Clear Sky Science · en
Comparative study of palmitoleic acid, sea buckthorn oil, and lovastatin in hepatocellular steatosis model
Why a fatty acid from berries and milk matters
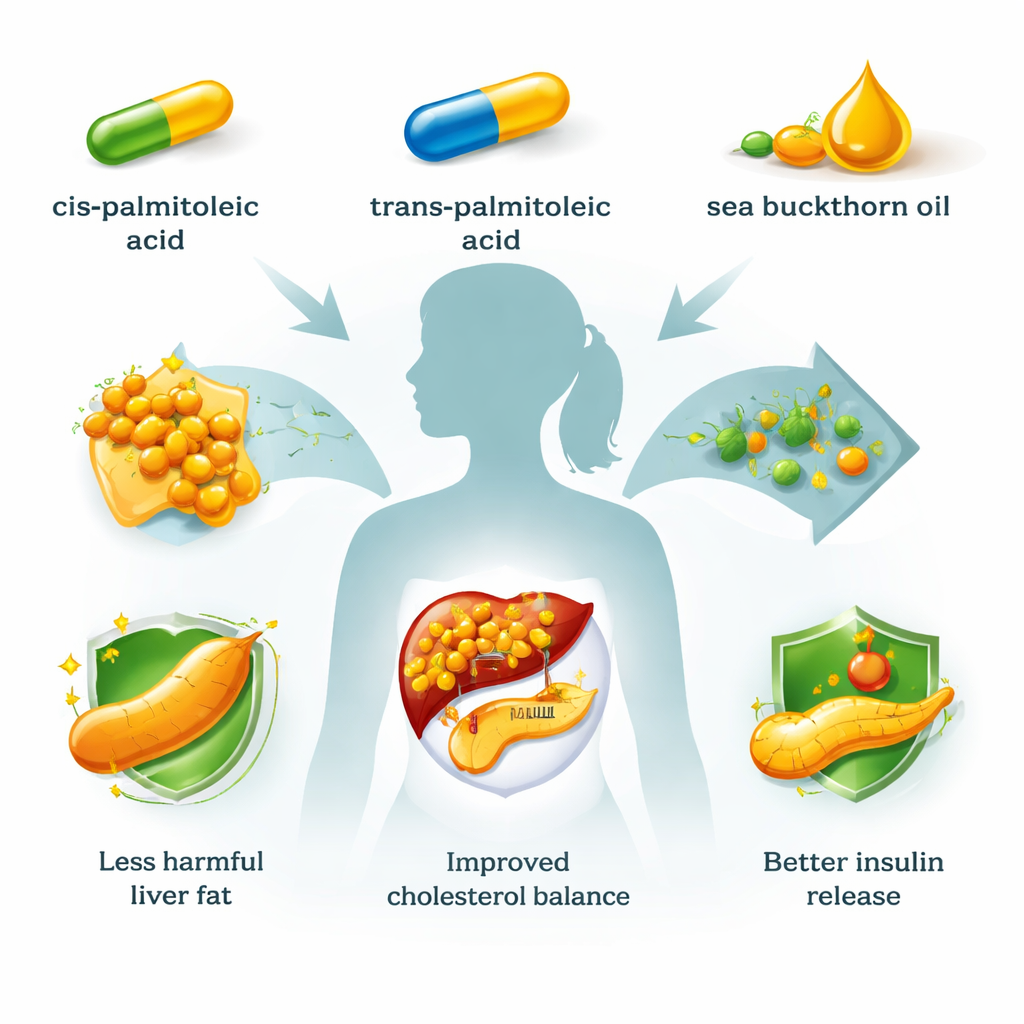
Fatty liver disease and type 2 diabetes are rising worldwide, and many people take statin drugs to control cholesterol at the same time. This study asks a timely question: could certain natural fats from foods—especially a fatty acid called palmitoleic acid and the bright orange oil of sea buckthorn berries—help tame liver fat and cholesterol while supporting, rather than harming, the body’s ability to release insulin? The researchers compared these natural substances head-to-head with the cholesterol drug lovastatin in human liver and pancreatic cell models.
