Clear Sky Science · en
A pro-inflammatory neutrophil subpopulation drives intestinal ischemia–reperfusion injury via the ATF4-mediated endoplasmic reticulum stress pathway
When Blood Flow Hurts the Gut
Surgeons and critical care doctors have long puzzled over a dangerous paradox: when blood supply to the intestine is cut off and then restored, the very act of restoring flow can severely damage the gut lining. This process, called intestinal ischemia–reperfusion injury, can lead to life‑threatening complications such as sepsis and multi‑organ failure. The study summarized here reveals that not all immune cells rushing to the scene are equal—one especially aggressive subset of white blood cells plays a starring role in turning a lifesaving response into a harmful one.
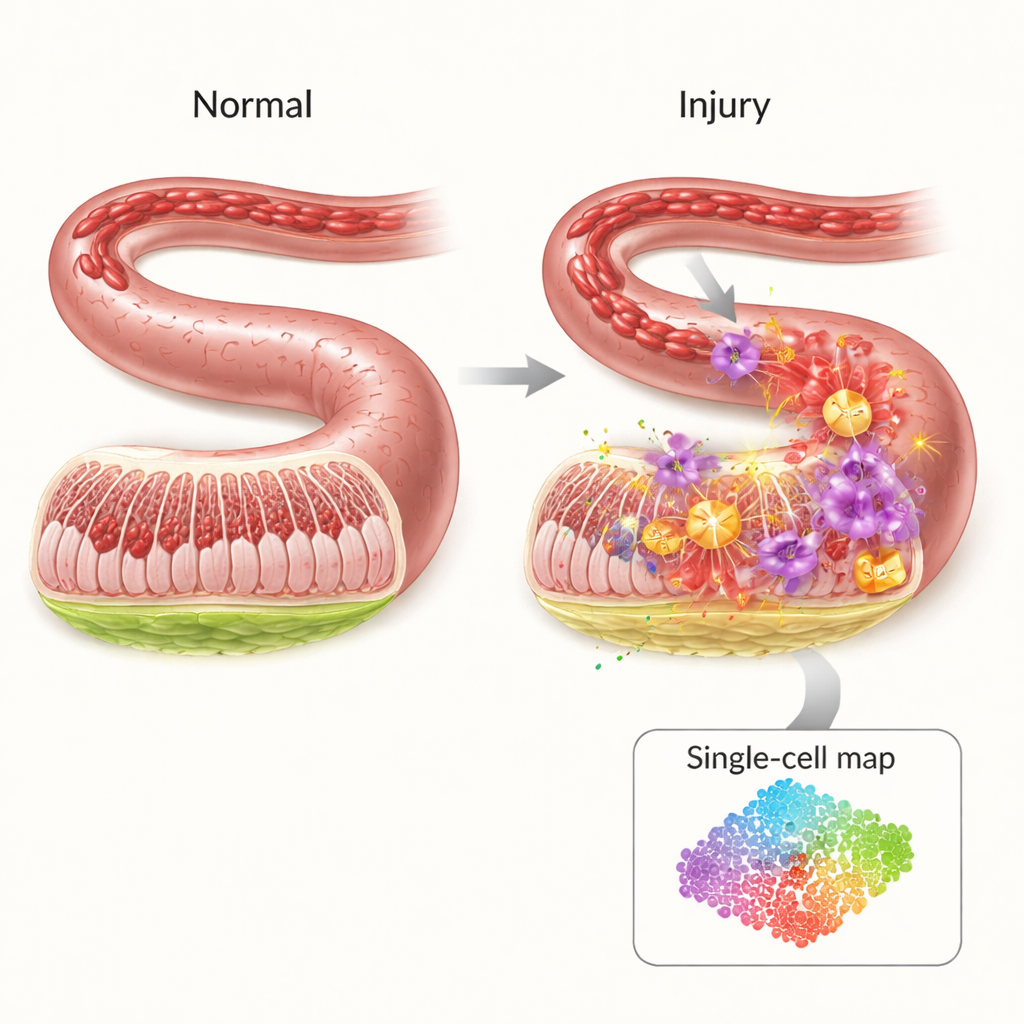
The Gut’s Fragile Protective Wall
The inside of the intestine is lined by a thin but highly organized barrier that keeps bacteria and toxins confined to the gut while allowing nutrients to pass through. When blood flow is interrupted—for example during shock, major surgery, or severe trauma—this barrier is starved of oxygen. Surprisingly, most of the damage occurs not during the lack of blood itself but when circulation returns. The sudden rush of oxygen and immune cells unleashes a storm of inflammatory signals and reactive molecules that can poke holes in the intestinal wall, allowing germs to leak into the bloodstream. Despite its clinical importance, doctors today have few targeted treatments to prevent this cascade.
Zooming In on Individual Cells
To uncover who does what during this crisis, the researchers used single‑cell RNA sequencing, a technique that reads out which genes are active in thousands of individual cells at once. In a mouse model of intestinal ischemia–reperfusion, they compared injured intestines to healthy ones and mapped 19 major cell types. They found that certain immune cells—especially neutrophils and inflammatory monocytes—expanded dramatically after injury, while protective cells such as T cells, B cells, and natural killer cells declined. Communication networks between cell types also shifted, with neutrophils emerging as central hubs sending and receiving many inflammatory signals, pointing to them as key drivers of the damage.
