Clear Sky Science · en
Impact of neuroendocrine neoplasm-specific systemic treatments on expression and function of CXCR4 in neuroendocrine tumor cells
Why this matters for future cancer care
When certain rare cancers of the gut and pancreas become more aggressive, they often stop responding to standard targeted treatments. Doctors and researchers urgently need new ways to see and attack these tougher tumors. This study asks a practical question with big implications: do the chemotherapy and targeted drugs that patients already receive change a key tumor marker called CXCR4, which is being developed as both a diagnostic “beacon” and a treatment target?
A shifting target on aggressive tumor cells
Neuroendocrine neoplasms are rare cancers that arise from hormone-producing cells, most often in the intestine and pancreas. In their early, more orderly form, these tumors usually display a surface molecule called somatostatin receptor 2 that doctors can image and treat with specialized radioactive drugs. As the disease becomes more aggressive, these tumors tend to lose that marker, making them harder to detect and target. At the same time, many of them switch on another surface molecule, a receptor called CXCR4, which is linked to faster growth, spread to distant organs, and poorer survival. Because CXCR4 can be imaged with a PET tracer and potentially attacked with radioactive or antibody-based drugs, it has become a promising “backup” target once standard options fail.
Testing real-world drugs in tumor cell models
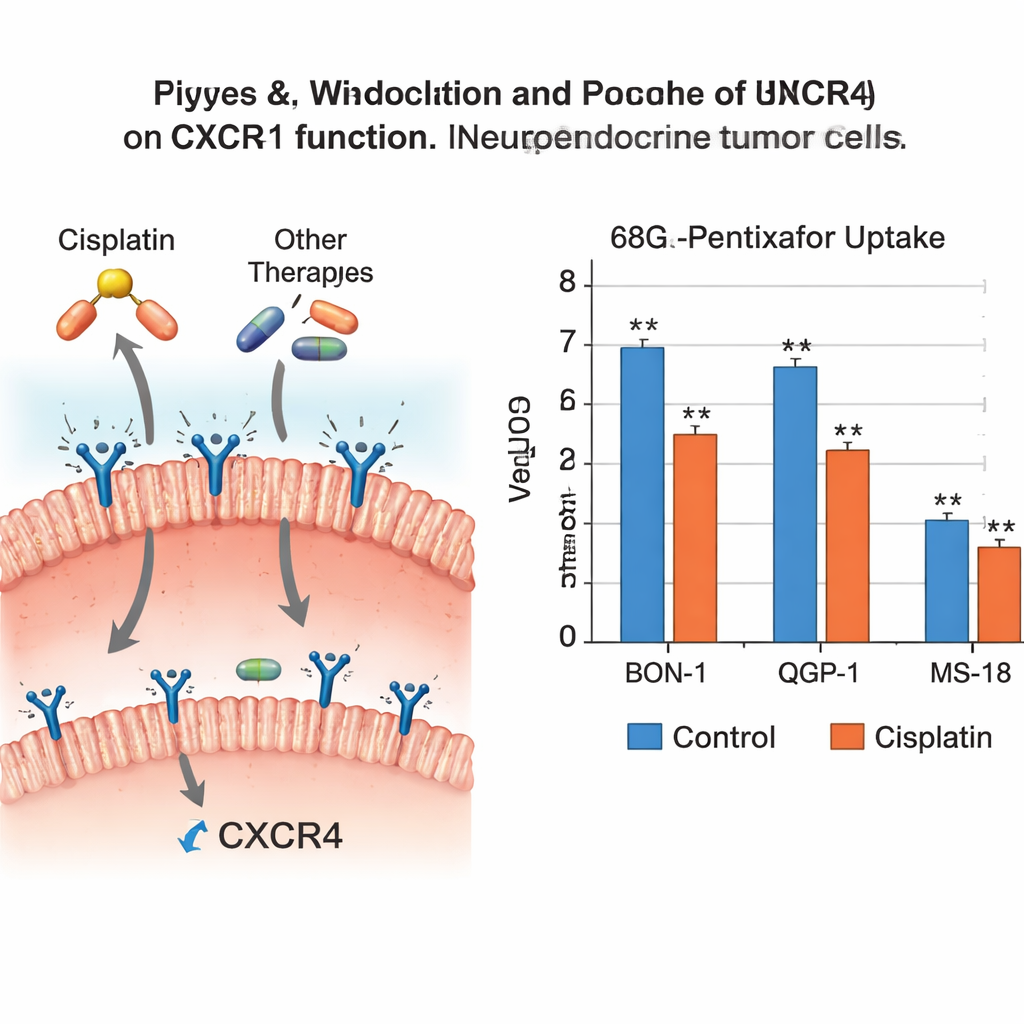
The researchers wanted to know how the drugs already used to treat high-grade neuroendocrine tumors affect CXCR4 itself. If these medicines accidentally lower CXCR4, they might weaken future CXCR4-based imaging or therapies—but they might also help tame the cancer’s aggressiveness. The team worked with three different human tumor cell lines representing aggressive forms of disease, including one hybrid line that closely mimics especially hard-to-treat tumors. They exposed these cells to six common agents: the chemotherapies cisplatin, etoposide, streptozotocin, 5‑fluorouracil, the oral drug temozolomide, and the targeted mTOR inhibitor everolimus. After treatment, they measured CXCR4 at the genetic and protein level and tested how well the cells could still take up a CXCR4-seeking PET tracer.
Some drugs turn down the CXCR4 signal
