Clear Sky Science · en
Cost of illness of Mycoplasma genitalium in Australia: an incidence-based approach incorporating resistance-guided therapy
Why this hidden infection matters
Most people have never heard of Mycoplasma genitalium, a tiny bacterium that spreads through sex and often causes no symptoms. Yet it can quietly inflame the reproductive tract, contribute to pelvic inflammatory disease in women, and is becoming harder to treat because of rising antibiotic resistance. This study asks a simple but important question for Australia: when someone is diagnosed with this infection, which treatment approach not only works better but also costs the health system less?
A quiet germ with growing impact
Mycoplasma genitalium is a sexually transmitted infection that can infect the urethra, cervix, and sometimes the rectum. Because people frequently do not feel sick, many infections go undetected. Unlike some other sexually transmitted infections, it is not routinely reported to national surveillance systems, so the true number of infections in Australia is uncertain. What is known is that the bacterium is linked with painful conditions such as urethritis in men, and cervicitis, pelvic inflammatory disease, and possibly long-term pelvic pain in women. These complications can require clinic visits, antibiotics, and sometimes hospital stays, all of which carry a financial as well as a personal toll.
Antibiotic resistance changes the game
For years, doctors commonly used a single dose of the antibiotic azithromycin to treat Mycoplasma genitalium. Over time, however, the germ has adapted. In Australia and much of the Western Pacific region, more than half of infections now carry genetic changes that make them resistant to azithromycin and related drugs. As a result, people are more likely to remain infected after standard treatment, need extra visits and stronger antibiotics, and may continue to pass the infection to others. This trend forces clinicians and health planners to weigh the benefits of newer, more tailored treatment strategies against their added upfront costs.
Two paths to care: guesswork versus guided treatment
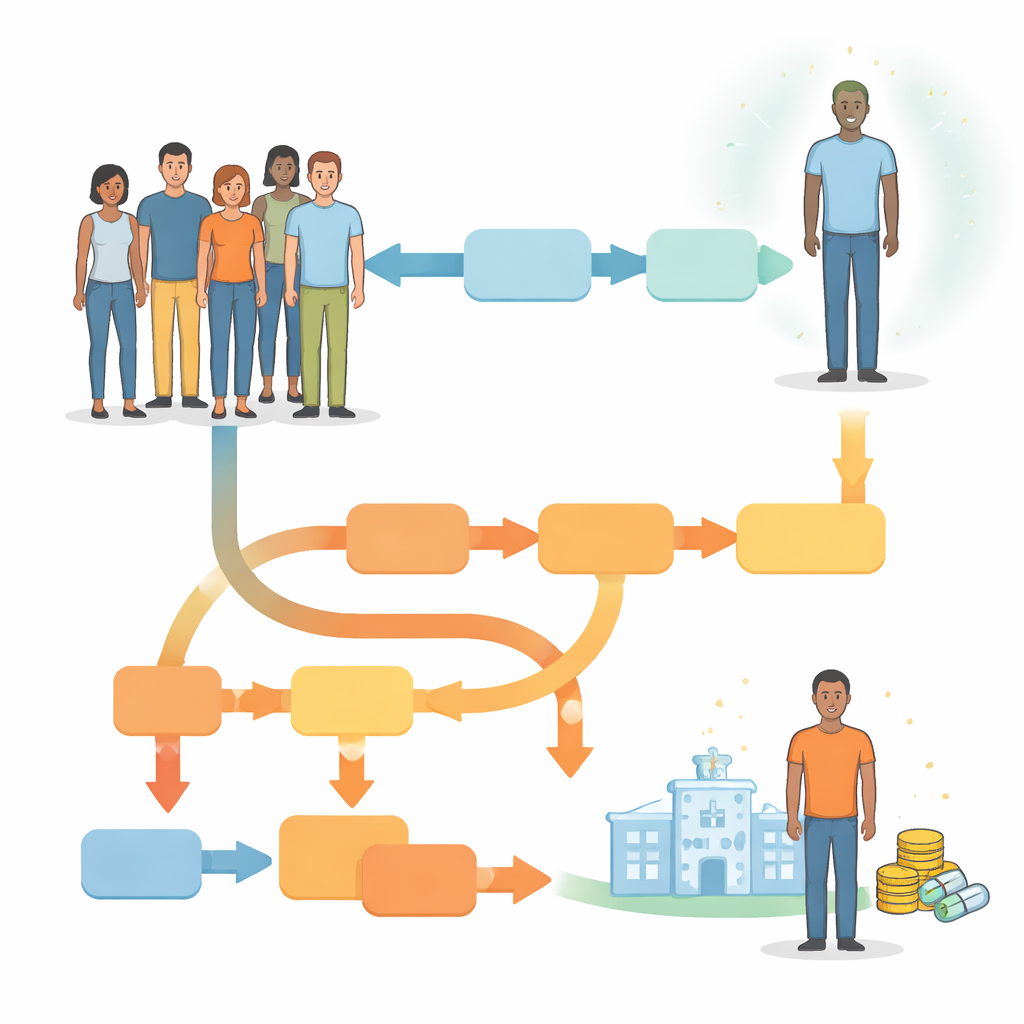
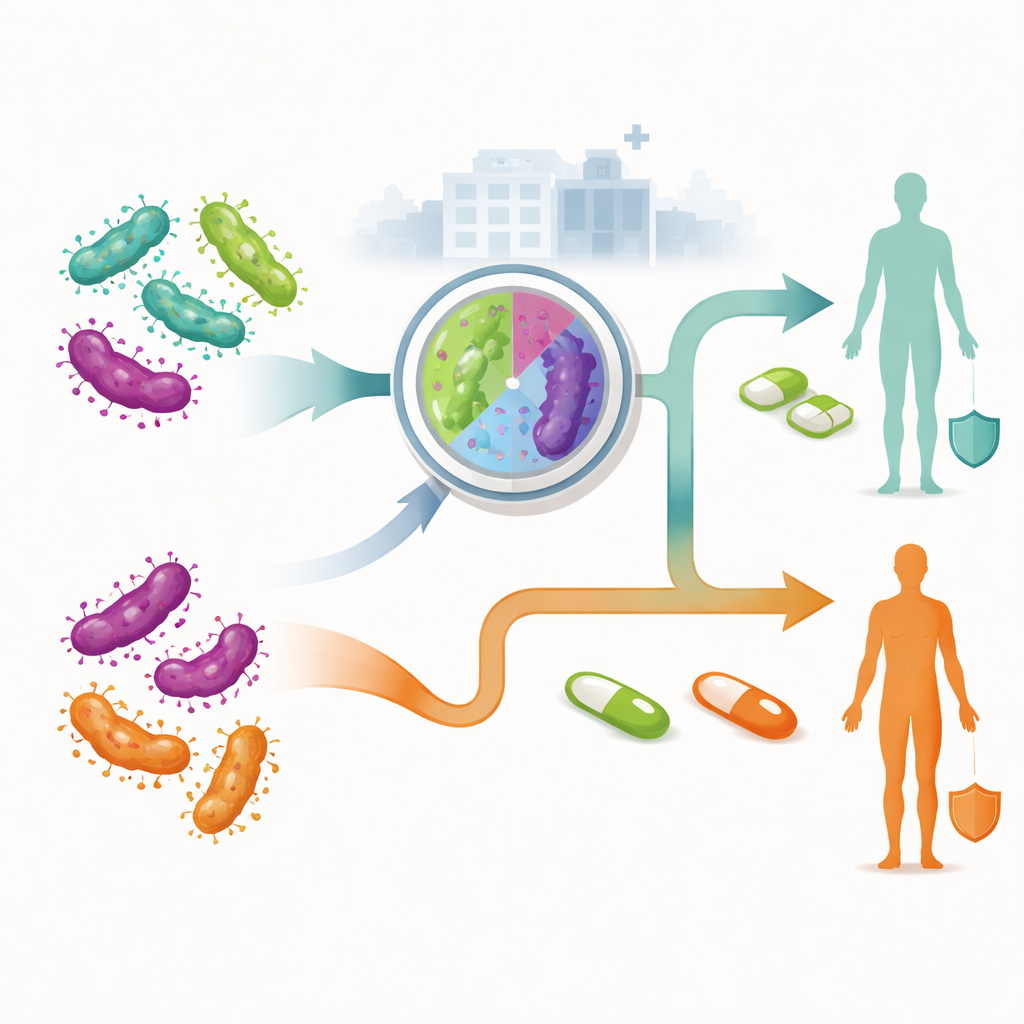
The researchers compared two broad strategies for managing newly diagnosed infections in adults attending Australian services. In the traditional, non-guided approach, everyone receives the same first antibiotic course without knowing whether their strain is resistant. Only if treatment fails do they move on to second- or third-line medicines. In the resistance-guided approach, a laboratory test at diagnosis checks if the germ is likely to resist macrolide antibiotics. People with susceptible infections start on one combination of drugs, while those with resistant infections start on a different regimen that avoids ineffective medicines. The team used a decision tree model to map out each possible route a patient might take, including whether they accept treatment, return for a cure check, need further therapy, or—among women—develop pelvic inflammatory disease and chronic pelvic pain.