A Harmful Subset of First‑Responder Cells
Neutrophils are among the body’s fastest first responders, rushing through the bloodstream to tackle infections and clear debris. But this study shows that within the neutrophil population, a particularly aggressive subgroup is responsible for much of the collateral damage in the gut. When the scientists depleted neutrophils in mice before inducing ischemia–reperfusion, the animals had less intestinal shortening, lower levels of injury markers in the blood, better tissue appearance under the microscope, and tighter sealing proteins in the gut lining. In co‑culture experiments, neutrophils taken from injured mice directly harmed intestinal cells, lowering their survival, increasing cell death, and disrupting key “zipper” proteins that keep the intestinal barrier intact.
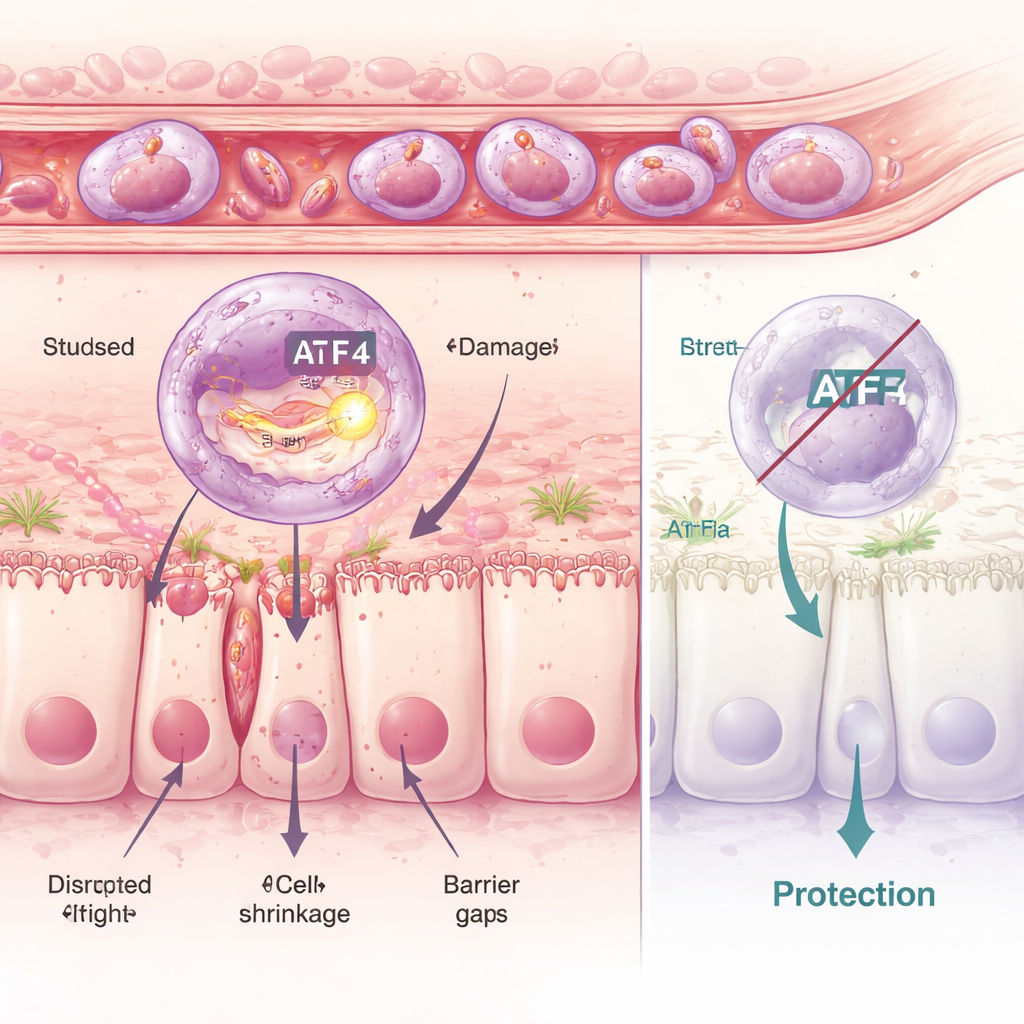
Stress Inside Cells: When Quality Control Goes Wrong
Diving deeper, the researchers examined neutrophils one by one and identified six distinct subgroups. One, called the C5 cluster, stood out for its strong inflammatory profile and for turning on a molecular stress program inside a structure called the endoplasmic reticulum, the cell’s protein‑folding workshop. A key controller of this program is a protein named ATF4. In the harmful C5 cells, ATF4 and its target genes were highly active. By using drugs that either ramp up or damp down this internal stress response, the team showed that more stress meant more gut damage, while blocking it preserved the intestinal barrier. Mice genetically engineered to lack ATF4 were largely protected: even when stress‑inducing drugs were given, their neutrophils could no longer fully execute their damaging program, and the gut lining remained far more intact.
What This Means for Future Treatments
Together, these findings paint a clear picture for non‑specialists: among the swarm of immune cells that flood the gut when blood flow returns, a specific, stress‑primed subset of neutrophils acts like an overzealous clean‑up crew that tears down the building along with the debris. Their destructive power depends on a built‑in stress pathway controlled by ATF4 inside the cells. Turning down this pathway—without wiping out all neutrophils—could offer a way to protect the intestine during high‑risk events such as major surgery, severe infection, or trauma. While more work is needed to confirm these results in humans and to find safe, precise drugs, the study opens a promising route toward therapies that keep the gut barrier intact when patients need it most.
Citation: Yang, Y., Zhou, Q., Liu, S. et al. A pro-inflammatory neutrophil subpopulation drives intestinal ischemia–reperfusion injury via the ATF4-mediated endoplasmic reticulum stress pathway. Sci Rep 16, 6117 (2026). https://doi.org/10.1038/s41598-026-36938-9
Keywords: intestinal barrier, ischemia reperfusion, neutrophils, endoplasmic reticulum stress, ATF4