Setting up a fatty liver in a dish
To mimic fatty liver disease in the lab, the team used HepG2 cells, a widely used human liver cell line. They overloaded these cells with a mix of two common dietary fats, palmitic acid and oleic acid, which drove the buildup of fat droplets inside the cells—similar to what happens in metabolic dysfunction-associated steatotic liver disease (MASLD). Once this “mini fatty liver” was established, they treated the cells with cis-palmitoleic acid, trans-palmitoleic acid, crude sea buckthorn oil, a digested form of that oil (to imitate what happens in the gut), or lovastatin. Across a broad range of doses, none of the palmitoleic acid forms or sea buckthorn preparations damaged cell viability over 24 hours, even in already fat-laden cells, suggesting that these interventions are not directly toxic to liver cells under the tested conditions.
Fat and cholesterol: a mixed but promising picture
When the researchers looked at fat storage, all the tested treatments generally increased triacylglycerol (TAG) content in normal liver cells, reflecting extra fat being drawn into storage. In steatotic, or already fatty, cells this effect was blunted, likely because the cells were near their storage capacity. Here an important nuance emerged: while most treatments still pushed TAG levels upward, trans-palmitoleic acid showed a modest fat-lowering tendency at some doses and did not significantly boost TAG at the highest dose—hinting at a potentially favorable profile for liver fat handling. Cholesterol told a different and clinically important story. In healthy cells, cholesterol levels barely changed. But in fatty liver cells, low and moderate doses of trans-palmitoleic acid, sea buckthorn oil, and especially its digested form clearly reduced cholesterol, in some cases approaching the cholesterol-lowering effect of lovastatin. The team also measured expression of HMG-CoA reductase, the key enzyme that statins block. Most palmitoleic and oil treatments dampened this enzyme’s gene expression in at least some settings, while computer docking simulations suggested that both cis- and trans-palmitoleic acid can sit inside the enzyme’s catalytic pocket, much like the natural substrate, potentially slowing cholesterol production directly.
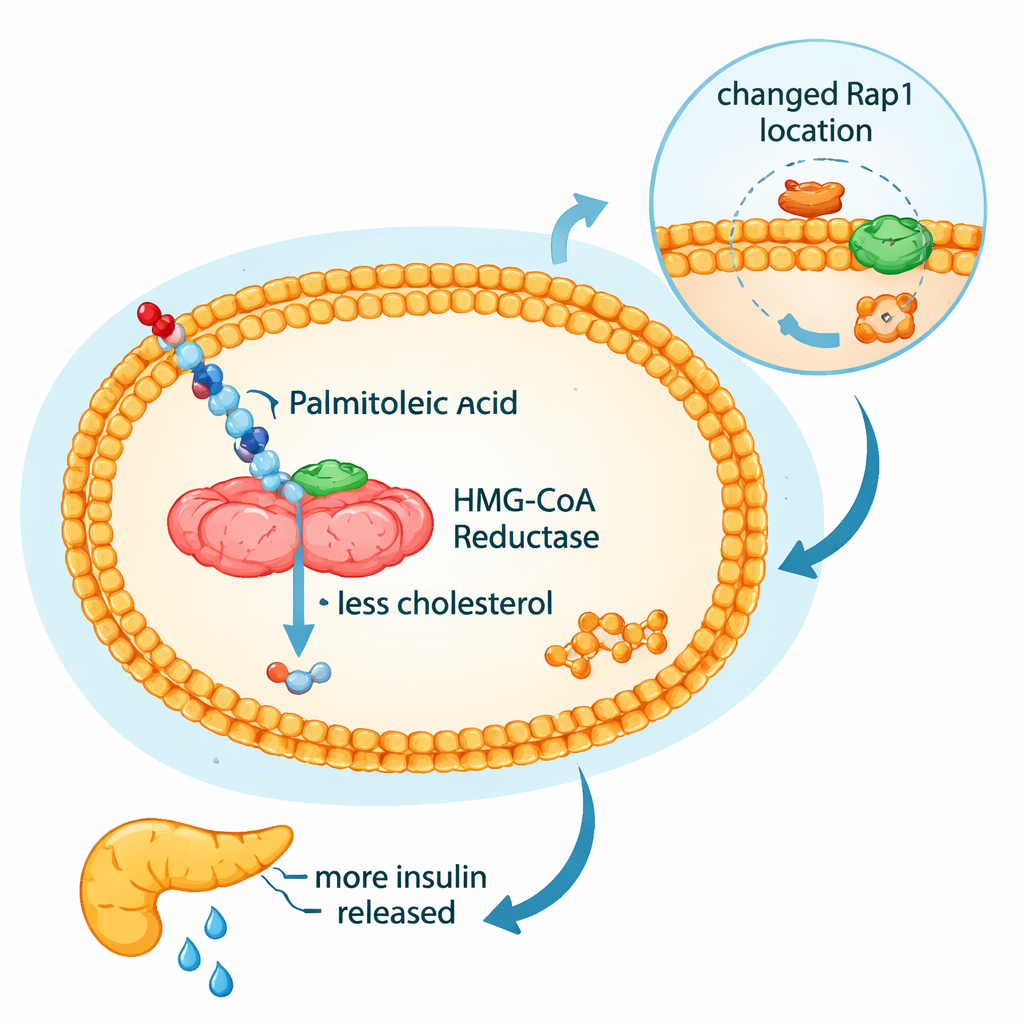
A closer look at a tiny switch inside liver cells
