Clear Sky Science · en
High purity lithium recovery from spent lithium-ion batteries using commercial nanofiltration membranes: a comparative performance assessment
Why Old Batteries Still Matter
Millions of lithium-ion batteries power our phones, laptops, and electric cars, and most will eventually be thrown away. Inside each “dead” battery, however, there is still valuable lithium and other metals that could be reused instead of mined from the earth. This study explores a water-based filtering approach that can pull high-purity lithium out of battery waste, helping to turn yesterday’s gadgets into tomorrow’s clean-energy materials.
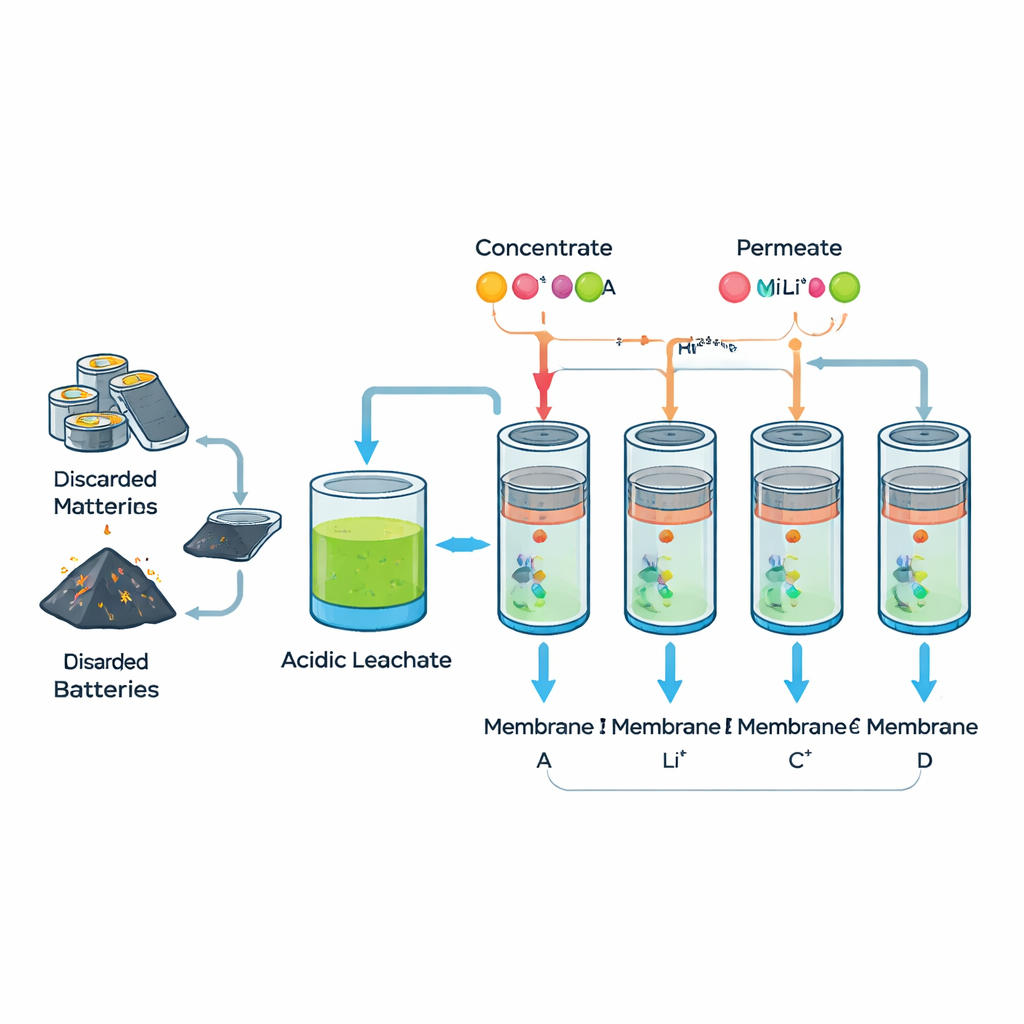
From Waste Batteries to Useful Liquid
When a lithium-ion battery reaches the end of its life, it does not go straight into a filter. First, its metal-rich parts are processed with acids in a step called leaching. This produces a strong liquid mixture that contains lithium along with several heavier metals such as nickel, cobalt, manganese, aluminum, and iron. In real recycling plants this liquid is messy and full of fine powders, so the researchers created a cleaner “synthetic leachate” with the same mix of dissolved metals. This allowed them to study how well different filters can separate lithium under controlled, realistic conditions.
Using Smart Filters Instead of Harsh Steps
The team tested four off-the-shelf nanofiltration membranes. These are thin, plastic-like sheets full of tiny water pathways that act a bit like sieves and a bit like electrically charged barriers. Water and small, singly charged particles such as lithium ions can slip through more easily, while larger or more highly charged metal ions are mostly held back. The researchers carefully measured each membrane’s thickness, roughness, and water-friendliness, then pushed the synthetic battery liquid across them in a lab setup similar to what could be used in industry. They also checked how the membranes changed after use, making sure they did not crack or fall apart.
