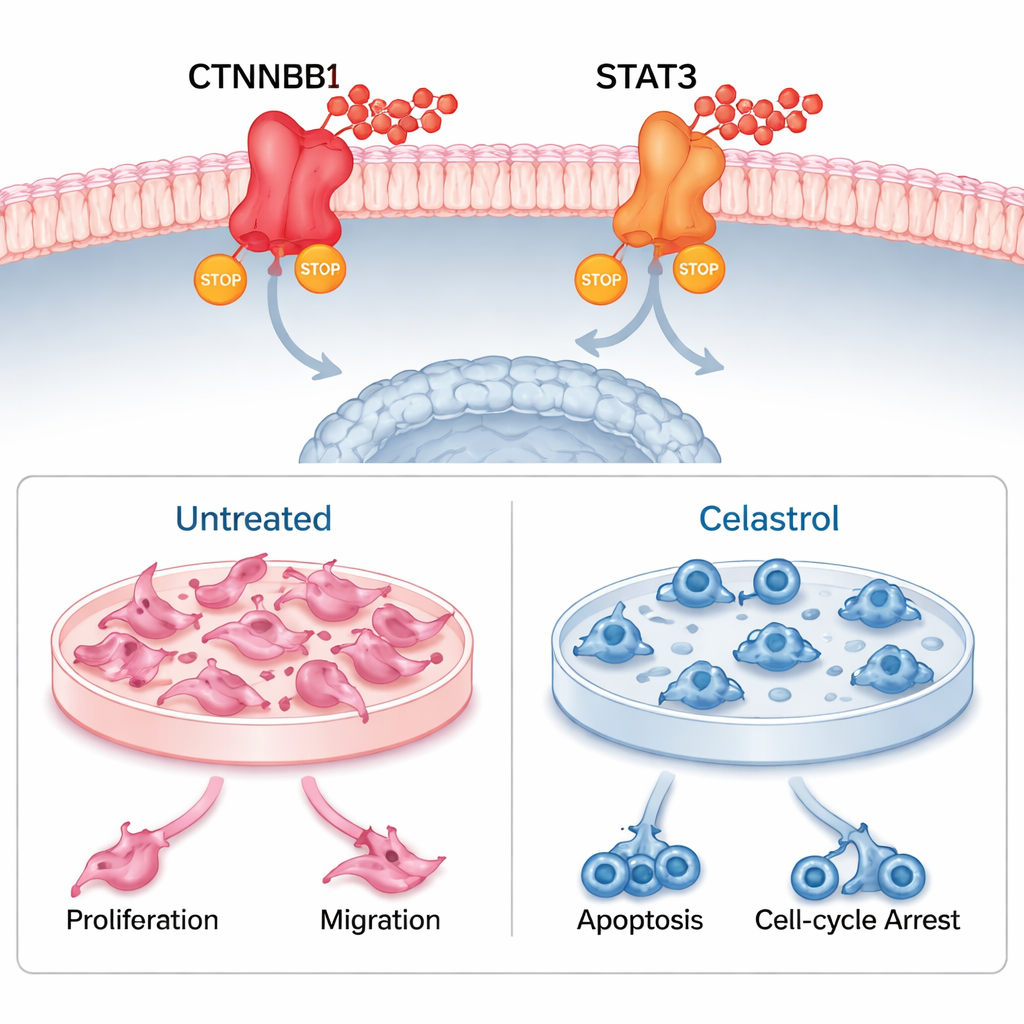
What this could mean for future treatments
Taken together, the results suggest that celastrol fights uveal melanoma by simultaneously switching off CTNNB1 and STAT3. This dual action not only slows tumor growth and spread, but may also reshape the tumor’s immune environment in ways that favor an anti-cancer response. While this work was done in cells and computer models—not yet in patients or animal models—it lays a strong foundation for further testing. For lay readers, the key message is that a compound rooted in traditional medicine has emerged as a scientifically plausible candidate for a new class of eye cancer treatments, one that targets both the tumor and the ecosystem of cells around it.
Citation: Li, Z., Xi, R., Han, X. et al. Research on the molecular mechanism of celastrol targeting CTNNB1/STAT3 to inhibit uveal melanoma based on network pharmacology and multi-omics analysis. Sci Rep 16, 6140 (2026). https://doi.org/10.1038/s41598-026-37061-5
Keywords: uveal melanoma, celastrol, eye cancer, cancer signaling, tumor microenvironment