Counting the dollars behind each decision
To estimate the cost of a single infection from diagnosis through recovery, the study tallied direct medical expenses: clinic visits, laboratory tests, drug costs, and hospital care for complications. Prices were based on Australian government fee schedules, hospital cost data, and market prices as of 2025. The model was run separately for three groups: non-pregnant women, men who have sex with men, and men who have sex with women, because their rates of resistance and treatment success differ. The researchers also tested how sensitive their results were to uncertainties, such as how common resistance is, how likely people are to come back for follow-up, and how often complications occur.
Fewer complications and lower bills
Across all three groups, resistance-guided care turned out to be cheaper overall, even though some of its drugs cost more per prescription. For women, the expected total cost per infection was about 34 Australian dollars lower with guided therapy than with the traditional approach. Men who have sex with men saw a saving of roughly 39 dollars per person, and men who have sex with women about 23 dollars. Guided treatment produced higher cure rates at the first attempt, meaning fewer repeat visits, fewer courses of antibiotics, and—among women—fewer cases of pelvic inflammatory disease and chronic pelvic pain. Sensitivity tests showed that while certain factors, such as the price of first-line drugs and how common resistance is, influenced the size of the savings, they did not overturn the main result: guided therapy remained the less expensive option.
What this means for patients and health services
Put simply, testing Mycoplasma genitalium for resistance before choosing antibiotics helps doctors match the right drug to the right patient, clearing infections more reliably and, in the long run, saving money for the health system. In a world where antibiotic resistance is steadily rising, this approach also helps slow the spread of hard-to-treat strains by avoiding drugs that no longer work. For patients, that translates into fewer clinic visits, shorter bouts of illness, and a lower chance of serious complications—benefits that extend well beyond the balance sheet.
Citation: Cheng, Q., Watts, C., Al Adawiyah, R. et al. Cost of illness of Mycoplasma genitalium in Australia: an incidence-based approach incorporating resistance-guided therapy. Sci Rep 16, 8034 (2026). https://doi.org/10.1038/s41598-026-36992-3
Keywords: Mycoplasma genitalium, antibiotic resistance, sexually transmitted infection, healthcare costs, resistance-guided therapy