Clear Sky Science · en
Experimental and theoretical evaluation of geometry-dependent doxorubicin loading onto cerium oxide nanoparticles via van der Waals interaction modeling
Why the shape of tiny drug carriers matters
Cancer drugs like doxorubicin are powerful but can damage healthy tissue. One strategy to make them safer is to hitch them onto nanoparticles that ferry the medicine straight to tumors. This study asks a surprisingly simple question with big implications: does the shape of the nanoparticle—whether it looks like a ball, a rod, or a sheet—change how much drug it can carry and how well it works?
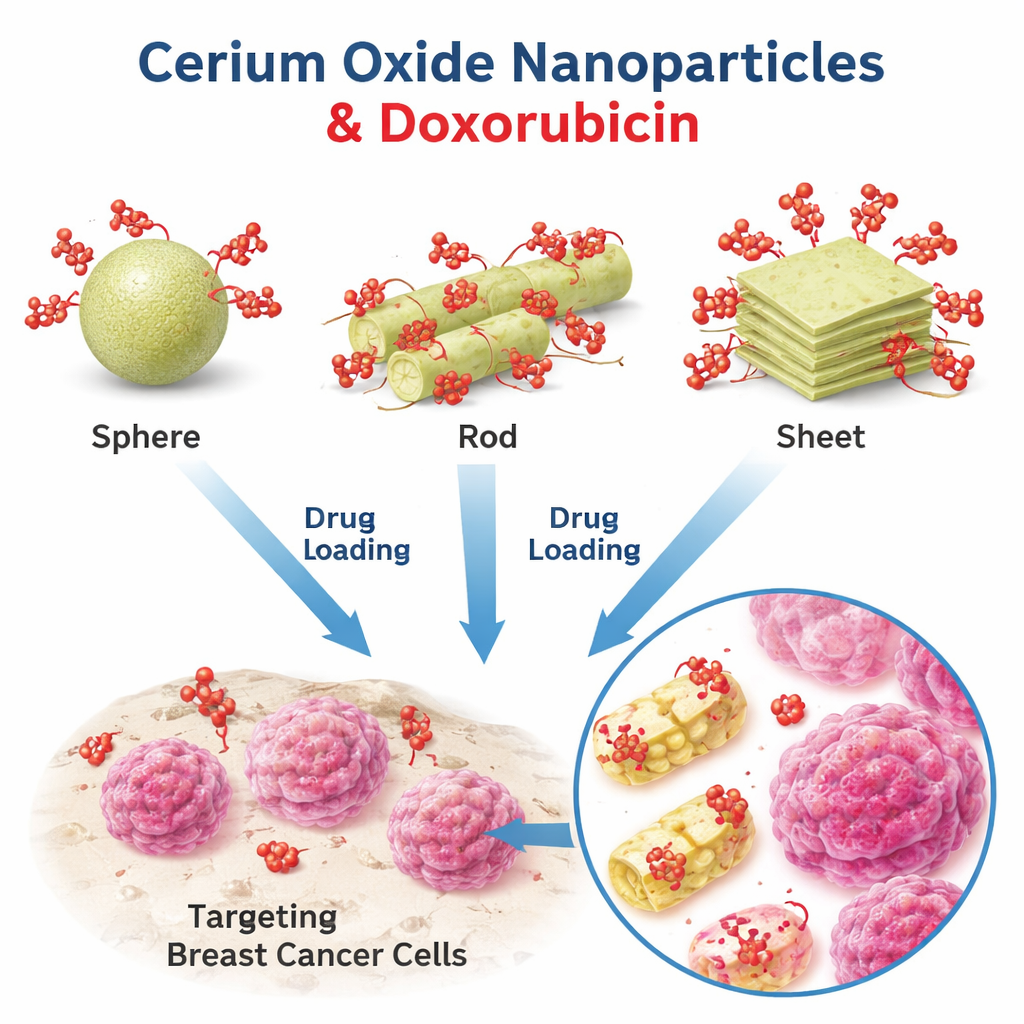
Three tiny shapes, one cancer drug
The researchers focused on nanoparticles made of cerium oxide, a material already known for its antioxidant, antibacterial, and wound-healing properties, and explored its potential as a carrier for doxorubicin, a widely used chemotherapy drug. They prepared three distinct shapes of cerium oxide: nearly perfect spheres, rod-like cylinders, and thin sheet-like flakes. Using electron microscopes and light-scattering measurements, they confirmed the particles’ sizes and shapes, with spheres forming compact beads, cylinders appearing as slender sticks, and sheets as broad, flat layers. This controlled set of shapes allowed them to ask how geometry alone changes drug behavior, while keeping the material itself the same.
Measuring how much drug each shape can hold
To see how effectively each shape could carry doxorubicin, the team mixed the nanoparticles with a solution of the drug and then measured how much drug remained in the liquid afterward. Less leftover drug meant more had been loaded onto the particles. Using precise light-based methods (UV–visible and fluorescence spectroscopy), they found that spherical nanoparticles captured the most doxorubicin, with about 86% of the drug ending up on or in the spheres. Cylinders followed at around 79%, and sheets trailed at roughly 67%. When these drug-loaded particles were tested against an aggressive breast cancer cell line, the sphere-based formulation was again the most effective at killing cancer cells, followed by the cylinders and then the sheets. Interestingly, the spheres also released the drug most slowly over time, hinting that a high load combined with slow release can boost the drug’s impact inside cells.
