Clear Sky Science · en
Label-free fluorescence lifetime imaging can distinguish cancer from healthy tissue in spontaneously occurring canine oral tumors
Seeing Cancer in a New Light
Oral cancers in both people and pets are notoriously hard to remove completely. Surgeons must cut wide “safety” margins around a tumor to avoid leaving behind hidden cancer cells, but in the mouth that can mean losing vital bone, teeth, and soft tissue that affect eating and speaking. This study explores a light-based imaging method used during surgery in pet dogs that could help surgeons distinguish cancer from healthy tissue in real time—potentially sparing more normal tissue while still keeping the cancer at bay.
Why Edges of Tumors Are So Hard to Find
In head and neck cancer surgery today, doctors often rely on quick frozen-tissue analysis to judge whether the edges of a removed tumor are truly cancer-free. That process is slow, only samples small areas, and can miss disease. Similar challenges plague treatment of dogs with oral tumors, where local recurrence remains common. Because there is no dependable way to “see” microscopic cancer during an operation, surgeons typically remove generous margins, which can impair chewing, swallowing, and appearance. The authors set out to test a non-invasive imaging approach that might draw a clearer line between cancer and normal tissue directly in the operating room.
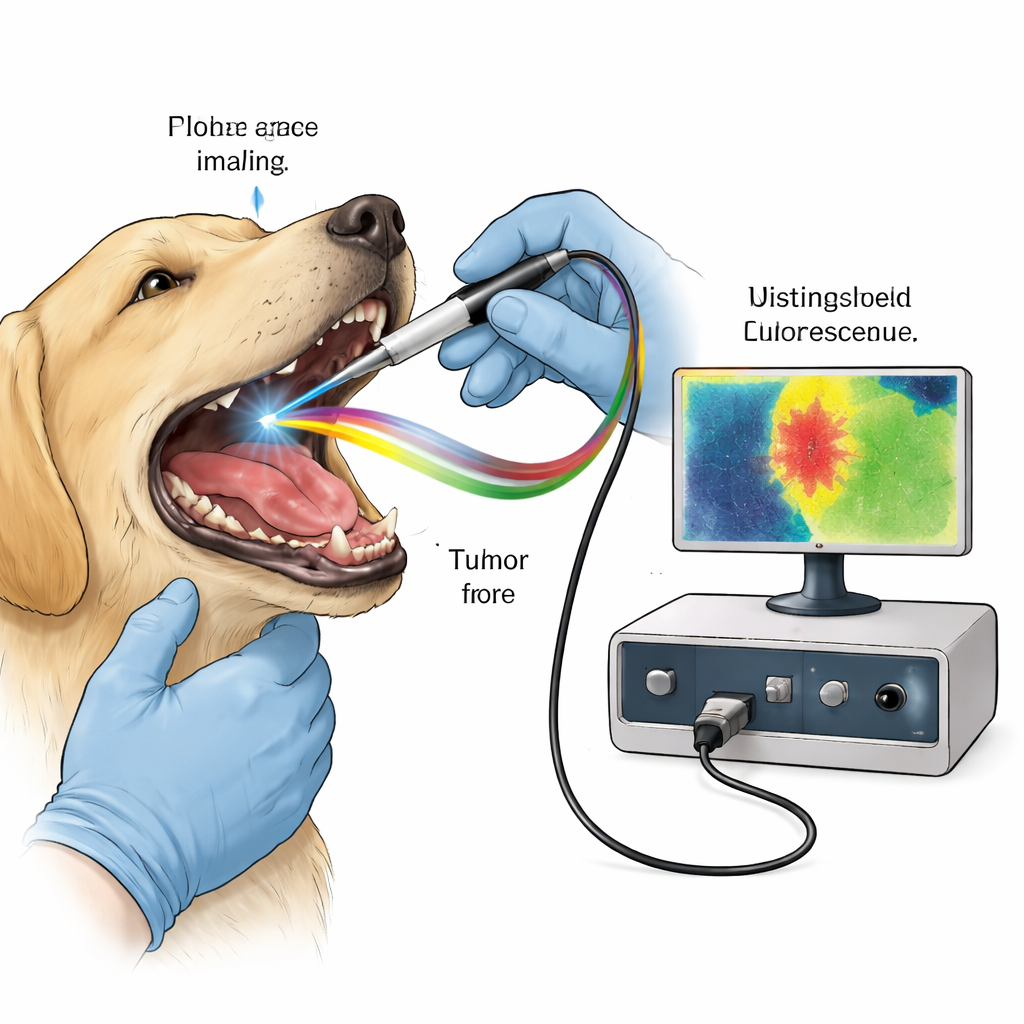
Using Natural Glow as a Surgical Guide
Many molecules in our tissues naturally emit faint fluorescent light after being excited by a brief laser pulse. Collagen in connective tissue, and molecules linked to cell metabolism such as NADH and FAD, each have characteristic colors and timing as they glow and fade. Cancer disrupts tissue structure and energy use, subtly changing this fluorescent fingerprint. Fluorescence lifetime imaging (FLIm) does not just measure how bright tissue glows; it measures how long that glow lasts in billionths of