Clear Sky Science · en
An engineered M13 phage–rGO electrochemical biosensor for rapid detection of viral protein in complex matrices
Why fast virus tests still matter
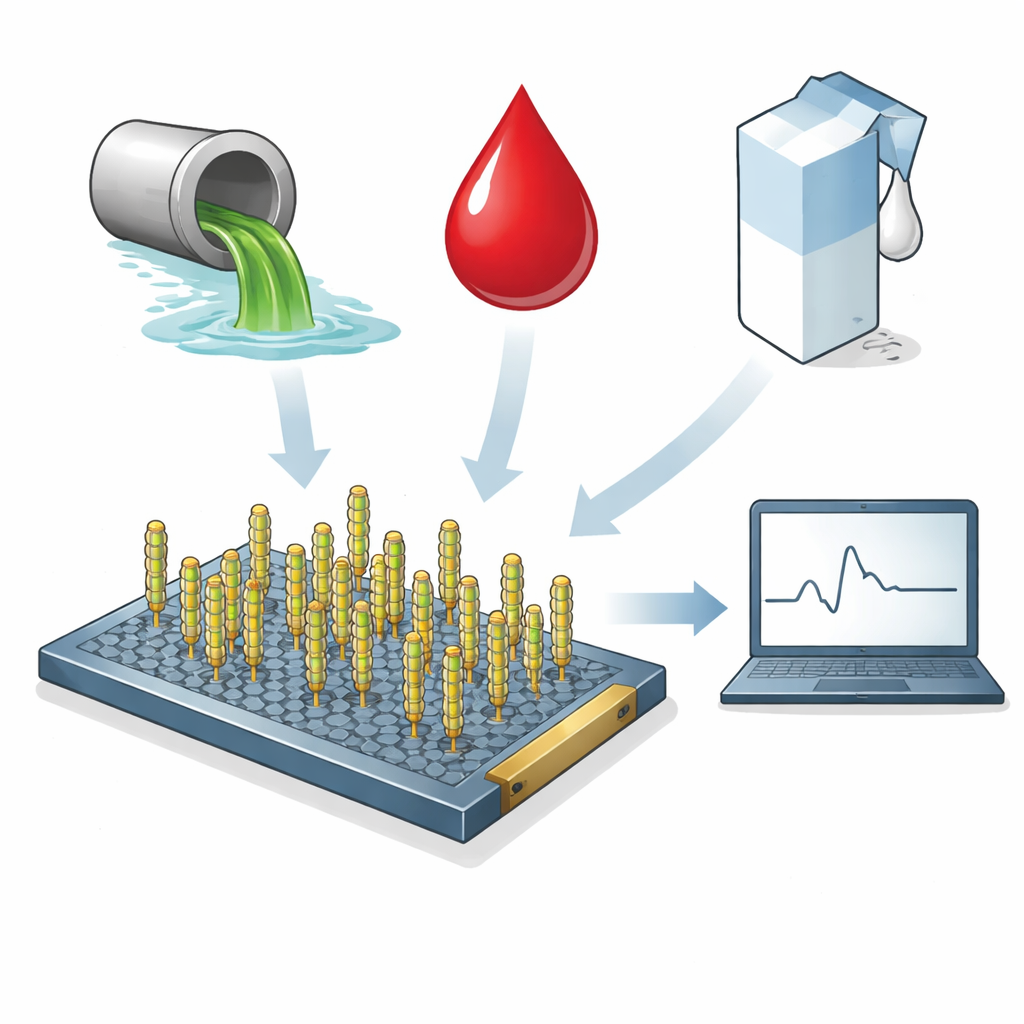
The COVID-19 pandemic highlighted how crucial it is to spot viruses quickly, not only in patients but also in places like wastewater and food-processing lines. Today’s lab tests are powerful but can be slow, expensive, and dependent on fragile biological ingredients that are hard to ship and store. This study presents a new kind of tiny electronic sensor that uses engineered viruses and a sheet of advanced carbon material to detect a key protein from SARS-CoV-2 in under a second, even in messy real-world samples such as blood serum, milk, and wastewater.
