Clear Sky Science · en
Origin of the high coercivity in FeNi inspired magnets
Why new magnets matter
From wind turbines and electric cars to smartphones and medical scanners, powerful permanent magnets quietly keep modern technology running. Today’s strongest magnets rely on rare-earth elements, which are costly to mine and process and raise environmental and geopolitical concerns. This study looks to nature—and to tiny structures inside meteorites—for inspiration, asking a simple question: can we build strong, stable magnets from ordinary metals like iron and nickel, without rare earths, by carefully controlling how the material is put together?
Space rocks as a starting clue
Iron meteorites contain a remarkable iron–nickel material known as tetrataenite, long thought to be a naturally occurring “cosmic” magnet. It forms an orderly atomic pattern that, in theory, should give rise to strong and stable magnetism. However, in meteorites this phase appears only as extremely small crystals embedded in a more complex mixture of other minerals, and it forms over millions of years of slow cooling in space. Reproducing this exotic structure on Earth, at useful scales and in a reasonable time, has proved extremely difficult. Yet some laboratory-made iron–nickel alloys already show surprisingly high magnetic hardness, measured by a property called coercivity, even when the special tetrataenite structure is barely present—or not clearly detectable at all.
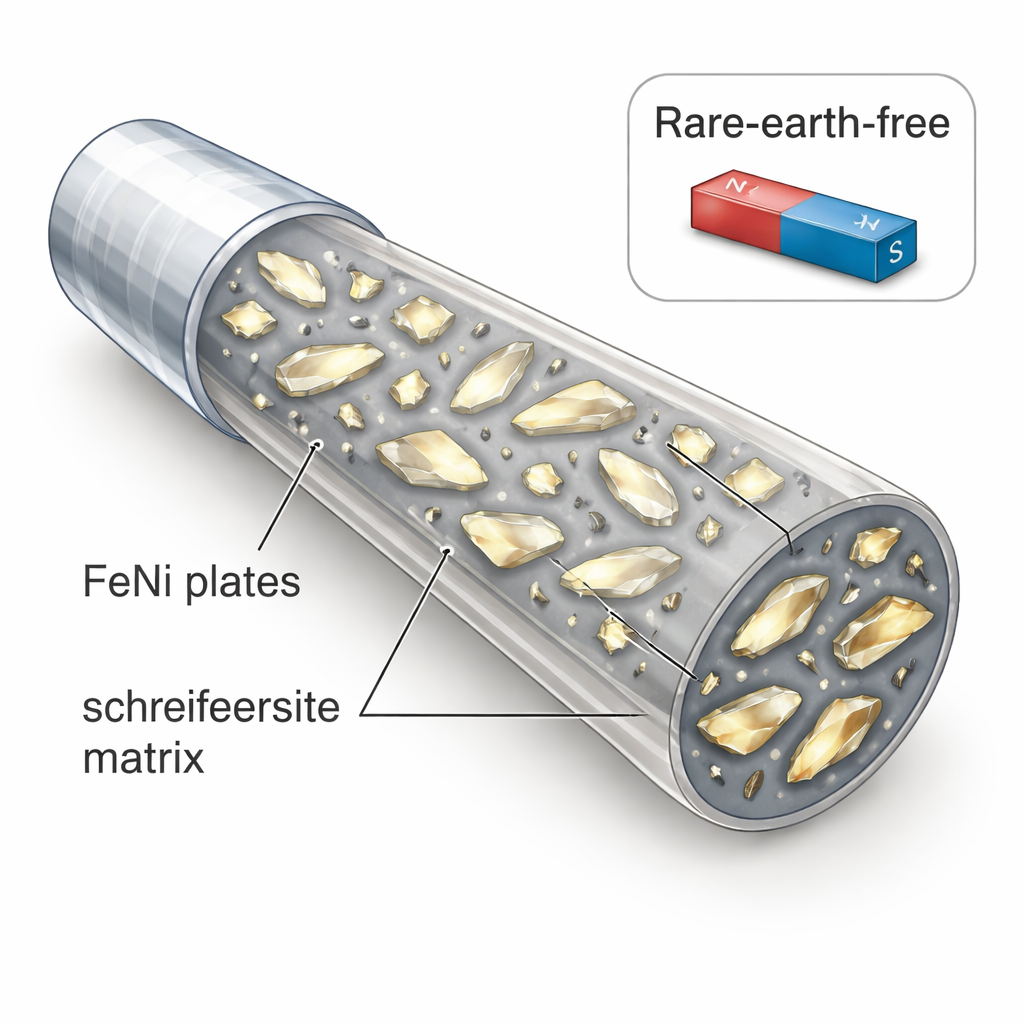
Building tiny wires with simple ingredients
The researchers set out to test whether the unusual magnetic strength seen in some iron–nickel samples really requires this rare ordered phase, or whether it could instead come from how the material is arranged on the nanoscale. They melted together iron, nickel, and phosphorus to make a “master alloy,” then rapidly cast this melt into very thin glass-coated microwires at different cooling speeds. X-ray diffraction experiments and electron microscope images showed that the resulting wires contained only two crystal types: a “soft” iron–nickel phase with a simple cubic atomic arrangement, and a phosphide phase called schreibersite. Importantly, the iron–nickel appeared as extremely small, flat plates—only about 20 nanometres across—dispersed within a continuous schreibersite matrix.