Cholesterol production feeds not only into blood lipids but also into small lipid attachments that anchor signaling proteins to cell membranes. One such protein, Rap1a, helps regulate glucose production in the liver. Statins, by strongly blocking the cholesterol pathway, can reduce these lipid attachments, dislodging Rap1a from membranes and contributing to higher blood sugar in some patients. In this study, lovastatin behaved as expected: it shifted Rap1a away from the membrane into the cell interior, especially in fatty liver cells. The palmitoleic acids and sea buckthorn oil showed a milder, condition-dependent version of this effect. Under steatotic conditions, both cis- and trans-palmitoleic acid, and to a lesser degree sea buckthorn oil, increased the pool of Rap1a in the cytosol and reduced the membrane-bound form. This suggests that these natural fats can partially mimic some “statin-like” actions on this pathway, but without the same intensity.
Insulin release: a key difference from statins
Because changes in Rap1a and cholesterol metabolism can affect how the pancreas secretes insulin, the researchers also studied MIN6 pancreatic beta cells. Under high glucose, these cells normally ramp up insulin release, mirroring the body’s response to a meal. Both cis- and trans-palmitoleic acid amplified this glucose-stimulated insulin secretion, with trans-palmitoleic acid giving a slightly stronger boost. Strikingly, the digested sea buckthorn oil—rich in free fatty acids such as palmitoleic acid—strongly enhanced insulin release, whereas the undigested oil did not, underscoring the importance of how these fats are processed during digestion. In sharp contrast, lovastatin dampened insulin secretion at high glucose, reinforcing clinical concerns that strong statin therapy can worsen blood sugar control.
What this could mean for everyday health
For non-specialists, the take-home message is that not all fats behave alike, and some may offer a gentler way to support metabolic health. In human cell models of fatty liver and pancreatic function, palmitoleic acid—especially its trans form from dairy fat—and sea buckthorn oil reduced cholesterol in stressed liver cells and boosted insulin release, all without the clear harm to blood sugar seen with a classic statin drug. These findings do not mean people should replace prescribed statins with supplements, nor do they prove benefit in real patients. But they suggest that targeted use of palmitoleic acid or carefully prepared sea buckthorn oil could one day complement existing therapies for fatty liver disease and metabolic disorders, potentially offering cholesterol control and better insulin support in the same package.
Citation: Szustak, M., Pichlak, M., Korkus, E. et al. Comparative study of palmitoleic acid, sea buckthorn oil, and lovastatin in hepatocellular steatosis model. Sci Rep 16, 6135 (2026). https://doi.org/10.1038/s41598-026-37006-y
Keywords: fatty liver disease, palmitoleic acid, sea buckthorn oil, cholesterol metabolism, insulin secretion