Clear Sky Science · en
Synthesis and anticancer evaluation of novel thioimidazole derivatives bearing a trimethoxyphenyl moiety
New chemical tools in the fight against cancer
Chemotherapy drugs often harm healthy cells along with tumors, leading to harsh side effects. This study explores a new family of laboratory-made molecules that aim to hit cancer cells much harder than normal tissue. By cleverly joining together two chemical building blocks that are already known to work well in medicines, the researchers created compounds that show strong, targeted killing of cancer cells in test-tube and 3D tumor models, while leaving healthy cells largely unharmed.
Building smarter cancer-killing molecules
The team focused on two “privileged” chemical structures that appear again and again in successful drugs. One is imidazole, a small ring that fits snugly into many biological targets. The other is a trimethoxyphenyl (TMP) group, a pattern of three oxygen-containing side chains on an aromatic ring that is central to several anticancer drugs that disrupt cell division. By adding a sulfur atom into the imidazole core (creating a thioimidazole) and linking it to a TMP group, the chemists hoped to fine-tune how the molecules move through the body and how tightly they bind to cancer-related proteins. Using a stepwise synthetic route, they built a library of related compounds, each subtly modified to probe what features matter most for anticancer activity. 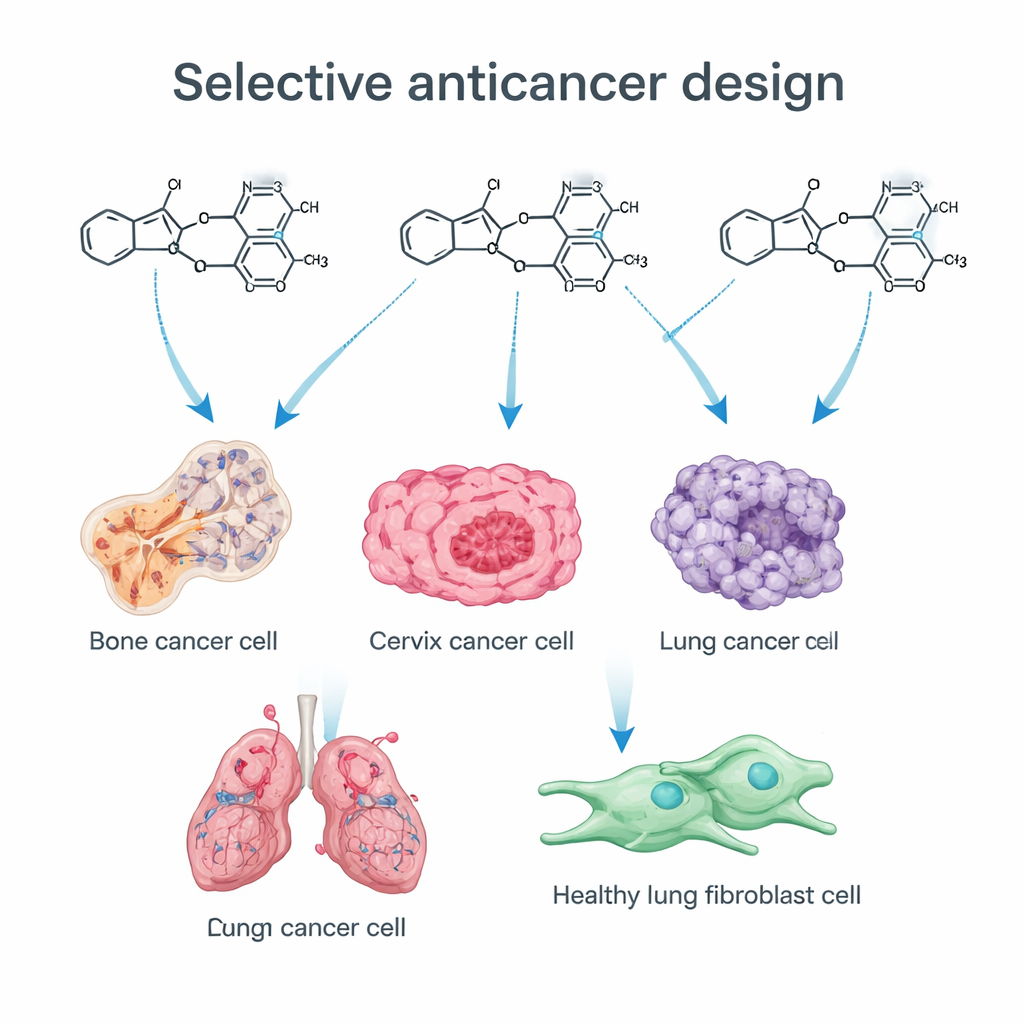
Testing cancer cells while sparing healthy tissue
The new molecules were tested on four human cancer cell lines—osteosarcoma (bone), cervical, colon, and lung cancer—as well as on non-cancerous lung fibroblasts. Many of the compounds slowed or stopped cancer cell growth, but one in particular, named 13b, stood out. It killed bone and cervical cancer cells at very low concentrations, yet showed no detectable toxicity toward normal fibroblasts even at doses more than 60 times higher. Several other compounds were moderately potent but less selective, underscoring how small structural changes—such as adding a chlorine atom or a nitro group in specific positions—can swing the balance between broad toxicity and cancer-focused action.
