Clear Sky Science · en
Broad spectrum antimicrobial nanoparticles with low toxicity to prevent biofilm formation on urologic devices
Why Medical Implants Sometimes Turn Against Us
Modern medicine relies on plastic tubes and implants to drain urine, support blocked kidneys, and restore sexual function. Yet these same devices can become magnets for bacteria, which build slimy protective communities called biofilms. Once a biofilm forms, infections are hard to treat, often forcing doctors to remove or replace the device. This study explores a new kind of microscopic coating, made from gold and silver wrapped in a carbon shell, designed to stop those biofilms before they start.
The Hidden Problem of Sticky Germs
Catheters, ureteral stents, and inflatable penile prostheses help hundreds of thousands of patients, but they also provide ideal real estate for microbes. Bacteria attach to the device surface and secrete a glue-like matrix of sugars, proteins, and fats, forming a fortress that shields them from antibiotics and the immune system. As a result, device-related infections make up more than a quarter of all hospital-acquired infections and cost hundreds of millions of dollars every year. Current protective coatings based on antibiotics or simple silver struggle to prevent biofilms over the long term and may contribute to growing antibiotic resistance.
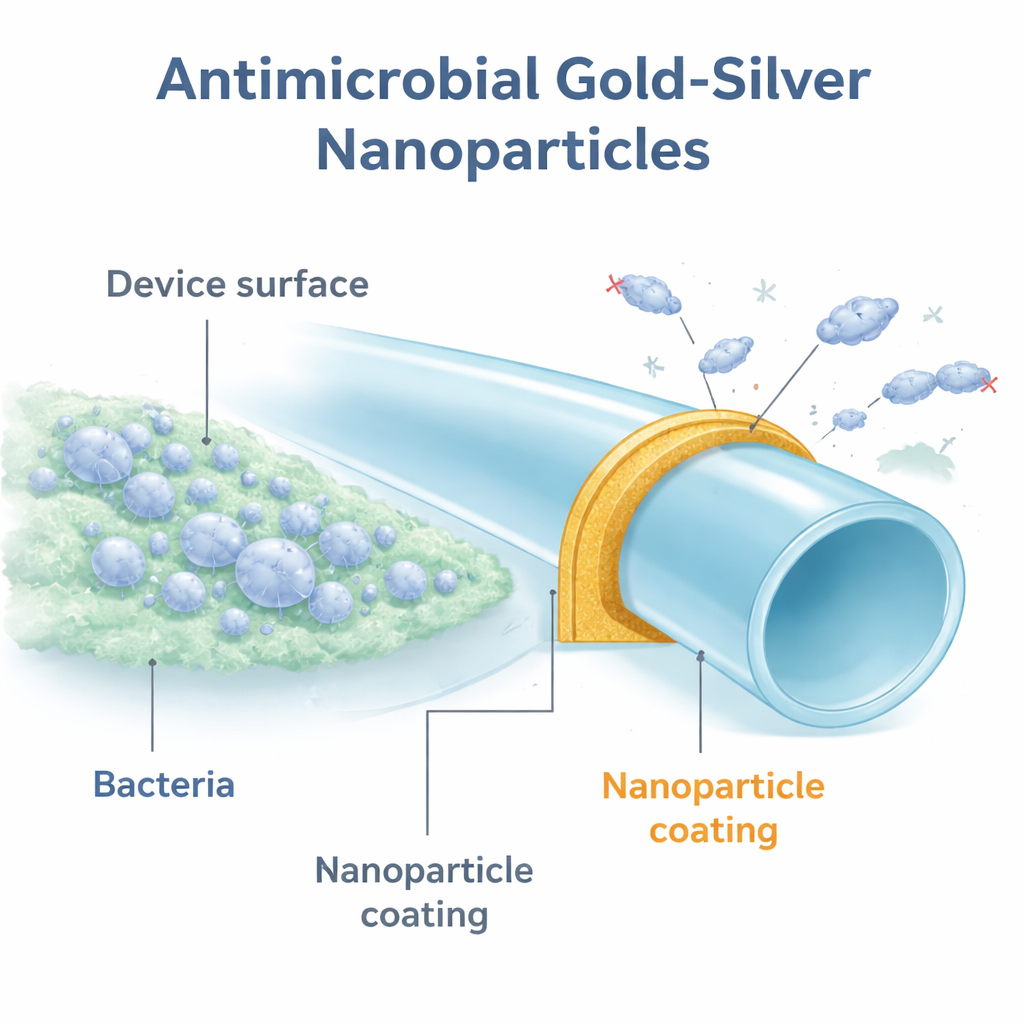
A New Kind of Protective Armor
The researchers focused on tiny metal particles only a few billionths of a meter wide. These nanoparticles were built around a carbon cage material called polyhydroxy fullerene, known to be non-toxic and to boost the performance of certain metals. They created several versions using different metals, then coated small discs of polyurethane, a plastic commonly used in urologic devices. When they tested these coated discs against a standard strain of Escherichia coli, only the silver-containing particles showed strong antibacterial effects. The clear standout was a gold–silver combination, referred to as GSNPs, which cut bacterial numbers by up to 100,000-fold compared with uncoated plastic.
Stopping Real-World Urologic Germs
To see if this effect held up beyond a laboratory strain, the team tested their best coating against six types of bacteria taken directly from infected ureteral stents and penile prostheses. These included both Gram-positive and Gram-negative species that are known to cause stubborn infections. At bacterial levels similar to those found in patients, the GSNP-coated surfaces achieved a 100% reduction in both free-floating and surface-attached bacteria for every strain tested. In separate experiments, the researchers exposed bacteria to different nanoparticle concentrations in liquid and estimated how much GSNP was needed to stop growth. All strains were fully inhibited at or below 3 micrograms of GSNP per milliliter of liquid, with slightly higher amounts required for some Gram-positive organisms.
