Clear Sky Science · en
Effectively detect doxycycline in variable sample fields through simple fluorescence strategy using nitrogen doped carbon dots as probe
Why tracking a common antibiotic matters
Doxycycline is a workhorse antibiotic used to treat everything from pneumonia to farm animal infections. Yet if too much lingers in the body—or in food and water—it can damage teeth, fuel antibiotic resistance and threaten long‑term health. The paper behind this summary describes a relatively simple light‑based test that can quickly measure doxycycline in blood, dairy products and tap water, potentially giving doctors, farmers and regulators a faster way to know when and where this drug is present.
Tiny glowing dots as smart sensors
At the heart of the method are “carbon dots”: nanometer‑scale specks of carbon that glow under ultraviolet light. The researchers made a special version called nitrogen‑doped carbon dots using two inexpensive, food‑related ingredients—citric acid and the amino acid glutamine. By heating these in water in a sealed vessel, then purifying and drying the product, they obtained uniform, water‑soluble particles only a few billionths of a meter across. These dots shine with blue fluorescence, are stable over a wide range of acidity, salt levels and temperatures, and keep their brightness for long storage, making them suitable for routine testing.
How doxycycline dims the glow
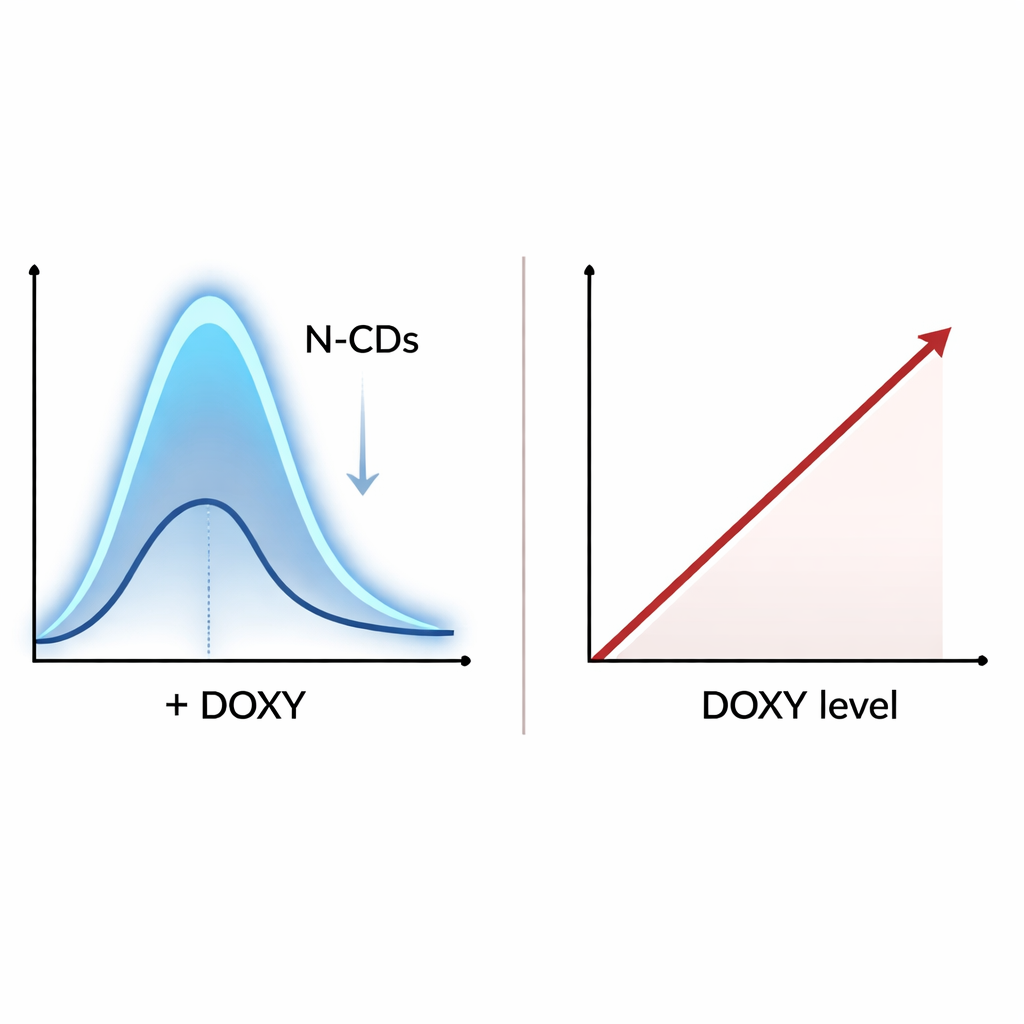
The team discovered that when doxycycline is present, it makes the carbon dots’ blue glow fade in a predictable way. Careful optical measurements showed that doxycycline’s own light‑absorbing bands overlap with the wavelengths used to excite and observe the dots. Instead of directly “poisoning” the dots, the drug mainly acts as a light filter, soaking up some of the incoming or outgoing light in a process called an inner filter effect. Tests of how fast the dots’ light decays, and how their absorption spectra change, ruled out other common quenching mechanisms, confirming that this filtering interaction dominates. The stronger the filter—meaning the more doxycycline in the sample—the weaker the observed fluorescence from the dots.
Tuning the test for real‑world use
To turn this effect into a practical assay, the authors optimized conditions such as solution chemistry, temperature and reaction time. They found that a mild phosphate buffer at slightly acidic pH, a temperature of 30 °C and a 20‑minute incubation gave the most reliable response. Under these conditions, the decrease in light from the dots scaled linearly with doxycycline concentration over a useful range, from 0.1 to 35 micrograms per milliliter. The method could detect levels as low as 0.018 micrograms per milliliter—sensitive enough for both medical monitoring and contamination checks. Importantly, common salts, sugars, amino acids, metal ions like calcium, and many other antibiotics barely affected the signal, indicating that the test is selective for doxycycline and closely related tetracycline drugs.
From hospital blood to farm and tap
The researchers then challenged their sensor with complex real‑world samples. They added known amounts of doxycycline to diluted human plasma from volunteers, to commercial milk and infant formula, and to tap water. After a simple dilution step, the same fluorescence protocol recovered between about 90 and 108 percent of the added drug, with very low variation between repeated measurements. When they compared results from blood and water samples against standard high‑performance liquid chromatography—a gold‑standard but more expensive lab method—the numbers closely matched, supporting the accuracy of the carbon‑dot assay.
What this means for everyday life
In plain terms, this work shows that tiny glowing carbon dots can act like a quick‑response “dimmer switch” for doxycycline: the more antibiotic is present, the more the light from the dots fades. Because the dots are easy to make from cheap ingredients and give a strong, stable signal, the method could be adapted into relatively simple instruments for clinics, food testing labs or environmental monitoring stations. That could help doctors fine‑tune doses in patients, help dairies and regulators keep drug residues out of milk and formula, and help track antibiotic pollution in waterways—supporting both safer treatments and a more cautious use of these powerful drugs.
Citation: Xu, L., Lin, X., Li, L. et al. Effectively detect doxycycline in variable sample fields through simple fluorescence strategy using nitrogen doped carbon dots as probe. Sci Rep 16, 6199 (2026). https://doi.org/10.1038/s41598-026-36947-8
Keywords: doxycycline detection, fluorescence sensor, carbon dots, antibiotic residues, water and food safety