Putting math to work on the nano world
Alongside the lab work, the researchers built an analytical model—a kind of simplified mathematical microscope—to calculate how strongly a single, roughly spherical doxorubicin molecule should stick to each nanoparticle shape. They focused on van der Waals forces, the weak but omnipresent attractions that help molecules cling to one another. By treating the drug as a small sphere near a spherical, cylindrical, or sheet-like cerium oxide surface, they derived exact formulas for the interaction energy as the drug approached or moved away. These calculations allowed them to predict which shape should give the most stable binding, both when the drug is buried inside the particle and when it sits on the surface, without running large computer simulations.
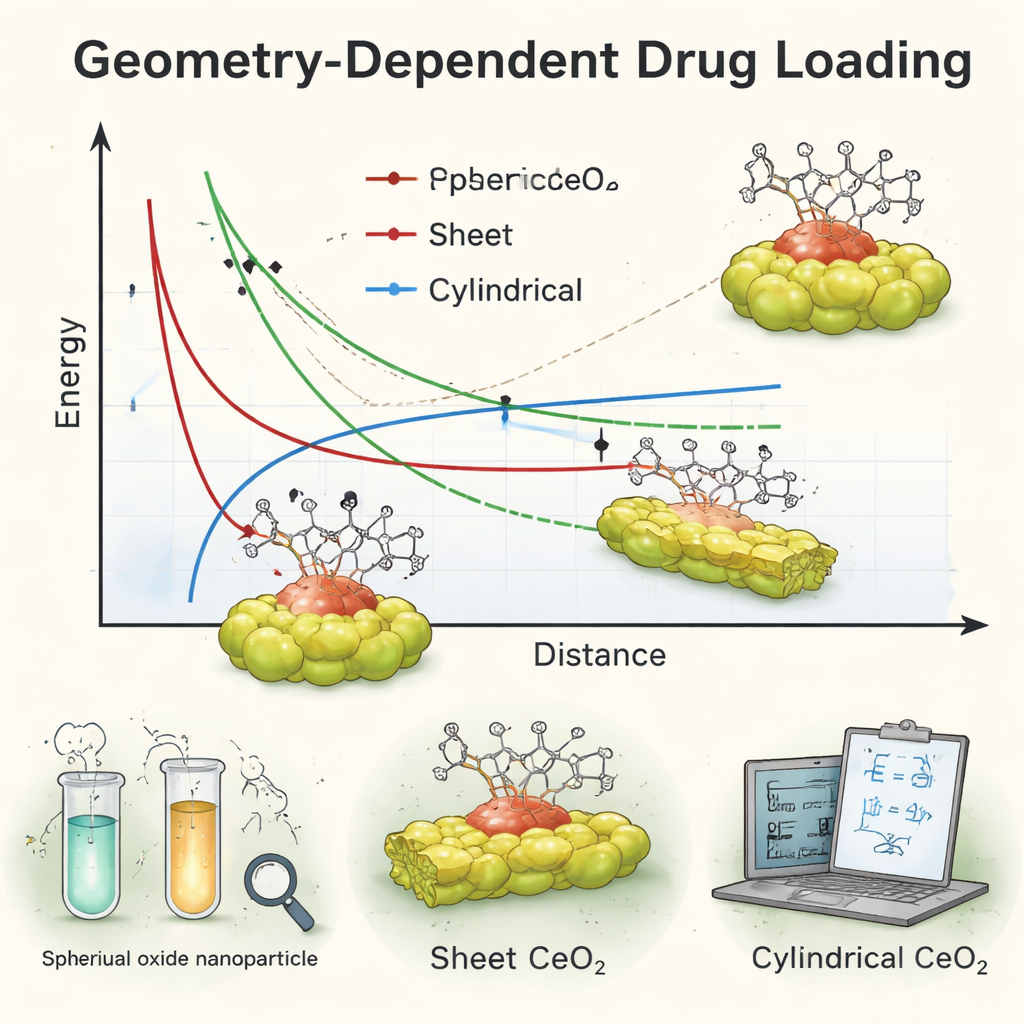
Where theory agrees—and where it breaks down
When the team compared their equations with the experimental data, they found a striking partial match. The math suggested that spherical and sheet-like nanoparticles should hold doxorubicin with very similar binding strengths, which fit well with the generally high loading seen for those two shapes. Moreover, when the drug was assumed to be trapped inside the particles, spheres came out as slightly more stable than the other shapes, echoing the strong performance of the spherical carriers in the lab. But there was a puzzle: the model predicted relatively weaker binding for cylinders, while experiments showed that rod-shaped particles still loaded the drug quite efficiently—almost as well as the spheres. This mismatch, especially for the cylindrical shape, revealed that a simple model that treats the surroundings as empty space and averages over surface details cannot fully capture the real behavior of drug–nanoparticle systems bathed in liquid and interacting with cells.
What this means for future cancer treatments
For non-specialists, the message is twofold. First, the shape of a nanoparticle is not a cosmetic detail; it directly affects how much cancer drug it can carry, how quickly it releases that drug, and how strongly it can attack tumor cells. Spherical cerium oxide particles in this study emerged as particularly promising carriers for doxorubicin, combining high loading, strong cancer cell killing, and slow drug leakage. Second, the study shows the limits of even elegant mathematical models when they oversimplify the messy reality of biology. To design truly reliable nanomedicines, future work will need to blend detailed experiments with more sophisticated theories that include the watery environment, complex particle surfaces, and particle clustering. Together, these advances could lead to smarter nanoparticle designs that deliver powerful drugs more safely and effectively.
Citation: Sripaturad, P., Keo, S., Wongpan, A. et al. Experimental and theoretical evaluation of geometry-dependent doxorubicin loading onto cerium oxide nanoparticles via van der Waals interaction modeling. Sci Rep 16, 6169 (2026). https://doi.org/10.1038/s41598-026-36893-5
Keywords: nanomedicine, cerium oxide nanoparticles, doxorubicin delivery, nanoparticle geometry, breast cancer therapy