Clear Sky Science · en
Quantitative proteomic analysis of plasma after remote ischemic conditioning in acute ischemic stroke
Why squeezing an arm might help protect the brain
When someone has a stroke caused by a blocked blood vessel, every minute of lost blood flow threatens brain cells. Standard treatments aim to reopen the blocked artery, but many patients either arrive too late or are not eligible. Researchers are exploring a surprisingly simple add‑on therapy: briefly cutting off blood flow to an arm or leg with a blood‑pressure cuff, a strategy called remote ischemic conditioning (RIC). This study asks a key question for turning that idea into real therapy: what exactly does RIC change in the blood of stroke patients, and could those changes point to who might benefit most?
A gentle stress signal sent from limb to brain
RIC works by repeatedly inflating a cuff on a limb for a few minutes, then deflating it, creating short, harmless episodes of low blood flow. Animal experiments and early clinical trials suggest this “controlled stress” can signal distant organs, including the brain, to switch on protective programs. In a large clinical trial called RICAMIS, patients with moderate ischemic stroke who received RIC in addition to usual care did better at 90 days than those who received usual care alone. Yet another trial (RESIST) did not see such a benefit, leaving doctors unsure when and for whom RIC really works. The current study digs into patients’ blood to look for molecular clues—measurable proteins, or biomarkers, that change over time with RIC.
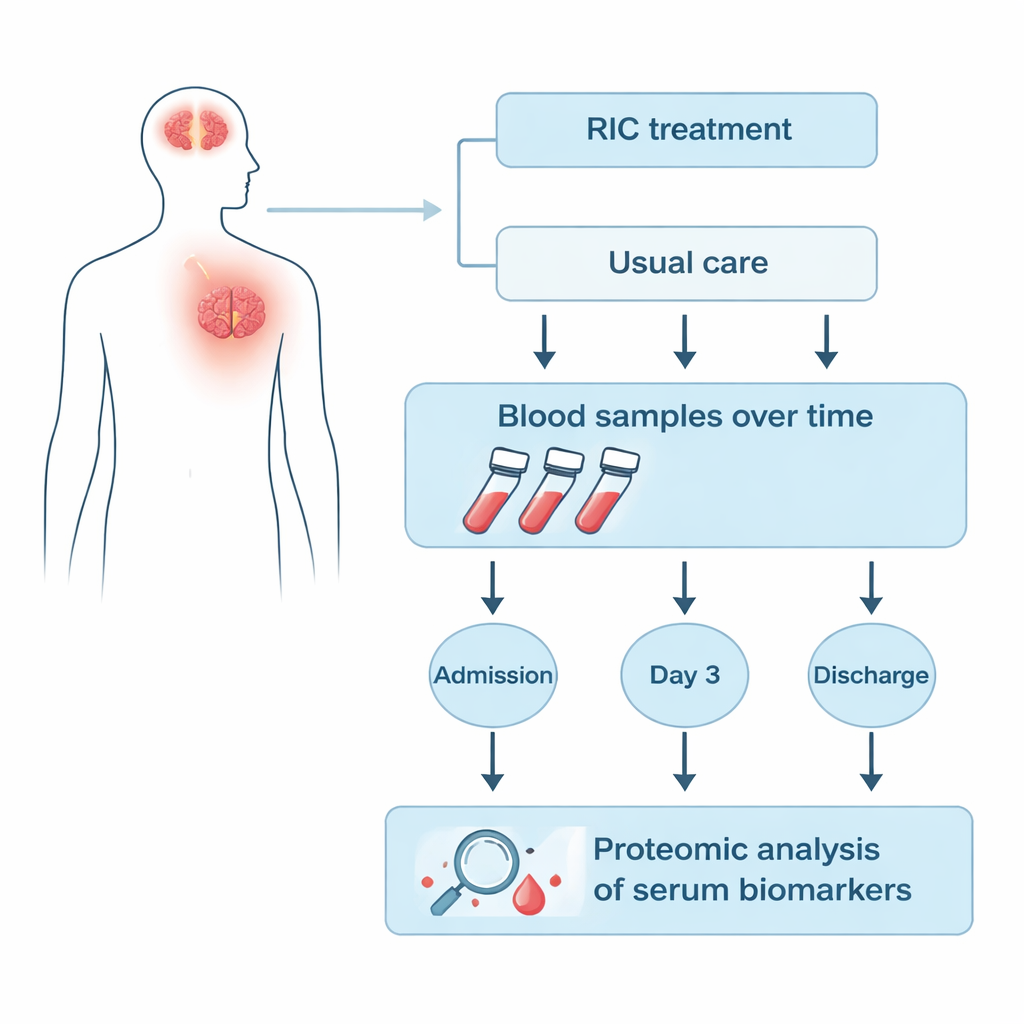
Following patients’ blood through their hospital stay
The researchers focused on 25 patients from the RICAMIS trial who had blood samples collected at three points: on admission, three days after treatment was assigned, and at hospital discharge. Nine patients received RIC plus usual care, while sixteen received usual care alone. Using a technique called quantitative proteomics, the team measured hundreds of proteins in the serum portion of the blood all at once, rather than looking at just a few pre‑selected markers. They then compared how each protein’s level rose or fell over time in the RIC group versus the control group to find signatures that were uniquely linked to RIC.
