Clear Sky Science · en
A unified multi modal transformer framework for breast cancer recurrence prediction and survival analysis
Why predicting cancer’s return matters
For many women, finishing breast cancer treatment brings relief mixed with a lingering question: will the disease come back, and if so, when and how severely? Today’s follow-up plans are often based on broad averages rather than the unique mix of factors that define each patient. This study introduces a new artificial intelligence system that aims to give doctors a clearer, more personalized view of both the risk that breast cancer will return and how long patients are likely to remain cancer‑free.
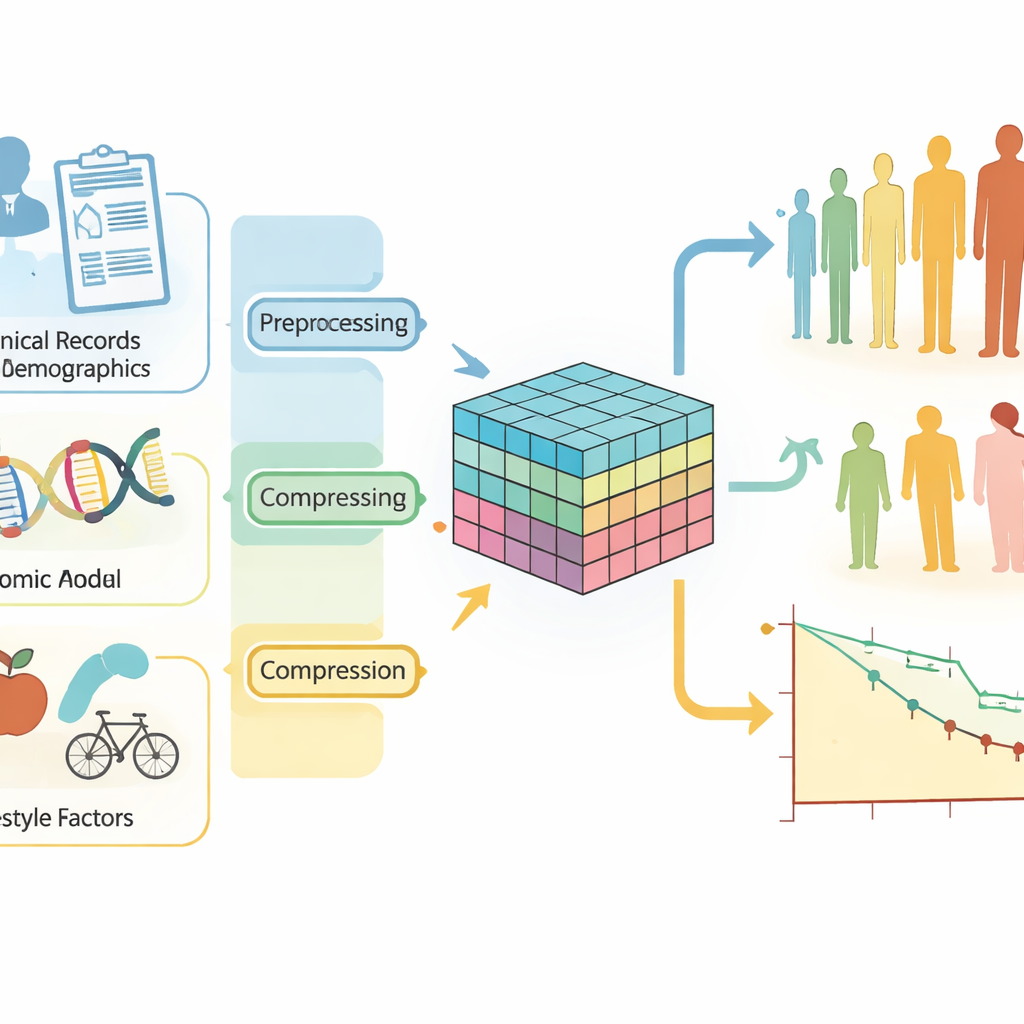
Bringing many kinds of patient data together
Breast cancer recurrence is not a single outcome. It can appear as a new tumor in the same breast, a spread to nearby lymph nodes, or distant metastases in organs such as the lungs or bones. Each pattern carries different implications for treatment and survival. At the same time, risk is shaped by many intertwined influences: tumor features, gene activity, age, menopausal status, body weight, smoking, and more. Traditional statistical tools struggle when faced with this mix of clinical, genetic, and lifestyle information. They typically assume simple, linear relationships and often rely on hand‑crafted risk scores that cannot capture the true complexity of modern cancer data.
A unified smart model instead of separate tools
The researchers designed a single deep learning framework that tackles two tasks at once: it predicts which of four recurrence types a patient is most likely to experience, and it estimates the timing of that event using survival analysis. Rather than building separate models for “will it come back?” and “when will it come back?”, the system learns both answers together. Under the hood, it uses a transformer architecture—the same family of models that power many cutting‑edge language tools—to discover subtle patterns and long‑range interactions in the data. This unified approach is meant to mirror how oncologists think, weighing many clues simultaneously instead of running isolated calculations.
