Clear Sky Science · en
A comprehensive investigation of expired dextromethorphan HBr drug as a carbon steel corrosion inhibitor using gravimetric, electrochemical, and theoretical computational approaches
Turning Old Medicine into New Protection
Most of us think of expired medicines as hazardous trash that should head straight for safe disposal. This study flips that idea on its head by asking: can an out‑of‑date cough syrup ingredient protect the steel that underpins refineries, pipelines, and chemical plants from destructive acid corrosion? By testing expired dextromethorphan hydrobromide (the familiar cough suppressant) as a shield for carbon steel in harsh hydrochloric acid, the researchers explore a surprising way to cut industrial costs and waste at the same time.
Why Steel Needs a Bodyguard
Carbon steel is the workhorse metal of industry, but in acidic environments it corrodes quickly, thinning pipes, weakening tanks, and forcing expensive shutdowns. Hydrochloric acid, widely used to clean and “pickle” steel, is especially aggressive. Companies usually dose these solutions with corrosion inhibitors, many of which are toxic or made from specialty chemicals. In parallel, tons of expired pharmaceuticals are discarded every year, even though many still retain most of their active ingredient. The authors realized that some of these complex, carbon‑rich molecules might stick to steel surfaces and act as a protective film, turning a disposal problem into a useful resource.
A Cough Drug Meets Harsh Acid
The team focused on expired dextromethorphan hydrobromide (EDM‑HBr), a common over‑the‑counter cough medicine component. First, they confirmed by high‑performance liquid chromatography that the expired drug still contained about 92% of its original active substance, meaning it was chemically largely intact. They then dissolved EDM‑HBr into a standard 1 molar hydrochloric acid solution and immersed polished carbon steel samples. By measuring how much weight the steel lost over several hours and by monitoring electrical signals associated with corrosion, they could quantify exactly how much damage the acid caused, with and without the drug present.
How Well the Expired Drug Shields Steel
