Clear Sky Science · en
A novel composite of chitosan and Bacillus subtilis exopolysaccharide for the removal of methylene blue from aqueous solutions
Why cleaning colored water matters
From the jeans we wear to the paper we print on, modern life relies on synthetic dyes. But when leftover dyes are washed down factory drains, they can turn rivers bright colors, block sunlight, harm aquatic life, and even pose health risks to people. Treating this “colored water” is expensive, especially in regions already struggling with water scarcity. This study explores a low-cost, biodegradable material made from natural polymers—one from shellfish waste and one from helpful bacteria—to pull a common blue dye out of water quickly and efficiently.
A natural team for dirty water
The researchers focused on removing methylene blue, a widely used blue dye that is tough to break down once it reaches the environment. They built a new material by combining chitosan—a sugar-based compound obtained from crustacean shells—with exopolysaccharides, long-chain sugars produced by a bacterium called Bacillus subtilis. Both ingredients are biodegradable and already known to stick to pollutants. The idea was that blending them into a single “composite” would create more and better binding sites for the dye than either material alone, while also reusing bacterial by-products that are usually discarded.
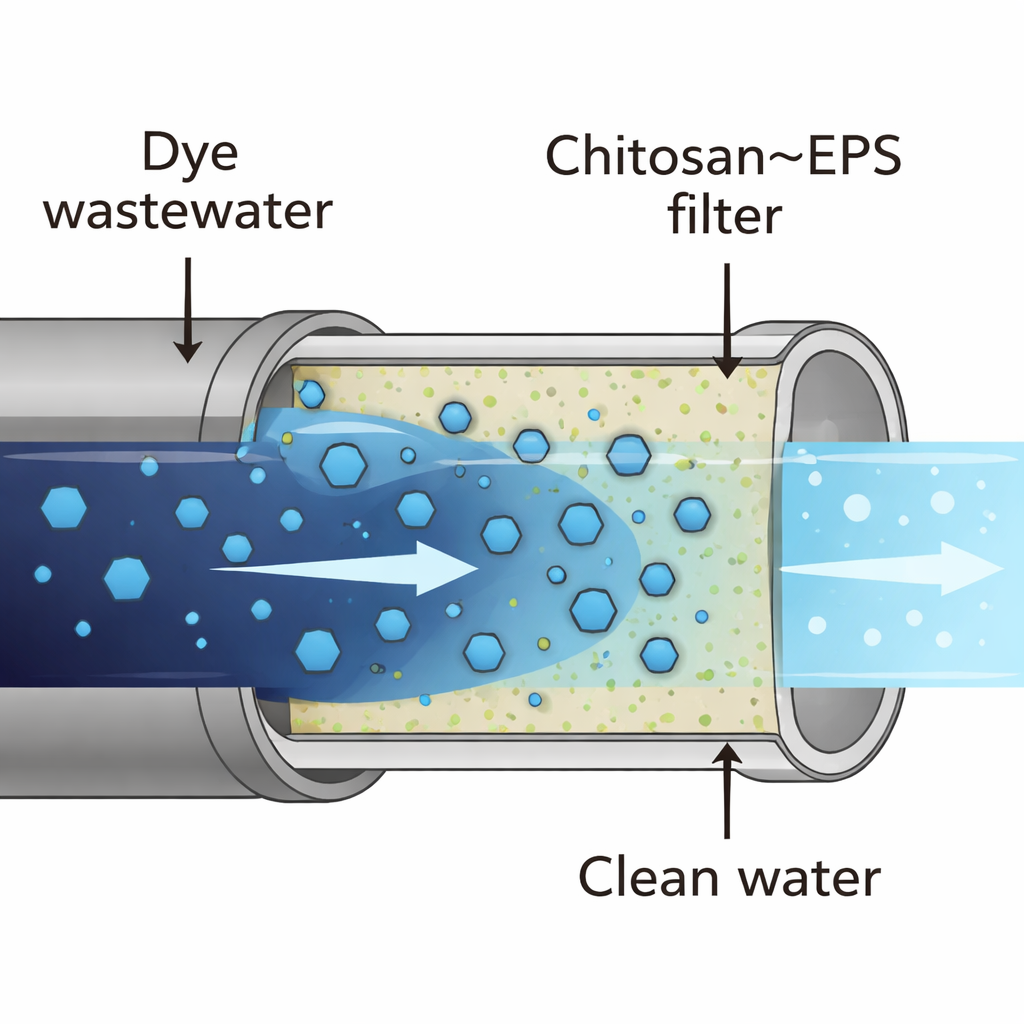
How the new filter material behaves
To understand how well this natural composite works, the team first examined its chemistry and structure using infrared spectroscopy and electron microscopy. These tools confirmed that the material had many active chemical groups—such as hydroxyl, amino, carboxyl, and phosphate groups—that can latch onto dye molecules. Under the microscope, plain chitosan appeared smooth and relatively dense, with few pores. In contrast, the chitosan–bacterial sugar composite looked rougher and more porous, with a sponge-like texture. This more open, irregular surface offers extra room for the blue dye molecules to slip in and attach.
Finding the best conditions for cleanup
The scientists then tested how different water conditions affected dye removal. They varied the acidity (pH), the contact time, and the starting dye concentration. The composite removed dye best at a mildly acidic to near-neutral pH of about 6, while plain chitosan worked best at pH 7. As pH increased from strongly acidic toward neutral, the material’s surface became more negatively charged, which strongly attracted the positively charged methylene blue molecules. Both materials removed most of the dye within roughly 30 minutes, but the composite consistently did better, clearing about 72 percent of the color compared with about 61 percent for chitosan alone. When the initial dye level was very high, the removal efficiency dropped, mainly because the limited number of binding sites became saturated.
