Clear Sky Science · en
Potential biomarkers for early periodontal inflammation: investigating CD5+ B cells, salivary cytokines and oral microbiome
Why your gums matter to your whole body
Bleeding gums are easy to ignore, but the inflammation behind them can quietly erode the bone that holds teeth in place and is linked to heart disease, diabetes and other illnesses. This study asked whether early warning signs of such damage can be found in an everyday sample—saliva—together with certain immune cells in the blood and the mix of bacteria hiding under the gum line. Finding reliable early markers could help dentists flag high-risk patients long before irreversible tooth loss occurs.
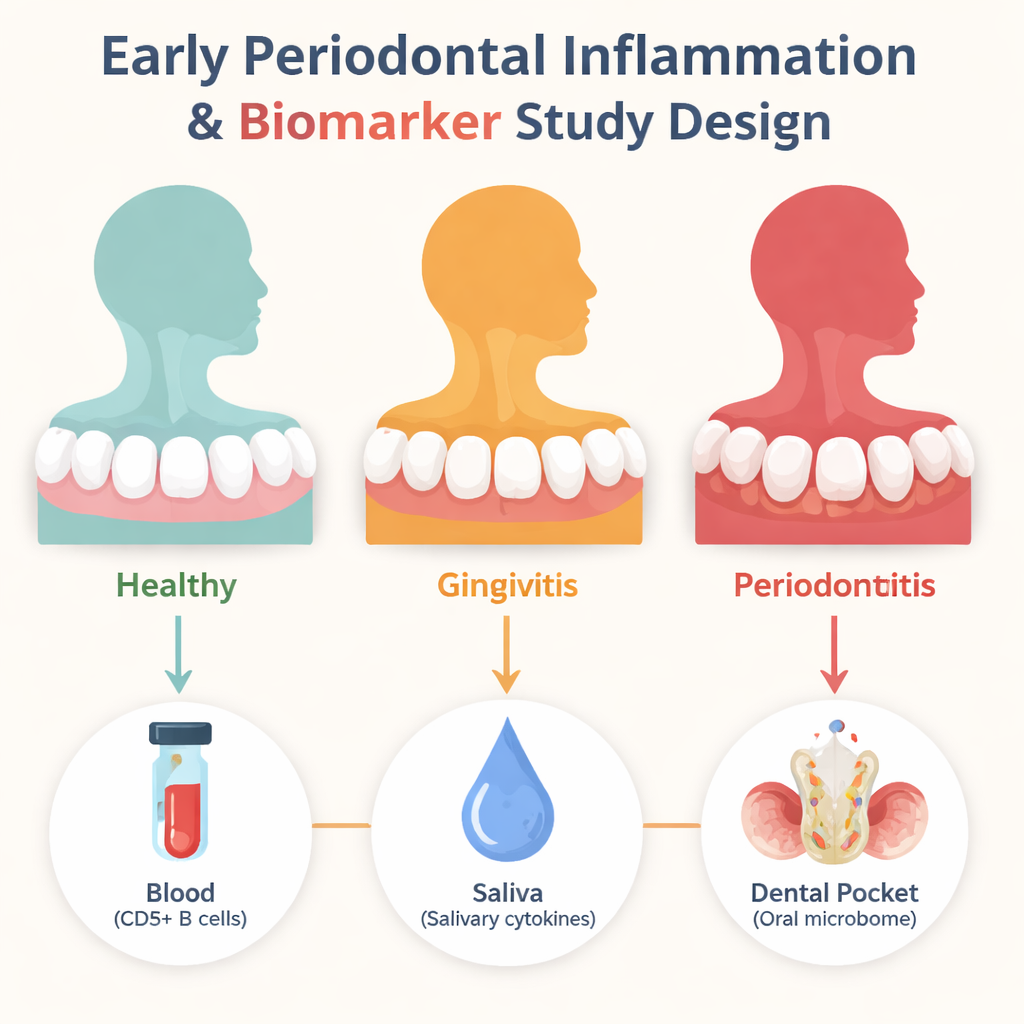
Looking for early warning lights
The researchers focused on three kinds of clues. First, they examined a special group of immune cells in the blood called CD5-positive B cells, which in severe gum disease and rheumatoid arthritis have been linked to bone breakdown and self-directed immune attacks. Second, they measured dozens of signaling proteins, or cytokines, in saliva that act as chemical alarms when tissues are irritated. Third, they sequenced bacterial DNA from fluid taken from gum pockets to see how the oral microbiome shifts as health gives way to gingivitis and then to moderate periodontitis. Sixty non-smoking adults were carefully grouped as healthy, gingivitis or moderate chronic periodontitis based on gum depth, bleeding and plaque scores.
Immune cells in blood stay quiet
Given earlier work in advanced disease, the team expected that people with periodontitis might already show raised levels of CD5-positive B cells in their bloodstream, marking a body-wide reaction to chronic gum infection. Surprisingly, total counts of these cells were essentially the same in all three groups, hovering around one-fifth of all B cells. Even when the scientists broke them down into developmental stages—immature, naive and several types of memory cells—no clear pattern reached statistical significance. Some memory cells, especially those linked to long-lasting responses, tended to be fewer in people with disease, hinting that they may be leaving the blood to accumulate in inflamed gum tissue, but larger studies will be needed to confirm this subtle trend.