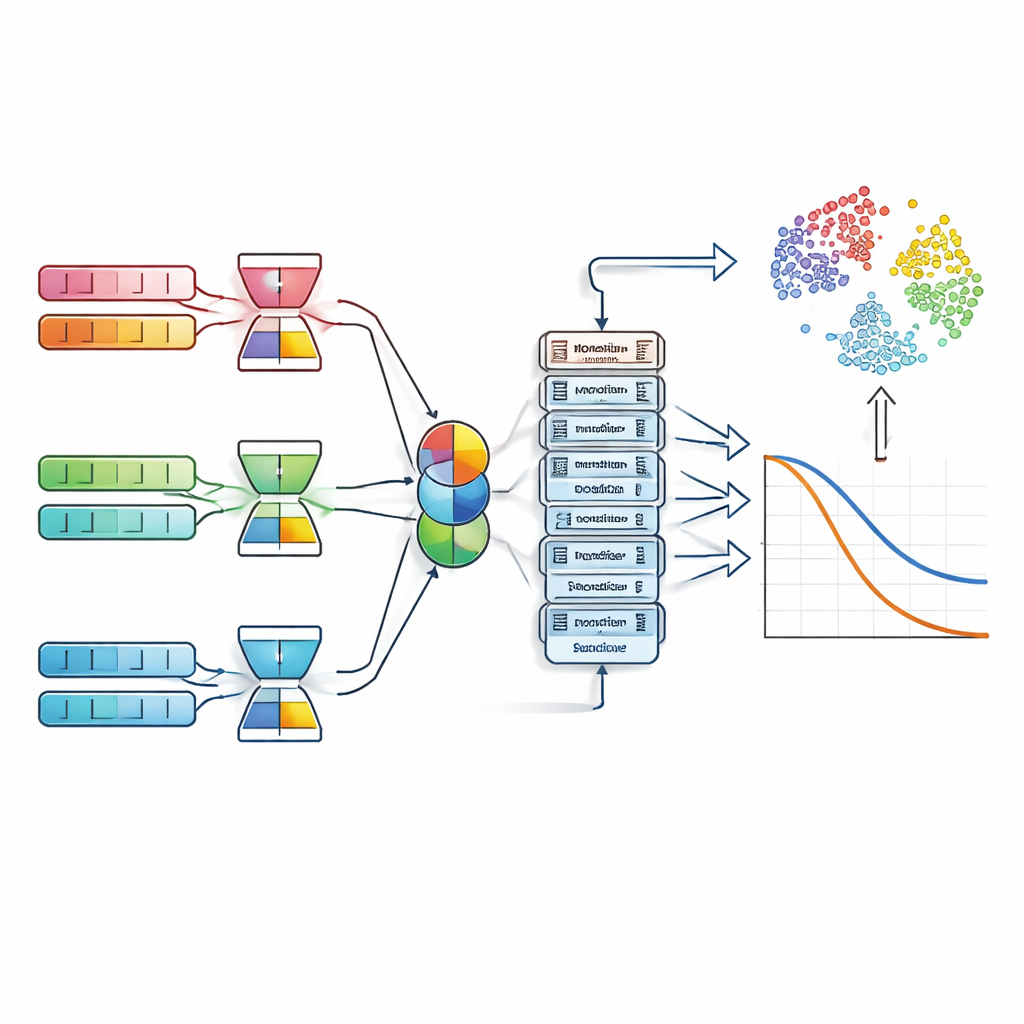
How the system reads patterns in health data
To feed the model, the team assembled a large multi‑center collection of breast cancer records from five well‑known sources. These include thousands of patients with detailed clinical measurements, gene expression profiles, demographic information, and lifestyle indicators. Because such data can be noisy and high‑dimensional—especially the tens of thousands of gene activity measurements—the system first passes each data type through a “denoising autoencoder.” This step compresses each modality into a cleaner, compact representation that keeps important biological signals while filtering out randomness.
Learning what matters most for each patient
After compression, the model does not simply glue all features together. Instead, it applies a modality‑attention mechanism that learns how much weight to give clinical, genetic, or lifestyle information for each individual. For some patients, tumor size and hormone receptor status may dominate; for others, a particular gene pattern or smoking history may be more telling. These weighted signals are fused into a single patient profile and processed by stacked transformer layers, which use self‑attention to model how different risk factors interact. From this shared representation, one branch predicts the type of recurrence, while another estimates a continuous risk score that can be translated into survival curves over five and ten years.
Performance, validation, and interpretability
In tests across the five datasets, the unified system consistently outperformed standard methods such as logistic regression, support vector machines, random forests, classical Cox survival models, and simpler neural networks. It achieved around 98–99% accuracy in classifying recurrence type and a high concordance index—an established measure of how well predicted survival order matches reality. Cross‑dataset experiments, where the model was trained on one cohort and tested on another, showed that it generalized better than competing approaches. To avoid becoming a mysterious “black box,” the authors also used explanation tools that highlight which features most strongly influenced each prediction. Tumor size, HER2 status, smoking, menopausal status, age at diagnosis, and BRCA1 mutations emerged as especially important, aligning well with current medical understanding.
What this means for patients and doctors
The study’s main message is that a single, carefully designed AI system can integrate many strands of information to give a richer, more reliable picture of breast cancer recurrence risk and survival. While it still needs prospective testing in real‑world clinics, the framework could one day help doctors tailor surveillance schedules, choose treatments, and counsel patients with greater confidence. For patients, this could translate into follow‑up plans that better match their true level of risk—reducing unnecessary anxiety and tests for some, while flagging others who might benefit from closer monitoring or more aggressive therapy.
Citation: Malik, S., Patro, S.G.K., Al-Nussairi, A.K.J. et al. A unified multi modal transformer framework for breast cancer recurrence prediction and survival analysis. Sci Rep 16, 8334 (2026). https://doi.org/10.1038/s41598-026-37046-4
Keywords: breast cancer recurrence, survival prediction, multimodal deep learning, transformer model, personalized oncology