What happens at the molecular scale
To dig deeper into how the dye sticks, the team analyzed how much dye the materials could hold and how fast they worked. Their measurements fit a model in which the dye forms a single, orderly layer on the surface, a sign of well-defined attachment sites. The composite stored slightly more dye per gram than chitosan alone and clung to it more strongly. Timing experiments showed that the process followed a “second-order” kinetic pattern, which in simple terms means the rate is controlled by how quickly dye molecules can form bonds with specific sites. Here, the composite was strikingly faster: its rate constant was about an order of magnitude higher than that of plain chitosan. Additional infrared measurements before and after dye removal revealed small but telling shifts in key chemical signals, showing that groups containing oxygen, nitrogen, and phosphorus were directly involved. Together, the evidence points to a combination of electrostatic attraction between opposite charges, hydrogen bonds, and stacking interactions between ring-shaped dye molecules and the sugar backbone of the composite.
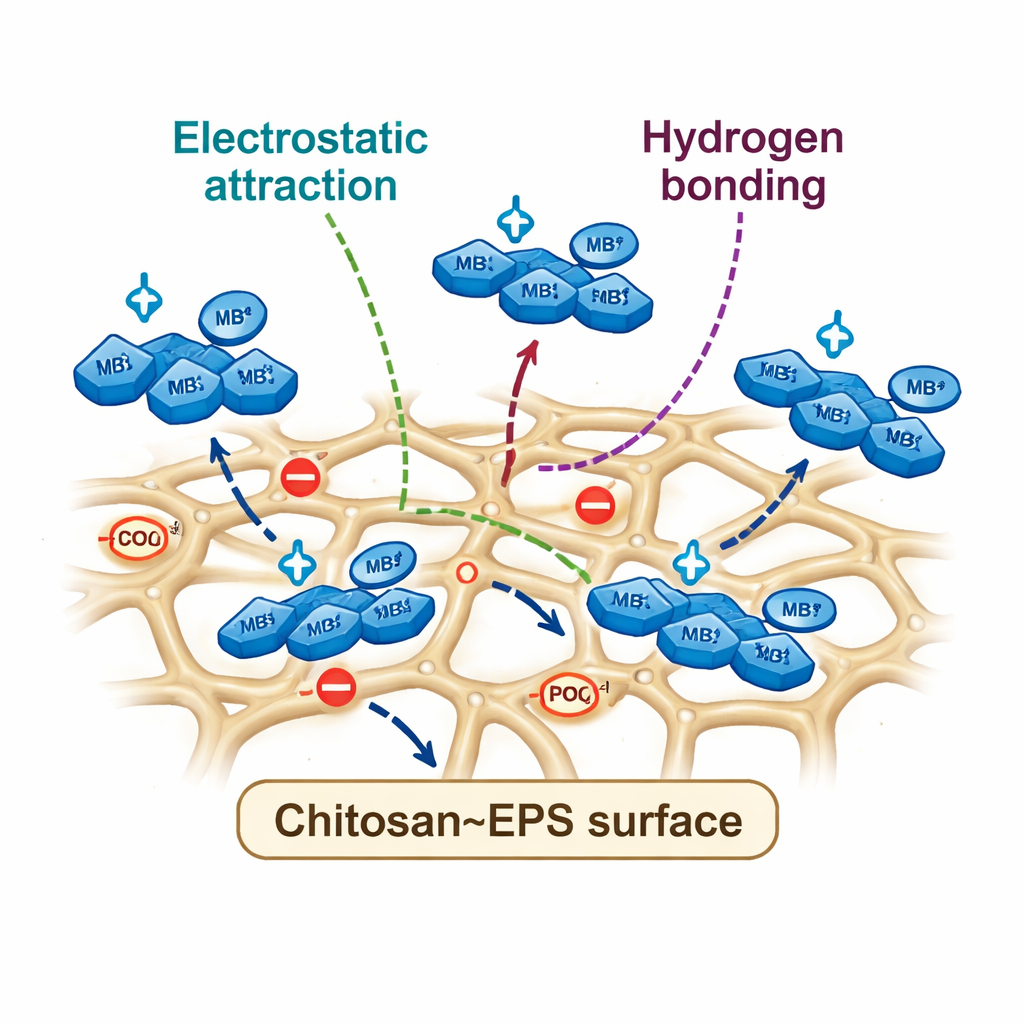
A greener path for treating colored wastewater
Overall, the study shows that blending chitosan with bacterial exopolysaccharides creates a fully biodegradable material that removes methylene blue from water more effectively and much faster than chitosan alone. Although some high-tech synthetic materials can hold even more dye, they often require harsh chemicals to make and take longer to work. In contrast, this natural composite is made from renewable ingredients, including a bacterial by-product that would otherwise go to waste, and it functions well under conditions similar to real industrial wastewater. That combination of speed, performance, and sustainability suggests this material could be developed into practical filters for factories that use bright dyes—helping keep our rivers clearer without adding new pollutants in the process.
Citation: Abd-Alla, M.H., Hassan, E.A., Mohammed, E.A. et al. A novel composite of chitosan and Bacillus subtilis exopolysaccharide for the removal of methylene blue from aqueous solutions. Sci Rep 16, 6349 (2026). https://doi.org/10.1038/s41598-026-36875-7
Keywords: wastewater treatment, biodegradable adsorbent, methylene blue removal, chitosan composite, bacterial exopolysaccharide