a second, a property that is less affected by viewing conditions. The team used a custom handheld probe that shines rapid ultraviolet pulses into the tumor region of anesthetized dogs and records the returning fluorescence in three spectral channels tuned to collagen, metabolic molecules, and a red-emitting compound called protoporphyrin IX (PpIX).
Adding a Cancer-Seeking Dye: Help or Hype?
PpIX can be made to accumulate in many tumors by giving patients a precursor drug, 5-aminolevulinic acid (5-ALA), by mouth. Under blue light, areas rich in PpIX often glow bright pink and are used by surgeons to spot cancer. The researchers first confirmed in canine oral cancer cell lines that 5-ALA led to strong PpIX fluorescence and showed that genes involved in producing and importing 5-ALA were more active in cancer cells. They then enrolled 15 pet dogs with naturally occurring oral tumors and gave them 5-ALA a few hours before surgery. In the operating room, most tumors visibly fluoresced, but some non-cancerous inflammatory or viral lesions also lit up, hinting that visual color alone might not reliably mark tumor borders.
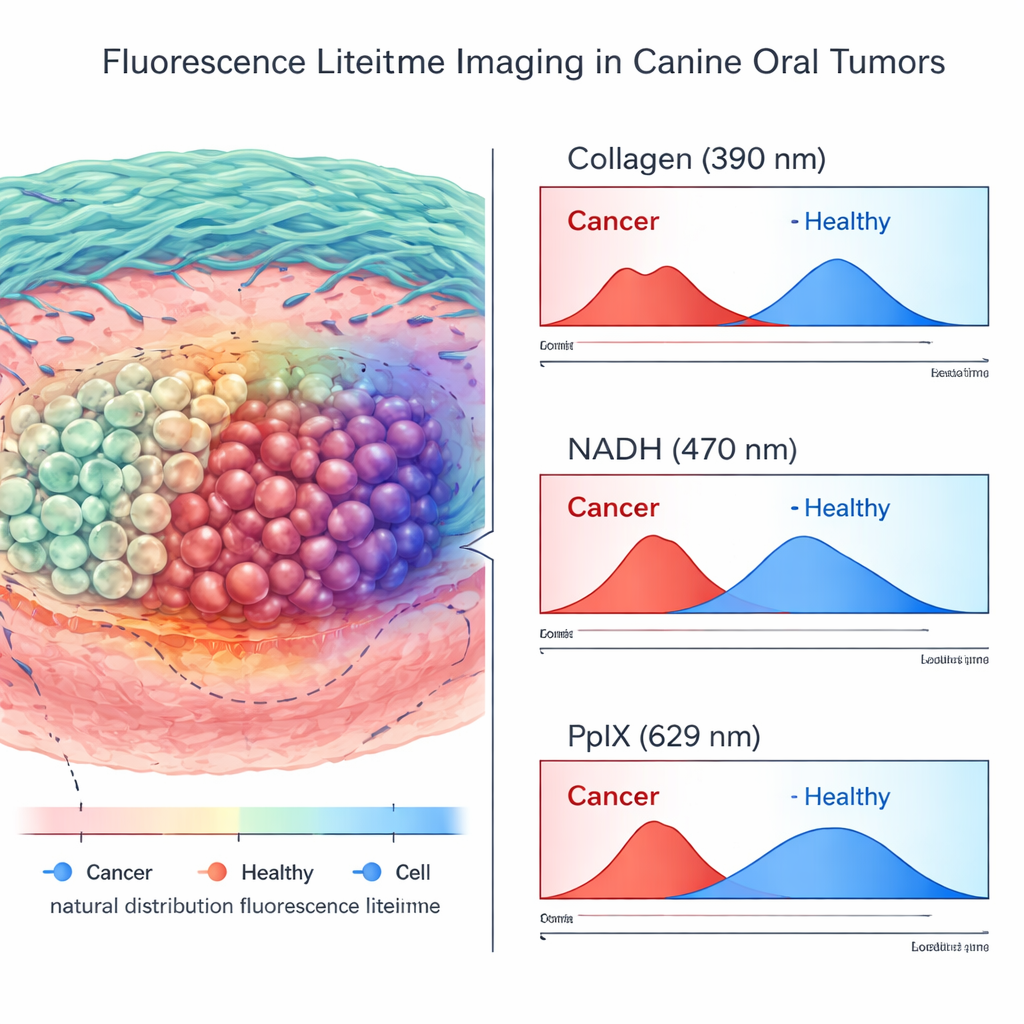
What the Lifetime Images Revealed
Across more than 200,000 high-quality measurement points from tumors and nearby normal tissues, the team compared FLIm signals with detailed pathology maps. They found that many lifetime and intensity features differed significantly between cancer and healthy tissue. However, the strongest and most consistent separation came from the label-free channels: shorter lifetimes in the collagen-sensitive band and characteristic shifts in the NADH-sensitive band were tightly linked to cancer. In contrast, the PpIX channel—the one boosted by the administered 5-ALA—gave less reliable information in living tissue. Its measurements varied widely in normal regions, likely because inflamed gums and mucosa either accumulated 5-ALA or contained naturally fluorescent porphyrins that mimicked the tumor signal.