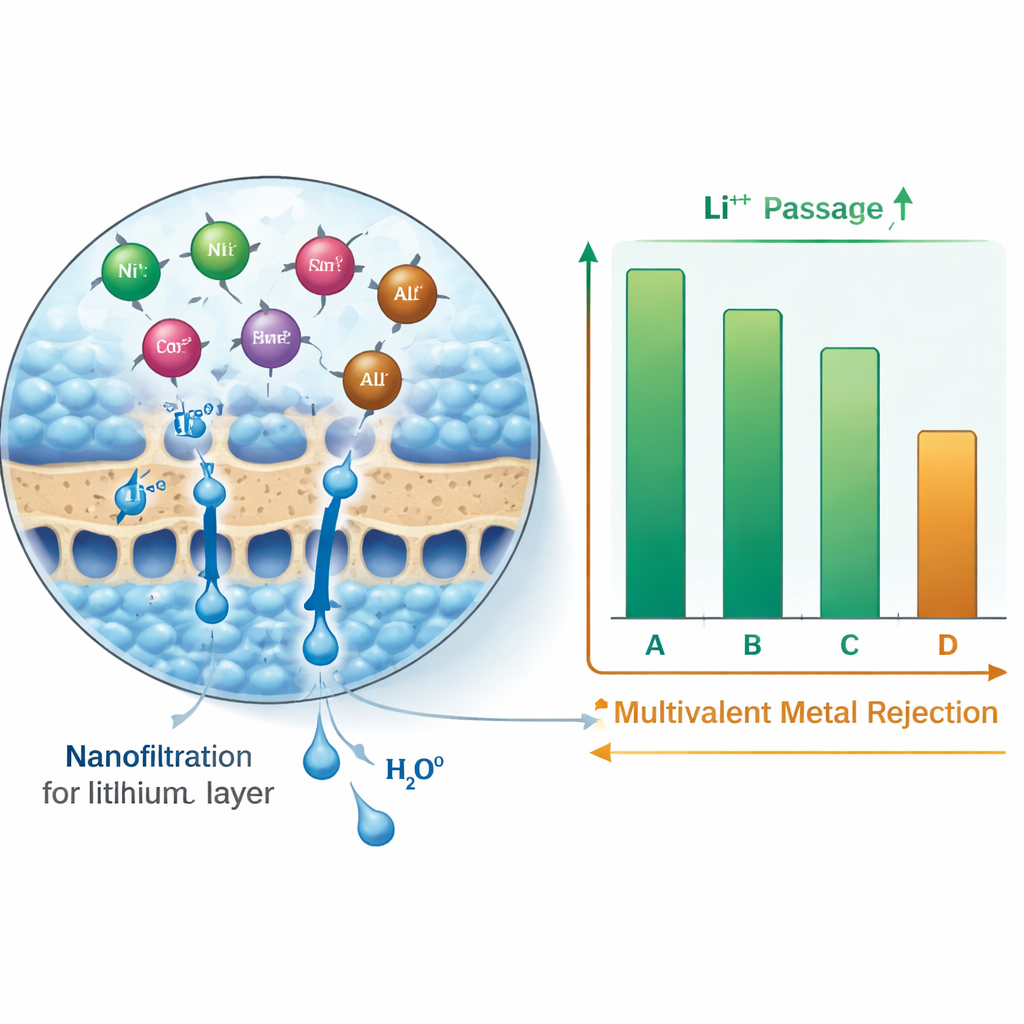
Letting Lithium Through, Holding Heavy Metals Back
All four membranes behaved in the same broad way: lithium, being small and carrying only a single positive charge, was weakly rejected and mostly passed through, while heavier metals with two or three positive charges were strongly blocked. Two of the “looser” membranes allowed the most lithium to pass, rejecting only about one fifth of it, yet still held back around 80–90% of the multivalent metals. The “tighter” membranes were more selective in the other direction: they rejected over 90% of nickel, cobalt, manganese, aluminum, and iron, but also stopped a larger share of lithium. When all metals were present together, the blocking of heavy metals became even stronger due to electrical crowding at the membrane surface, while lithium still flowed through in significant amounts.
Designing the Best Filter Mix
By comparing the four membranes side by side, the researchers built a simple rulebook for choosing filters in a recycling plant. If the main goal is to get as much lithium as possible into the clean liquid on the far side of the membrane, a more open membrane is best, because it offers low resistance to lithium while still capturing most heavier metals. If a process needs to scrub out heavy metals as thoroughly as possible, a tighter membrane is preferable, even though it sacrifices some lithium flow. The study also showed how features such as surface roughness, water contact angle (how easily water spreads over the surface), and chemical makeup all work together to control which ions pass and which stay behind.
What This Means for Everyday Life
For non-specialists, the take-home message is that simple, commercially available filters can already help turn worn-out batteries into a reliable secondary source of lithium, easing pressure on mines and sensitive salt flats. By choosing the right combination of membranes, recycling companies could both recover high-purity lithium for new batteries and keep toxic heavy metals out of the environment. In other words, the work points toward a future where the batteries in our devices form part of a circular loop—being reborn as new batteries rather than ending up as hazardous waste.
Citation: Alam, M., Bruggen, B.V.d., Ahsan Khan, M. et al. High purity lithium recovery from spent lithium-ion batteries using commercial nanofiltration membranes: a comparative performance assessment. Sci Rep 16, 6129 (2026). https://doi.org/10.1038/s41598-026-36924-1
Keywords: lithium recycling, spent batteries, nanofiltration, membrane separation, circular economy