Clear Sky Science · en
Metadynamics reveals luteolin-mediated conformational stabilization against avapritinib-resistant PDGFRα D842V/G680R GIST
Why this matters for cancer treatment
Targeted cancer drugs can work remarkably well—until tumors evolve ways to escape them. This article explores how a common plant compound, luteolin, might help restore the power of an important drug used against certain gastrointestinal stromal tumors (GIST), a type of cancer in the digestive tract. By using advanced computer simulations, the researchers show how luteolin could steady a misshapen protein in cancer cells so that the existing drug can latch on again, hinting at a new way to fight drug resistance without reinventing treatment from scratch. 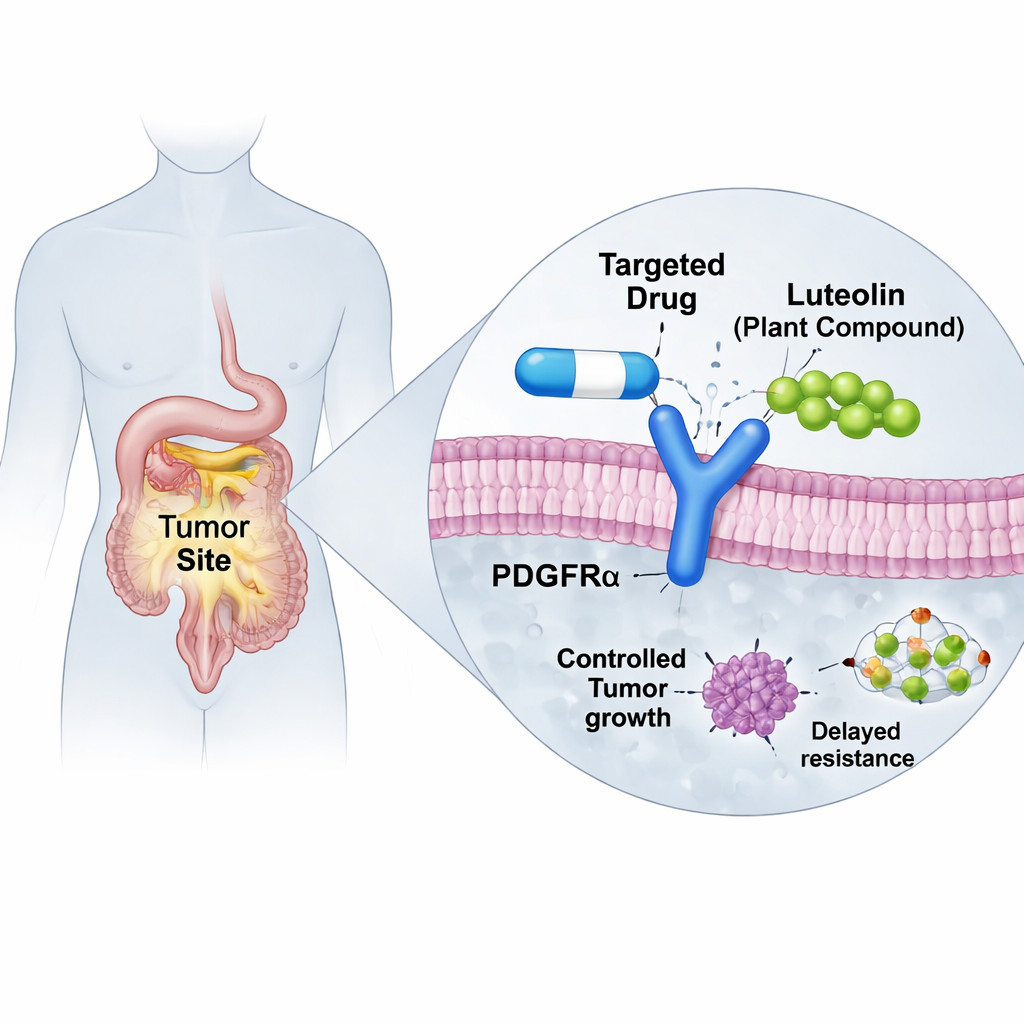
The problem of tumors that learn to dodge drugs
GISTs often grow because of faulty "on switches" in cell-surface proteins known as KIT or PDGFRα, which drive constant growth signals. Modern pills called tyrosine kinase inhibitors were designed to shut these switches off and have transformed outcomes for many patients. But a frequent mutation in PDGFRα, called D842V, makes the older drug imatinib largely useless. A newer drug, avapritinib, was built specifically to tame this stubborn mutant and has shown striking success in clinical trials. Unfortunately, tumors can still adapt. Some patients develop additional changes in PDGFRα, such as a secondary G680R mutation, that physically block avapritinib from fitting comfortably into its binding pocket, causing the drug to fall out and resistance to rise.
A plant molecule with an intriguing shape
Natural products are a rich source of drug ideas, and flavonoids—colorful compounds found in many fruits, vegetables, and medicinal plants—are known to interact with growth-control proteins inside cells. Luteolin, one such flavonoid present in a Mediterranean shrub called Retama monosperma, has already been studied for its anticancer and anti-inflammatory properties and appears safe at levels achievable through oral supplements. Based on earlier screening work, the authors suspected luteolin might stick to PDGFRα in a spot near, but not overlapping, the usual drug-binding site. If true, this could let luteolin act like a subtle brace, nudging the protein into a shape that once again welcomes avapritinib.