How the lead compound makes cancer cells self-destruct
To understand what 13b actually does inside cells, the researchers looked for hallmarks of programmed cell death, or apoptosis. Cancer cells treated with 13b became positive for Annexin V and activated the enzymes caspase-3 and -7, classic markers that a cell has been pushed into an orderly suicide program rather than simply bursting. Microscopy revealed bright spots of γH2AX, a signal of broken DNA strands, in the nuclei of treated cells. Over time, the internal scaffolding of the cells began to reorganize and break down, especially in cervical cancer cells, which rounded up and detached—visual cues that apoptosis was well underway. Together, these findings show that 13b damages cancer cell DNA and then drives the cells down a controlled death pathway.
From flat cell layers to 3D mini-tumors
Most lab tests grow cancer cells in thin layers, which do not fully mimic the dense, layered structure of real tumors. To address this, the team grew the cancer cells into three-dimensional spheroids—tiny, ball-shaped clusters that better resemble a tumor’s architecture and drug penetration challenges. When these spheroids were treated with 13b, they shrank in a dose-dependent manner. Bone cancer spheroids were especially sensitive, becoming small, loose, and fragmented at higher drug levels. Fluorescent staining showed waves of apoptosis moving from the outside of the spheroid inward as the concentration increased, proving that 13b can penetrate and kill cells throughout these compact structures. 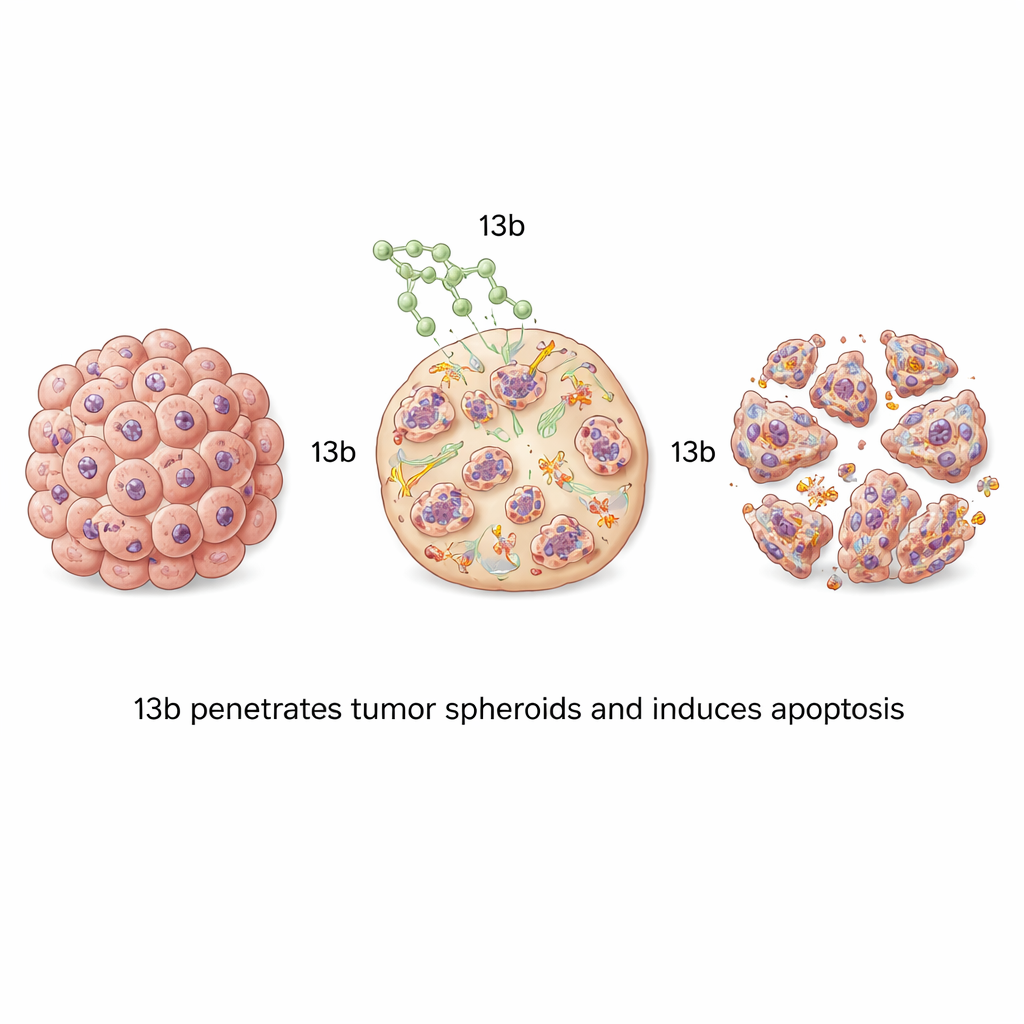
Why this work matters for future cancer treatments
For a non-specialist, the key message is that the researchers have created a new class of small molecules that combine the best features of two proven drug components into one hybrid design. Their leading compound, 13b, is able to recognize and destroy cancer cells efficiently while largely sparing healthy cells in lab tests, and it remains effective even in more realistic 3D tumor models. Although much work remains—such as pinpointing its exact molecular targets, improving its solubility, and testing it in animals—this study shows that blending thioimidazole and TMP frameworks is a promising strategy for crafting more selective and powerful anticancer agents.
Citation: Maciejewska, N., Grybaitė, B., Anusevičius, K. et al. Synthesis and anticancer evaluation of novel thioimidazole derivatives bearing a trimethoxyphenyl moiety. Sci Rep 16, 6271 (2026). https://doi.org/10.1038/s41598-026-36890-8
Keywords: anticancer agents, imidazole derivatives, trimethoxyphenyl, apoptosis, 3D tumor spheroids