From Operating Room to Bench and Back
When the researchers imaged the removed specimens on the back table, the picture changed slightly. Outside the body, some PpIX-based features became more useful for distinguishing cancer from healthy tissue, suggesting a potential role in “chairside” margin checks. Still, when sophisticated statistical models and machine learning were applied, the best-performing classifiers for both in-surgery and ex-surgery data leaned heavily on the natural autofluorescence signals rather than on the added dye. Label-free FLIm achieved respectable accuracy in telling cancer from normal tissue on its own, while including PpIX added little and sometimes introduced confusion.
What This Means for Future Surgeries
For pet owners and, ultimately, human patients, the key message is that surgeons may one day have a light-based tool that helps them see where cancer ends and healthy tissue begins, without relying on extra drugs or dyes. This work in dogs—a realistic large-animal model of human oral cancer—shows that the body’s own fluorescent signatures can be enough to guide decisions in the operating room. Adding 5-ALA and PpIX in this context did not improve accuracy enough to justify the extra cost, complexity, and potential side effects. The authors conclude that future efforts should focus on refining label-free fluorescence lifetime imaging itself—possibly tailoring the analysis to specific anatomic sites—rather than chasing more contrast agents. If successful, such technology could reduce repeat surgeries and preserve more normal tissue while still keeping cancer under control.
Citation: Goldschmidt, S., Marcu, L., Ehrlich, K. et al. Label-free fluorescence lifetime imaging can distinguish cancer from healthy tissue in spontaneously occurring canine oral tumors. Sci Rep 16, 6077 (2026). https://doi.org/10.1038/s41598-026-37001-3
Keywords: oral cancer imaging, canine oral tumors, fluorescence lifetime, surgical margins, 5-ALA PpIX