How microstructure turns soft into hard
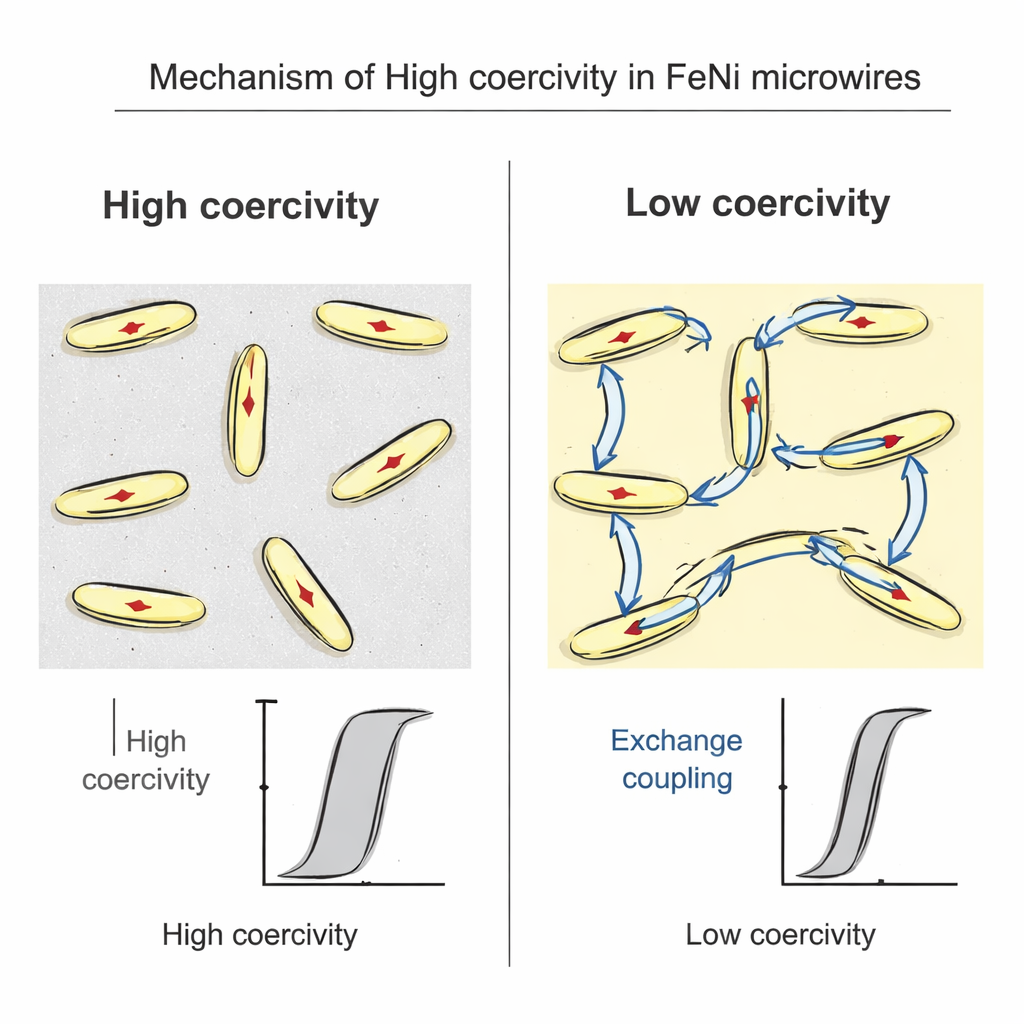
Magnetic measurements at room temperature revealed that these nanostructured microwires, made only of soft iron–nickel embedded in schreibersite, had coercive fields around 400–440 oersteds—values similar to those reported for materials claimed to contain the hard tetrataenite phase. Detailed analysis showed why. Each tiny iron–nickel plate is smaller than the size at which it can split into separate magnetic regions, so it behaves as a single magnetic domain. Because these plates are thin and elongated, their shape strongly resists flipping their magnetic direction, an effect known as shape anisotropy. At the same time, the surrounding schreibersite is not magnetic at room temperature, so it acts like an insulating spacer: it prevents neighboring plates from “talking” to each other magnetically. Together, this single-domain size, plate-like shape, and magnetic isolation make the overall material hard to demagnetize.
When the matrix joins in
The team then explored what happens when the surrounding matrix does become magnetic. By cooling the wires below about 190 kelvin (–83 °C), the schreibersite phase turns ferromagnetic, allowing the iron–nickel plates to couple through it. Under these conditions, the coercivity drops sharply: the once-isolated domains now reverse their magnetization collectively, making the sample much easier to magnetize and demagnetize. A separate set of microwires made from pure iron and a ferromagnetic iron–phosphide matrix showed similarly low coercivity at room temperature. These comparisons clearly demonstrate that the magnetic character of the matrix—whether it is magnetically “silent” or actively connects the grains—plays a central role in setting how hard the magnet really is.
What this means for future magnets
The study concludes that large coercive fields in these Fe–Ni–P microwires do not require the presence of the exotic tetrataenite phase. Instead, they arise primarily from a combination of microstructure and shape: very small, plate-like iron–nickel crystals, each acting as a single domain, dispersed and magnetically isolated in a non-magnetic schreibersite matrix. When the matrix becomes magnetic, the coercivity collapses, revealing that the key to hardness lies in how the grains are arranged and separated, not in some special intrinsic property of a rare ordered phase. For the design of future rare-earth-free magnets, this insight is powerful: by engineering size, shape, and spacing of common metallic phases, it may be possible to achieve robust magnetic performance using abundant elements and scalable processing methods.
Citation: Hernando, A., de la Presa, P., Jiménez-Rodríguez, J.A. et al. Origin of the high coercivity in FeNi inspired magnets. Sci Rep 16, 6014 (2026). https://doi.org/10.1038/s41598-026-36990-5
Keywords: rare-earth-free magnets, iron-nickel alloys, nanocrystalline microwires, magnetic coercivity, microstructure