Turning a harmless virus into a smart detector
At the heart of the device is M13, a harmless virus that normally infects bacteria. Its long, rod-like body is covered with many identical coat proteins that scientists can reprogram genetically. The team inserted a short custom-made peptide onto one of these proteins so that the M13 particles would recognize and latch onto the S1 fragment of the coronavirus spike protein. A second version of the virus, carrying a scrambled peptide, served as a control to show that the response comes from true recognition rather than random sticking.
Building on a sheet of atom-thin carbon
To turn this programmable virus into a sensor, the researchers attached it to a flat film of reduced graphene oxide, a highly conductive form of carbon made by chemically processing graphite. After spreading graphene oxide on small glass chips and heating it to convert it into reduced graphene oxide, they added a linker molecule that sticks to the carbon surface and also binds to amine groups on the virus. This created a dense layer of M13 particles anchored to the conductive sheet. Electron microscopy and atomic force microscopy confirmed that each fabrication step changed the surface in the expected way, and electrical measurements showed that adding the linker and then the virus steadily increased the resistance, a sign that the surface was being successfully coated.
Reading virus binding as an electrical pulse
Unlike many biosensors that need added chemicals or moving parts, this platform works as a simple resistor under a tiny constant voltage. When the S1 protein lands on the virus-covered surface and binds to the displayed peptide, it slightly alters the way charge moves through the graphene layer. This shows up as a brief spike in electrical current that appears roughly 300 milliseconds after a drop of sample is placed on the chip, then fades as the system settles. By tuning the applied voltage, the team found a sweet spot at about minus 0.8 millivolts, where the signal from true S1 binding was strong while background noise and responses to unrelated proteins, such as bovine serum albumin, stayed low.
Working in messy real-world samples
The researchers then challenged the sensor with the kinds of complex mixtures that often defeat delicate lab reagents. They tested the device in buffer, municipal wastewater, fetal bovine serum (a stand-in for blood), and pasteurized milk, with and without added S1 protein. Using a statistically defined cutoff to mark a positive result, the sensor detected extremely low protein levels in simple buffer—down to about 10⁻⁴ picograms per milliliter—comparable to or better than many antibody-based systems. In wastewater, the device reliably flagged higher S1 levels, while in serum and milk it consistently detected lower concentrations as well, all within a fraction of a second. Importantly, a control sensor built with the scrambled virus showed little response to S1, confirming that the signal depends on the engineered binding sequence. A parallel sensor using a conventional antibody on the same graphene platform performed similarly, suggesting that the virus-based system can match antibody sensitivity while potentially being cheaper and easier to produce.

What this could mean for everyday testing
Antibodies, the workhorses of many diagnostics, are costly to manufacture, sensitive to heat, and typically require cold storage from factory to clinic. In contrast, M13 viruses can be grown in bacteria like a simple culture, tolerate harsher conditions, and can be reprogrammed by rewriting their genetic code. By marrying this robustness and flexibility with a fast, low-power electronic readout on graphene, the study outlines a path toward portable, low-cost devices that could be adapted to detect many different disease markers or contaminants simply by changing the displayed peptide. The work is still at a proof-of-concept stage and has not yet been tested in human clinical samples, but it points toward a future where handheld sensors could screen for viral proteins and other biomarkers in clinics, sewers, or even food products within seconds, without the logistical burden of traditional antibody-based tests.
Citation: Alshehhi, H.Y., Tizani, L., Palanisamy, S. et al. An engineered M13 phage–rGO electrochemical biosensor for rapid detection of viral protein in complex matrices. Sci Rep 16, 9279 (2026). https://doi.org/10.1038/s41598-026-37008-w
Keywords: biosensor, graphene, bacteriophage, SARS-CoV-2, electrochemical detection