The results showed that not all cancer drugs are equal when it comes to this receptor. Cisplatin clearly reduced CXCR4 genetic activity in two of the three cell lines and significantly cut tracer uptake in two, meaning there were fewer functioning receptors for the tracer to bind. Temozolomide and everolimus also lowered CXCR4 genetic and protein levels across several cell types, although this did not always translate into a statistically clear drop in tracer uptake. In contrast, etoposide and streptozotocin had little effect on CXCR4, while 5‑fluorouracil produced modest changes that did not strongly alter tracer binding. Overall, the pattern suggested that cisplatin, temozolomide, and everolimus can all dampen CXCR4 in these aggressive tumor cells, while the other agents mainly leave it unchanged.
What this means for scans and treatment timing
These findings carry a double message for future patient care. On one hand, turning down CXCR4 may be part of how drugs like cisplatin, temozolomide, and everolimus slow tumor spread, because high CXCR4 is linked to invasiveness and metastasis. On the other hand, if a tumor’s CXCR4 is suppressed by ongoing treatment, CXCR4-based PET scans may underestimate how much disease is present, and CXCR4-targeted radioactive therapies may have fewer docking sites to hit. The study suggests that combining CXCR4-targeted treatments directly with these systemic drugs is unlikely to make those CXCR4-focused therapies work better, and might even blunt them.
A new piece of the puzzle for personalized therapy
For non-specialists, the key takeaway is that a single tumor marker can play two roles at once: it can be a warning sign of aggressive disease and, at the same time, a useful handle for imaging and targeted treatment. This research shows that some established cancer drugs can quietly adjust that marker up or down. In practice, this could influence when to schedule CXCR4-based scans and therapies—ideally when the receptor is most abundant—and may help explain why certain treatments are effective against fast-growing neuroendocrine tumors. While these results come from cell models and need confirmation in patients, they provide an important step toward smarter sequencing of therapies for people with these challenging cancers.
Citation: Däubler, C., Böttcher, C., Landwehr, LS. et al. Impact of neuroendocrine neoplasm-specific systemic treatments on expression and function of CXCR4 in neuroendocrine tumor cells. Sci Rep 16, 4339 (2026). https://doi.org/10.1038/s41598-026-37026-8
Keywords: neuroendocrine tumors, CXCR4, cisplatin, everolimus, targeted imaging