Clear Sky Science · en
CKAAKN peptide-conjugated long-circulating nanoliposomes for the targeted delivery of oridonin to pancreatic cancers
Why this research matters
Pancreatic cancer is one of the deadliest cancers, in part because it is usually discovered late and often resists standard chemotherapy. This study explores a smarter way to deliver an herbal-derived anti-cancer compound, oridonin, directly to pancreatic tumors using tiny fat-based bubbles called nanoliposomes. By helping more drug reach the tumor while sparing healthy tissue, this work points toward gentler, more effective treatment options for a cancer that desperately needs them.
A deadly cancer that is hard to treat
Pancreatic cancer has earned the grim nickname “king of cancers” because survival rates remain in the single digits for most patients. Only a small fraction of people are eligible for surgery, and even then, many tumors come back. Chemotherapy drugs such as paclitaxel and cisplatin can slow the disease, but they cause strong side effects and tumors often become resistant. Researchers are therefore searching for drugs that are both more precise and less toxic, along with delivery systems that can concentrate medicine inside the tumor instead of flooding the whole body.
Turning an herbal molecule into a modern medicine
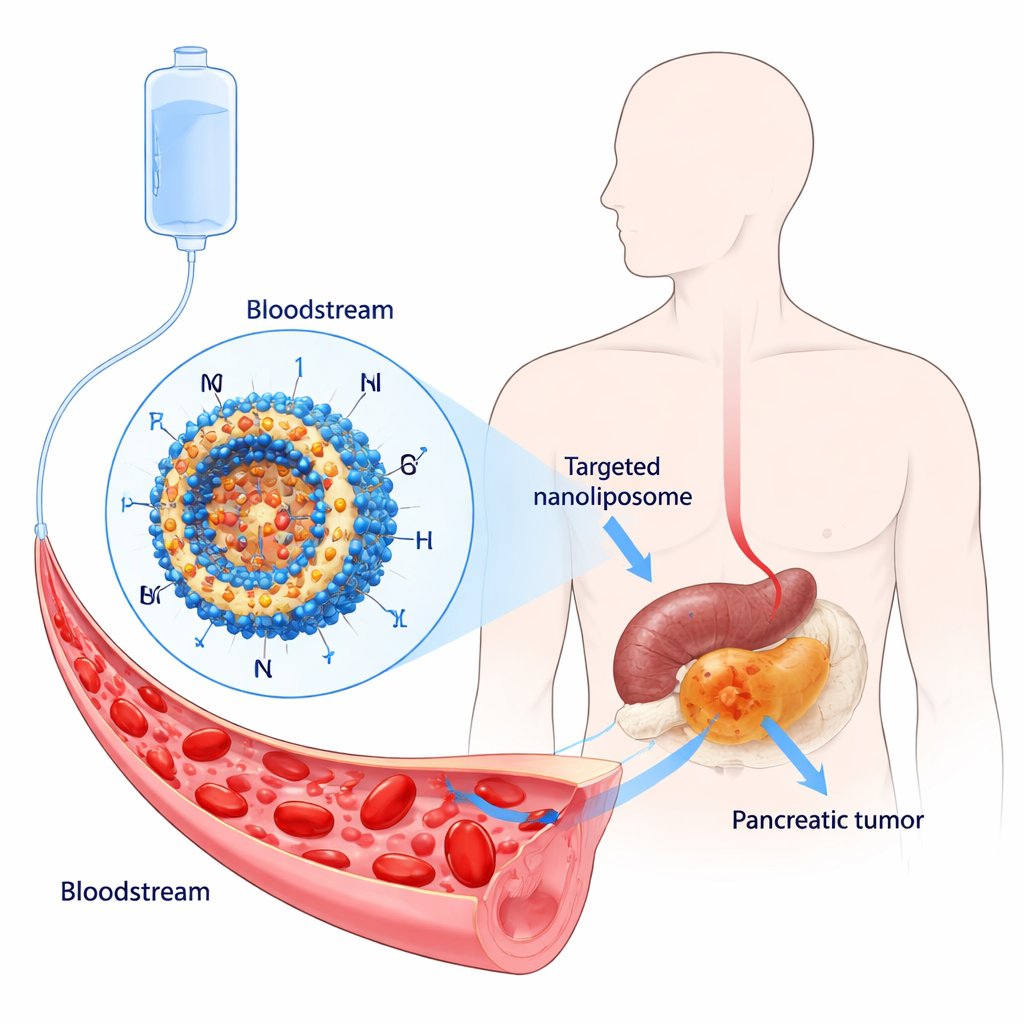
Oridonin is a natural compound extracted from the traditional Chinese medicinal plant Rabdosia rubescens. Laboratory studies have shown that it can kill or slow the growth of many types of cancer cells, including those from the pancreas, by interfering with cell division and triggering cell suicide. However, oridonin on its own is difficult to use as a medicine: it does not dissolve well in water, is poorly absorbed, and can be toxic at higher doses. To harness its promise, the authors set out to package oridonin inside nanoliposomes—tiny spheres made from lipids similar to those in cell membranes—designed to carry the drug safely through the bloodstream and release it slowly at the tumor site.