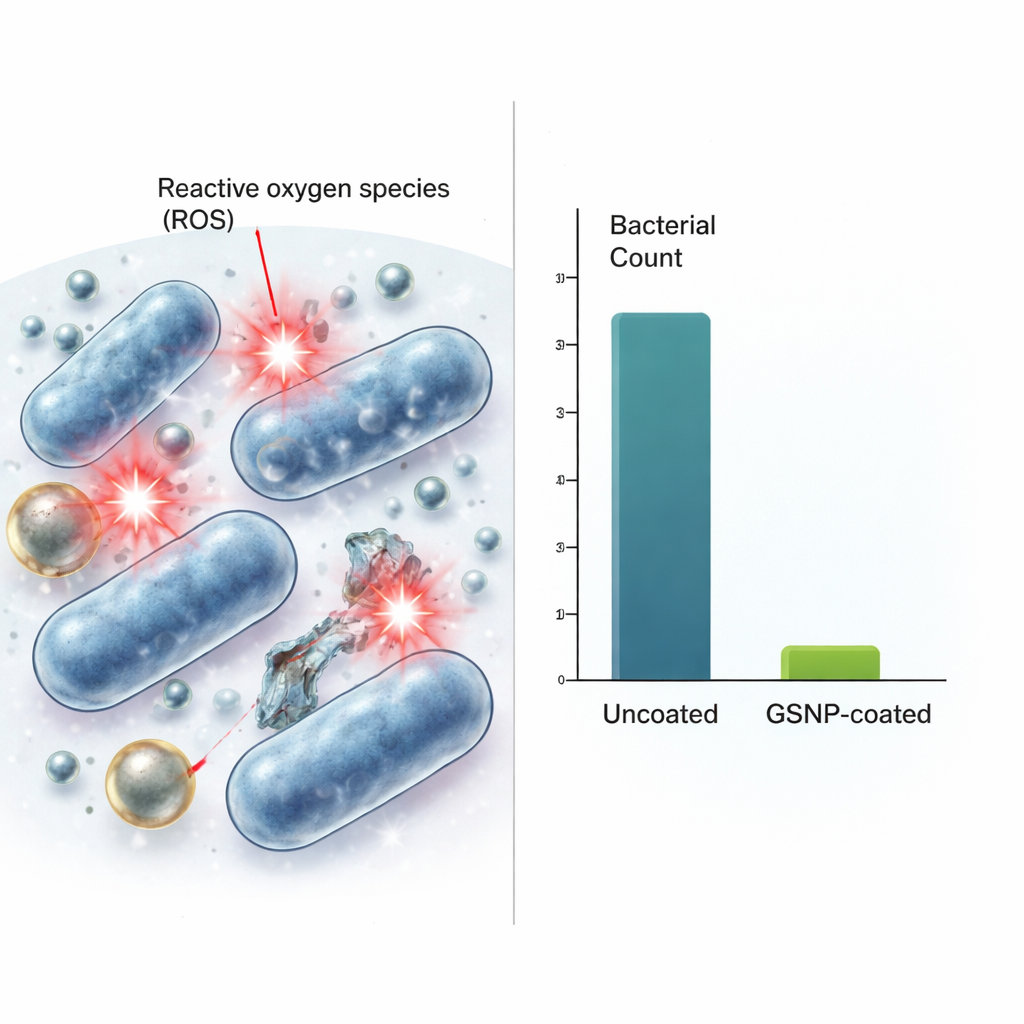
How They Work and How Safe They Are
The team next asked how these coatings kill bacteria and whether they might also harm human cells. Their data point to reactive oxygen species—chemically reactive forms of oxygen—as a key part of the killing mechanism. While the nanoparticles alone did not generate these reactive molecules, bacteria exposed to GSNPs produced much higher levels than untreated bacteria, suggesting the particles trigger damaging chemistry inside or near the microbes. When mouse fibroblast cells were exposed to similar or higher concentrations of GSNPs, more than 80% of the cells remained alive at the worst-case antibacterial dose. In other words, the levels that wiped out bacteria had only a modest impact on mammalian cells. The GSNPs were also dramatically more effective than a commercial gold–silver nanoparticle product tested side by side, achieving roughly a million-fold greater bacterial reduction at equivalent doses.
What This Could Mean for Patients
Although this work was done in the lab and not yet in animals or people, the findings point to a promising new strategy for safer medical implants. A thin layer of GSNPs on a catheter or prosthesis could provide broad protection against many different bacteria, reduce the need for device removal, and do so without relying on traditional antibiotics. The coating uses low amounts of metal, shows relatively low toxicity in initial tests, and is straightforward to make. With further engineering to improve long-term stability and careful testing in realistic models, these gold–silver nanoparticle coatings could help keep vital urologic devices working for patients instead of becoming a home base for infection.
Citation: Rodriguez-Alvarez, J.S., Xu, Y., Gutierrez-Aceves, J. et al. Broad spectrum antimicrobial nanoparticles with low toxicity to prevent biofilm formation on urologic devices. Sci Rep 16, 7333 (2026). https://doi.org/10.1038/s41598-026-36969-2
Keywords: biofilm, urologic devices, antimicrobial coating, nanoparticles, gold-silver