The results were striking. In plain acid, the steel dissolved rapidly. When EDM‑HBr was added, the corrosion rate fell sharply as the dose increased. At the highest tested concentration (600 milligrams per liter), the steel lost about twenty times less mass, corresponding to roughly 95% protection in weight‑loss tests and over 97% protection in electrochemical measurements. The benefit persisted for days: protection peaked around 12 hours and then slowly declined but still remained above 90% after 72 hours. Tests at higher temperatures showed that some efficiency is lost as heat encourages both faster corrosion and some unpeeling of the protective layer, but the inhibitor still performed very well across the range studied.
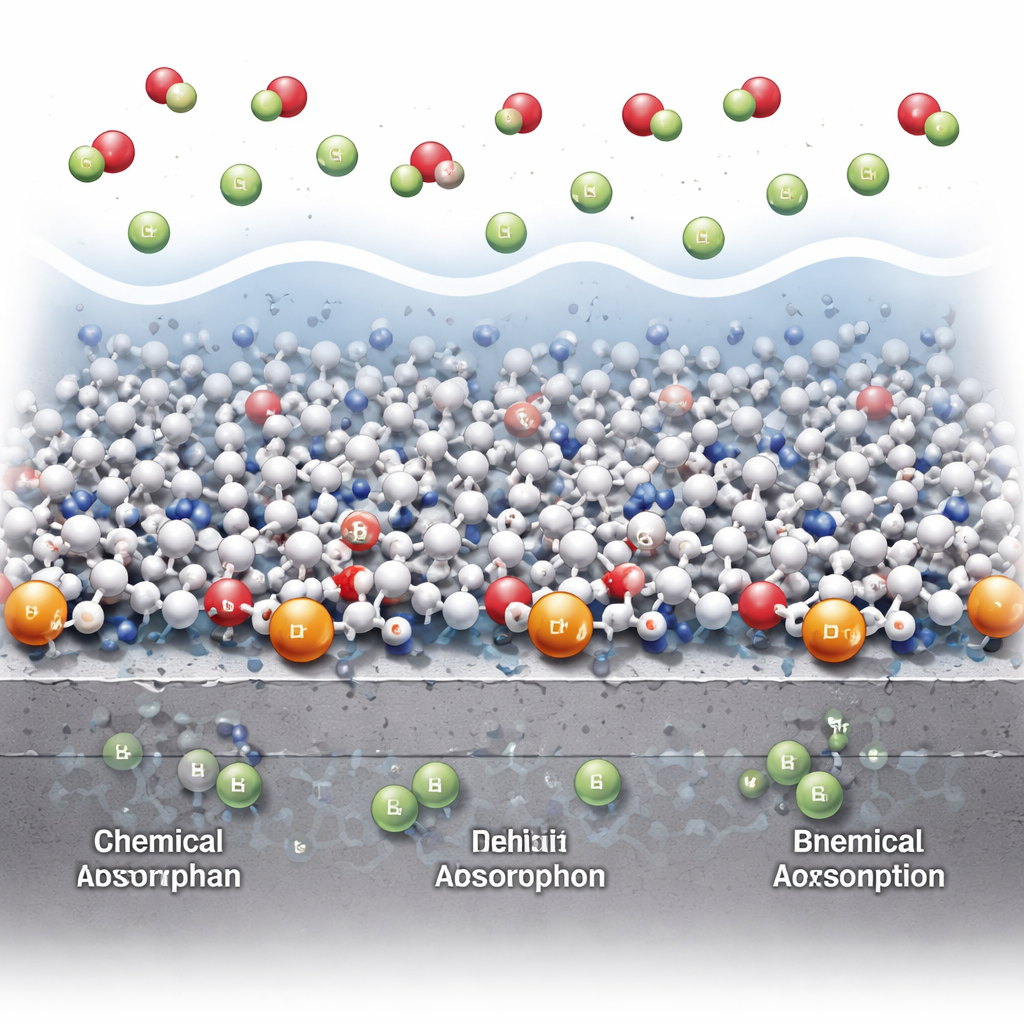
Peeking into the Invisible Film on Steel
To understand what was happening at the steel surface, the researchers analyzed how the protection changed with temperature and concentration and used quantum‑chemical calculations on the EDM‑HBr molecule. The patterns matched a so‑called Freundlich adsorption behavior, which simply means the molecules spread unevenly over a somewhat patchy surface rather than forming a perfectly uniform coat. Energy and entropy measurements suggested that at room temperature the drug mostly clings to the steel through physical attraction—like weak molecular “stickiness”—while at higher temperatures some true chemical bonding through nitrogen and oxygen atoms kicks in. Computer calculations showed that EDM‑HBr has electronic properties that make it especially good at sharing electrons with iron atoms, helping it form a tight, mixed physical‑and‑chemical barrier that keeps acid and chloride ions away from bare metal.
From Waste Medicine to Green Industrial Helper
For a non‑specialist, the takeaway is simple: an expired cough‑syrup ingredient can act as an exceptionally strong rust‑stopper for steel in acid, rivaling or beating other drug‑based inhibitors. Because the drug remains largely intact after expiry and its breakdown products and bromide ions may even boost its grip on steel, it offers a promising way to repurpose pharmaceutical waste. While real‑world deployment will still require checks under flowing conditions, harsher acids, and full environmental safety reviews, this work points toward a future where discarded medicines help protect vital infrastructure instead of filling landfills and incinerators.
Citation: Khafagy, ES., Lila, A.S.A., Ashmawy, A.M. et al. A comprehensive investigation of expired dextromethorphan HBr drug as a carbon steel corrosion inhibitor using gravimetric, electrochemical, and theoretical computational approaches. Sci Rep 16, 3047 (2026). https://doi.org/10.1038/s41598-026-36977-2
Keywords: corrosion inhibitor, expired drugs, carbon steel, hydrochloric acid, green chemistry