Simulating a molecular partnership
To test this idea in silico, the team built detailed three-dimensional computer models of the resistant PDGFRα protein carrying both the D842V and G680R mutations. They simulated how avapritinib behaved on its own and how it behaved when luteolin was present nearby, tracking atomic motions over hundreds of nanoseconds—billions of a second in real time. When only avapritinib was bound, the drug gradually drifted out of its pocket as the bulky G680R change pushed it away, ending up completely detached from the protein. In contrast, when luteolin was also bound at a separate “allosteric” site next to a key helix, avapritinib stayed largely in place. The protein as a whole flexed less, key structural segments held their shape, and important contacts between the drug and the protein persisted far longer. 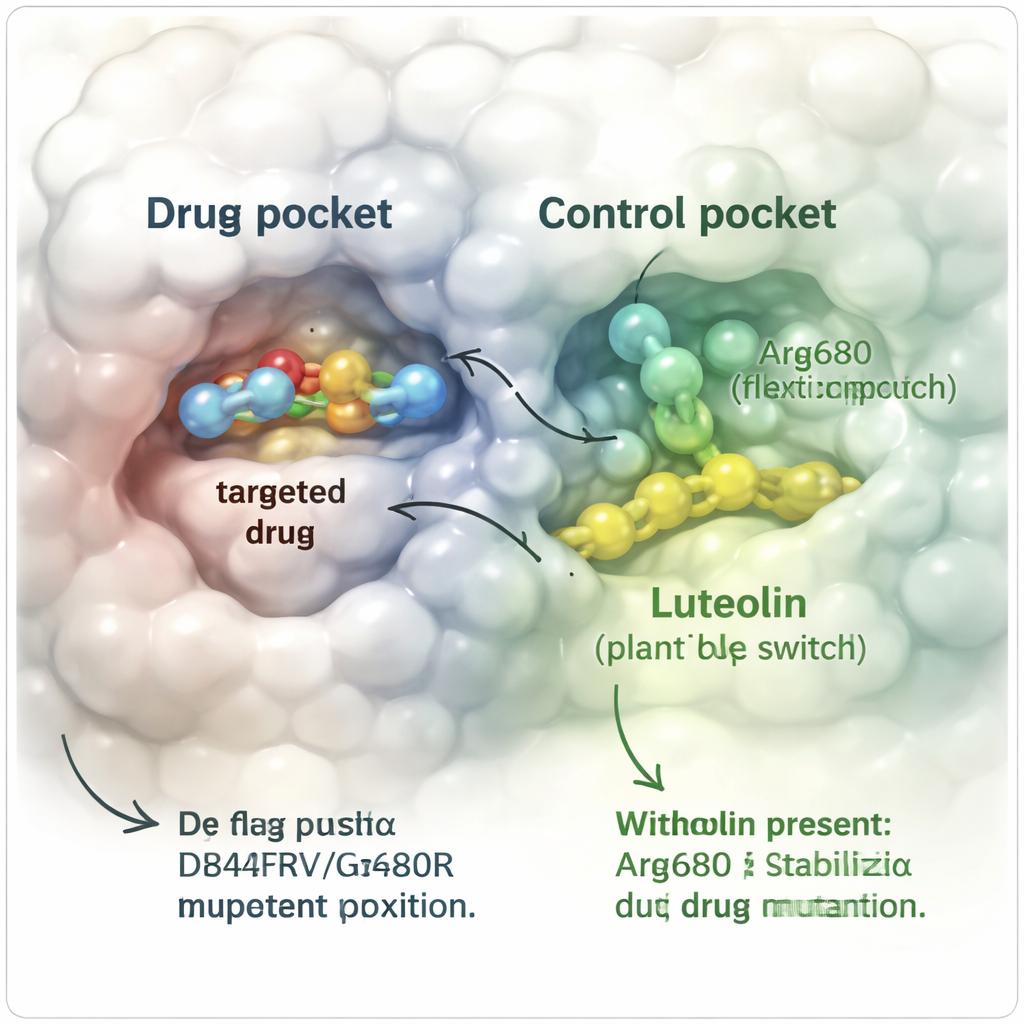
Pinning down a wayward molecular switch
Standard simulations still miss rare but important shape changes, so the researchers turned to an enhanced approach called metadynamics to more fully explore how the protein might twist and bend. They focused on the behavior of the altered amino acid Arg680, which acts like a small flexible arm near the drug pocket. Without luteolin, this arm sampled many positions and the drug readily slipped away, with relatively low energy barriers to escape. With luteolin in place, Arg680 was held in a specific orientation that no longer clashed with the drug. The energy landscape shifted: the system now preferred conformations in which avapritinib stayed bound, and the energetic “hill” the drug would have to climb to escape grew several kilocalories higher, making dissociation much less likely.
What this could mean for patients
In plain terms, the study suggests that luteolin may act like a molecular splint, steadying a warped cancer protein so that an existing targeted pill can grip it again. Instead of designing an entirely new drug for every resistance mutation, pairing the current medicine with a carefully chosen helper compound might restore its effectiveness. The work is based on powerful but purely computational methods, so lab and animal studies are still needed to confirm whether luteolin, or a related molecule engineered for better drug-like properties, truly boosts avapritinib in living systems. Still, the concept of using small allosteric helpers to re-stabilize resistant cancer targets could open a fresh path for extending the life of today’s precision therapies.
Citation: El Khattabi, K., Akachar, J., Lemriss, S. et al. Metadynamics reveals luteolin-mediated conformational stabilization against avapritinib-resistant PDGFRα D842V/G680R GIST. Sci Rep 16, 6534 (2026). https://doi.org/10.1038/s41598-026-36898-0
Keywords: drug resistance, gastrointestinal stromal tumor, tyrosine kinase inhibitors, luteolin, allosteric modulation