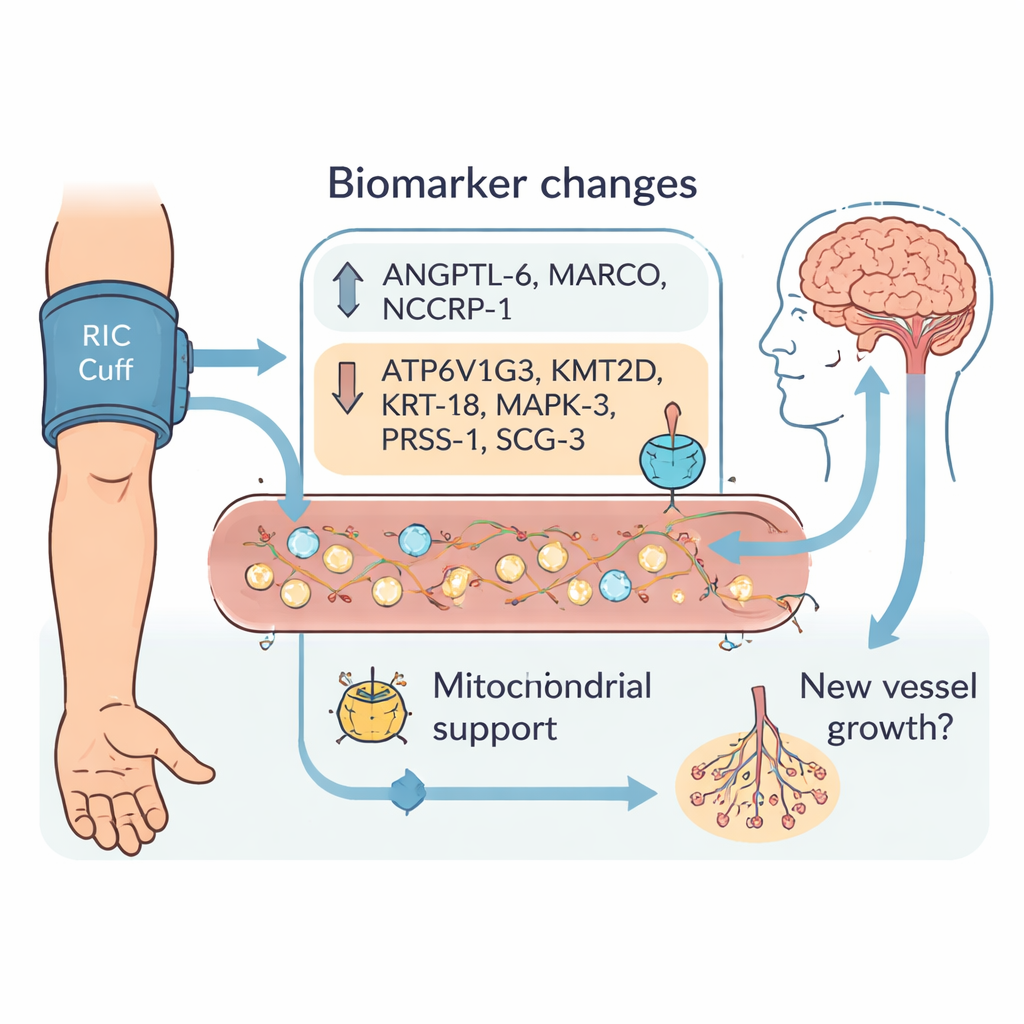
Nine stand‑out signals in the bloodstream
Across the hospital stay, dozens of proteins shifted differently between the two groups. After careful filtering, the team highlighted nine biomarkers whose changes from admission to day 3 and to discharge clearly separated RIC‑treated patients from controls. Three proteins—ANGPTL‑6, MARCO, and NCCRP‑1—increased more with RIC. Six others—ATP6V1G3, KMT2D, KRT‑18, MAPK‑3, PRSS‑1, and SCG‑3—tended to decrease. Many of these molecules are involved in how cells manage energy in their tiny power plants, the mitochondria, or in how new blood vessels grow and remodel. This fits with the idea that RIC does not simply shield neurons from damage, but may help injured brain regions reorganize, repair their blood supply, and recover function over days to weeks.
Hints about energy use and new vessel growth
To better understand what these nine proteins might be doing, the authors mapped them onto known biological pathways. Two of them, KMT2D and MAPK‑3, are connected to how DNA is packaged and how genes are switched on or off, especially in programs related to vessel growth. Others, like ATP6V1G3 and KRT‑18, have been linked in prior work to maintaining healthy mitochondria and protecting heart tissue after injury. Together, these patterns point toward RIC nudging the body toward more efficient energy use and encouraging the growth or remodeling of blood vessels—potentially improving blood flow around the stroke‑damaged area. At the same time, some markers, such as NCCRP‑1 and MARCO, may reflect the body’s acute stress and immune responses to the cuff‑inflation procedure itself, underscoring that RIC triggers a complex, whole‑body reaction.
What this means for future stroke care
This small exploratory study cannot prove exactly how RIC protects the brain, and it involved only 25 patients from a single country, so the findings need confirmation in much larger and more diverse groups. Still, identifying a short list of nine proteins that consistently move in opposite directions with and without RIC is an important step. In the future, such biomarkers could help doctors quickly tell whether RIC is “taking hold” in a given patient, guide the choice of who should receive it, or even inspire drug treatments that mimic its beneficial effects without the cuff. For now, the work strengthens a simple but powerful idea: by safely stressing one part of the body, we may be able to coax the brain into a state that is more resilient and better able to heal after stroke.
Citation: Cui, Y., Liu, F., Cai, JR. et al. Quantitative proteomic analysis of plasma after remote ischemic conditioning in acute ischemic stroke. Sci Rep 16, 6106 (2026). https://doi.org/10.1038/s41598-026-36968-3
Keywords: acute ischemic stroke, remote ischemic conditioning, serum biomarkers, proteomic analysis, neuroprotection