Designing a “stealth” homing package for the drug
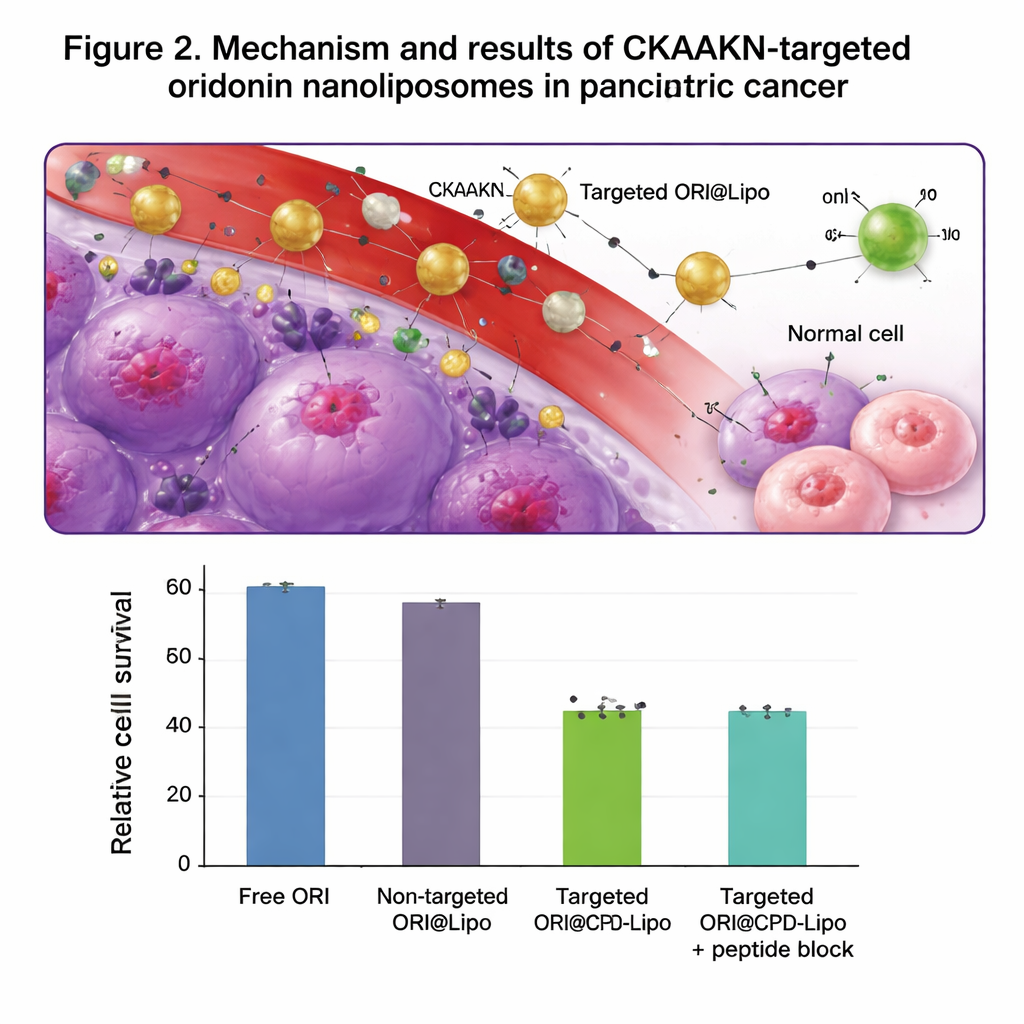
The team engineered a two-layered delivery system. First, they created long-circulating liposomes by coating their surface with polyethylene glycol (PEG), a flexible, water-loving polymer that helps the particles evade immune cells and stay in the blood longer. Then they added a short peptide, CKAAKN, that acts like a homing tag for pancreatic cancer. This peptide mimics part of the Wnt signaling pathway, which is abnormally active in many pancreatic tumors, allowing the modified liposomes (called ORI@CPD-Lipo) to latch onto cancer cells that display matching receptors. The final particles were about 100 nanometers in diameter, carried high amounts of oridonin, and released the drug slowly over many hours instead of all at once.
How the targeted particles behave in cells and animals
In cell culture tests, the CKAAKN-tagged liposomes were taken up far more efficiently by human pancreatic cancer cells than by normal pancreatic cells, especially during the first few hours of contact. When excess free CKAAKN peptide was added as a decoy, the cancer cells absorbed fewer particles, supporting the idea that a specific binding step is involved. Empty liposomes showed low toxicity and did not damage red blood cells, suggesting good safety. When loaded with oridonin, the targeted liposomes killed cancer cells more effectively than either free oridonin or non-targeted liposomes, and they more strongly slowed cancer cell migration across a scratch “wound” in a dish. In mice bearing pancreatic tumors, fluorescently labeled targeted liposomes built up in the tumor for up to 48 hours and accumulated there more than non-targeted liposomes, while still largely clearing through the liver and spleen as expected.
What this could mean for future treatments
Taken together, the results show that combining a traditional herbal compound with a modern, precision delivery vehicle can significantly boost its anti-tumor power in laboratory models. The CKAAKN-decorated, long-circulating nanoliposomes funnel more oridonin into pancreatic cancer cells while limiting exposure to normal tissue, enhancing cancer cell death and reducing movement that could lead to spread. Although these findings are still preclinical and the authors note that full animal treatment studies and clinical trials are needed, the work lays a solid foundation for targeted nanomedicine approaches to pancreatic cancer and suggests a general strategy for turning other promising natural molecules into practical, tumor-seeking drugs.
Citation: Zhang, F., Luo, K., Xuan, S. et al. CKAAKN peptide-conjugated long-circulating nanoliposomes for the targeted delivery of oridonin to pancreatic cancers. Sci Rep 16, 6065 (2026). https://doi.org/10.1038/s41598-026-36920-5
Keywords: pancreatic cancer, nanoliposomes, targeted drug delivery, oridonin, tumor-homing peptide