Saliva and bacteria tell a stronger story
In contrast, saliva and the bacterial community beneath the gums clearly reflected disease stage. People with periodontitis had roughly double the median level of the chemokine IL-8 in saliva compared with healthy or gingivitis participants, and were much more likely to have detectable IL-17A as well as signs of IL-6 and IL-1β activity. These molecules help call in immune cells and drive bone-resorbing processes, so their rise suggests that inflammatory circuits are already active even in moderate disease. At the same time, DNA sequencing showed that healthy mouths were dominated by harmless, oxygen-loving bacteria such as Rothia and Streptococcus. With gingivitis and especially periodontitis, the balance shifted toward oxygen-shy, gram-negative species including Tannerella, Fusobacterium, Treponema and Fretibacterium, which are known or emerging culprits in gum breakdown.
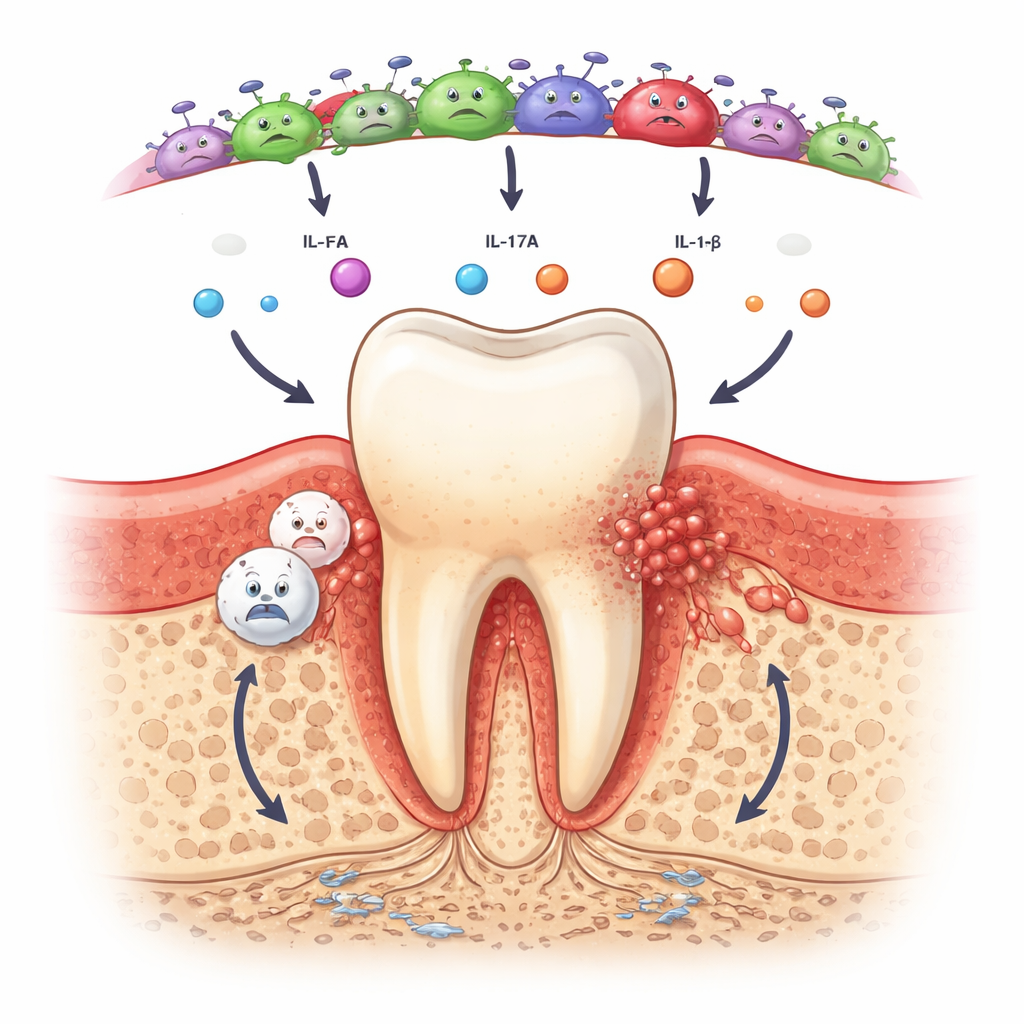
Early hints of a high-risk signature
By combining statistics and machine learning, the team identified bacterial groups that best separated healthy from diseased sites. Two strains of Tannerella forsythia and several Fretibacterium species emerged as particularly informative, while a Rothia species signaled healthier gums. An intriguing finding came from the gingivitis group: levels of IL-8 in saliva tracked closely with the abundance of a bacterial genus called Megasphaera. This link did not appear in healthy mouths or in full-blown periodontitis, suggesting that Megasphaera plus IL-8 might act as an early-stage indicator while damage is still limited and potentially reversible. However, the sample size was modest, and the authors stress that such patterns must be tested in larger, long-term studies before they can guide care.
What this means for patients
For people worried about their gums, the take-home message is that the body does send early biochemical signals of trouble, but these show up more in local saliva and gum bacteria than in circulating immune cells when disease is still moderate. Routine blood tests for CD5-positive B cells are unlikely to flag early periodontitis. Instead, the most promising approach may be a combined saliva-and-microbiome panel that tracks several cytokines—especially IL-8 and IL-17A—together with key bacterial species such as Tannerella, Fretibacterium and Megasphaera. If validated, such a simple mouth-based test could help dentists spot high-risk patients sooner, tailor cleanings and therapies, and perhaps prevent both tooth loss and some of the broader health problems tied to chronic gum inflammation.
Citation: Gottschalk, E.C., Chabanovska, O., Vasudevan, P. et al. Potential biomarkers for early periodontal inflammation: investigating CD5+ B cells, salivary cytokines and oral microbiome. Sci Rep 16, 7192 (2026). https://doi.org/10.1038/s41598-026-37044-6
Keywords: gum disease, oral microbiome, salivary biomarkers, periodontitis, inflammation