Clear Sky Science · en
Structural and functional foetal renal abnormalities as a mechanism of oligohydramnios in a guinea pig model of growth restriction
Why the Womb’s Water Matters
Late in pregnancy, a baby floats in a protective bath of fluid that cushions movement and reflects how well the baby is doing. When that pool shrinks too much—a condition called low amniotic fluid—it signals trouble and raises the risk of complications at birth. Doctors know that small, growth‑restricted babies often have too little fluid, but the exact reason has been unclear. This study uses pregnant guinea pigs to show that, beyond problems with the placenta and blood flow, the baby’s own kidneys can be structurally damaged, reducing urine production and helping to dry up that vital pool.
Small Babies and a Shrinking Fluid Pool
The researchers created a model of poor fetal growth by modestly limiting the food given to pregnant guinea pigs. Compared with well‑fed mothers, undernourished mothers had smaller fetuses and smaller placentas, mimicking human growth restriction. Crucially, the volume of fluid around these fetuses fell by about half, even after accounting for their lower body weight. Because most amniotic fluid in mid‑to‑late pregnancy is made from fetal urine, this sharp drop suggested that something was going wrong with how the fetal kidneys were working, not just how much blood they were receiving.
Looking Inside Tiny Kidneys
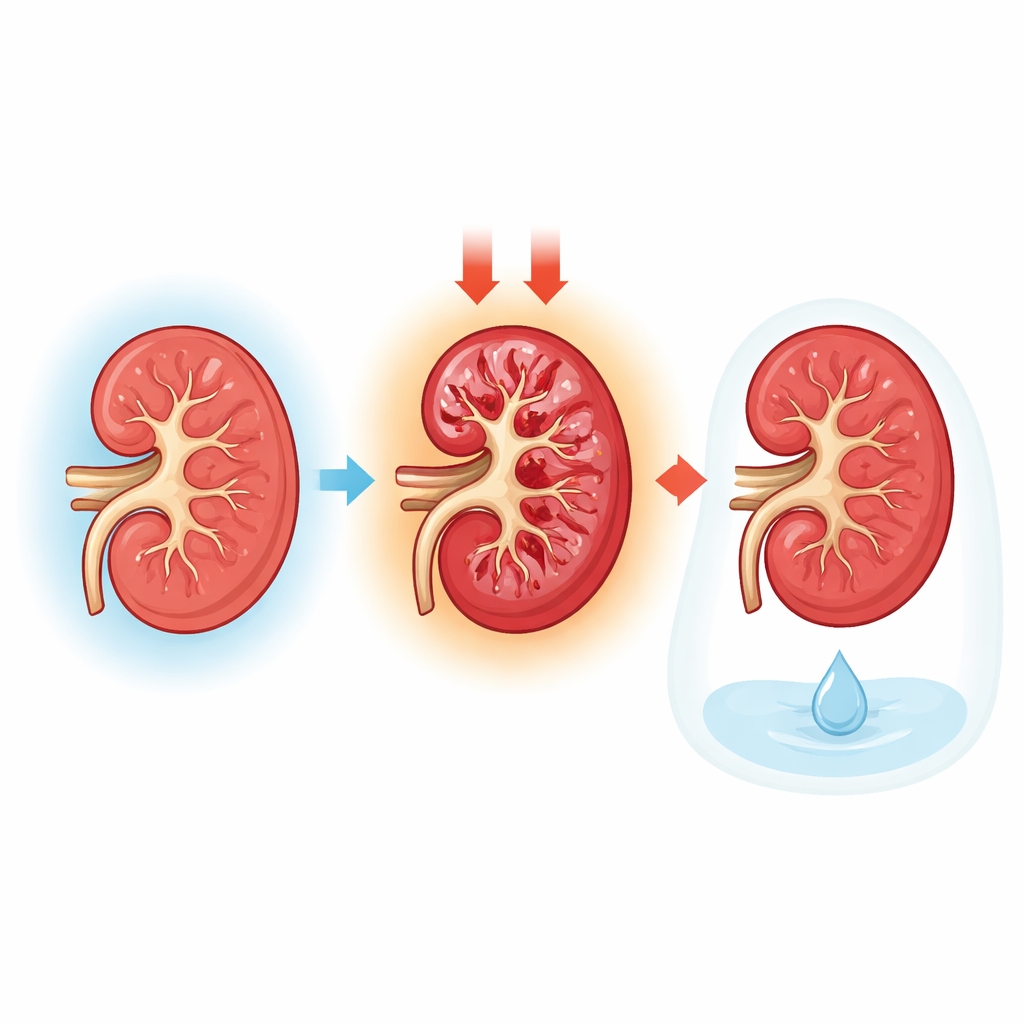
To find out what was happening at a tissue level, the team examined the fetal kidneys under the microscope. In well‑grown fetuses, the filtering units and tiny tubes that process urine were neatly organized and intact. In growth‑restricted fetuses, by contrast, the kidney tissue showed clear signs of injury: tubules were flattened and swollen, cells had lost their nuclei, and debris had collected inside the tubular spaces. High‑magnification imaging revealed that key filter cells on the surface of the glomeruli had thinned or disappeared, and the supporting membranes of both filters and tubules were frayed or broken. Together, these changes point to filters that leak and tubules that can no longer properly reclaim water and other useful substances.
Chemical Clues in the Surrounding Fluid
The team also searched the amniotic fluid for chemical fingerprints of kidney damage. They measured three proteins that normally are kept out of the urine or reabsorbed by healthy tubules. In the growth‑restricted group, all three were markedly higher when adjusted for creatinine, a standard way to account for concentration. Elevated albumin suggested that the kidney filters were letting protein escape. Higher levels of cystatin C and a protein called L‑FABP suggested stressed and injured tubule cells struggling with their workload. Blood tests from the fetuses showed increased urea, another sign that overall kidney function was strained, even though a more stable marker, creatinine, had not yet risen.
Kidneys Fighting Back Under Stress
Beyond structural damage, the researchers looked for signals of inflammation and repair within the kidneys. They found increased amounts of two immune messenger molecules, IL‑6 and IL‑10, and activation of a downstream signaling protein, STAT3, especially in the kidney tubules. In other kidney diseases, these signals can play a protective role, helping tissues cope with injury. Their presence here suggests that the fetal kidneys are not only being harmed by poor nutrition and reduced oxygen, but are also mounting a biological response that may limit or reshape the damage, with unknown consequences after birth.
What This Means for Pregnancies at Risk
Taken together, these findings paint a picture in which low amniotic fluid in growth‑restricted pregnancies is not simply the result of less blood reaching the kidneys. Instead, the kidneys themselves are structurally and functionally compromised, so they filter blood and handle urine less effectively, and overall urine output drops. The same molecules that leaked into the surrounding fluid in guinea pigs could serve as early warning signals in human pregnancies, helping doctors identify fetuses whose kidneys are under strain. Ultimately, recognizing the baby’s kidneys—not just the placenta—as a key player in low amniotic fluid may open the door to new ways of monitoring and protecting vulnerable pregnancies.
Citation: Inatomi, A., Katsura, D., Tokoro, S. et al. Structural and functional foetal renal abnormalities as a mechanism of oligohydramnios in a guinea pig model of growth restriction. Sci Rep 16, 8285 (2026). https://doi.org/10.1038/s41598-026-36964-7
Keywords: fetal growth restriction, amniotic fluid, kidney injury, maternal undernutrition, pregnancy complications