Clear Sky Science · en
Benefit and risk associated with interleukin-6 receptor inhibitor administration during severe COVID-19: a retrospective multicentric study
Why this matters for patients and families
During the COVID-19 pandemic, doctors urgently needed treatments to help the sickest patients survive, especially those struggling to breathe. One promising option was a class of drugs that block interleukin‑6 (IL‑6), a powerful messenger of inflammation in the body. These medicines, such as tocilizumab and sarilumab, can calm an overactive immune response—but they may also weaken the body’s defenses against germs. This study asks a simple but crucial question: when used in real hospitals during severe COVID-19, do these drugs help more than they harm?
How the study was set up
Researchers in Paris looked back at the medical records of 2,587 adults who were hospitalized with severe or critical COVID‑19 between March 2020 and December 2021. Some patients stayed only in regular hospital wards, while others needed intensive care units (ICUs) because of organ failure or the need for mechanical ventilation. Within these groups, a minority—224 people, or about 9%—received at least one dose of an IL‑6 receptor inhibitor (IL‑6Ri), mainly tocilizumab and, less often, sarilumab. The team compared patients who did and did not receive these drugs, focusing on serious side effects such as hospital‑acquired infections, bleeding in the digestive tract, and blood cell problems like low platelets or white cells.
What happened to patients in regular wards
Among the 1,603 patients who stayed in hospital wards, those treated with IL‑6Ri tended to be sicker: they more often needed high‑flow oxygen, stayed longer in the hospital, and more frequently received other treatments such as corticosteroids and blood thinners. In this group, the risk of dying in the hospital was higher for patients who got IL‑6Ri (about 19%) compared with those who did not (about 10%). At the same time, the length of stay was longer—10 days versus 7 days on average. While these differences could partly reflect that doctors reserved IL‑6Ri for the most fragile patients, they raised concerns that the drugs might not be clearly improving outcomes outside of the ICU.
What happened in intensive care
In the 984 patients who needed ICU care, treatment patterns were somewhat different. Here, 133 patients received an IL‑6Ri. Both treated and untreated ICU patients required high rates of advanced support such as mechanical ventilation, prone positioning, and sometimes extracorporeal membrane oxygenation (ECMO), a form of heart‑lung bypass. In this sicker group, hospital death rates were similar whether or not patients received IL‑6Ri—about one in four died in each group. Lengths of stay in the ICU and overall hospital time were also roughly the same. In other words, in this real‑world ICU population, blocking IL‑6 did not clearly improve survival, but it also did not worsen it.
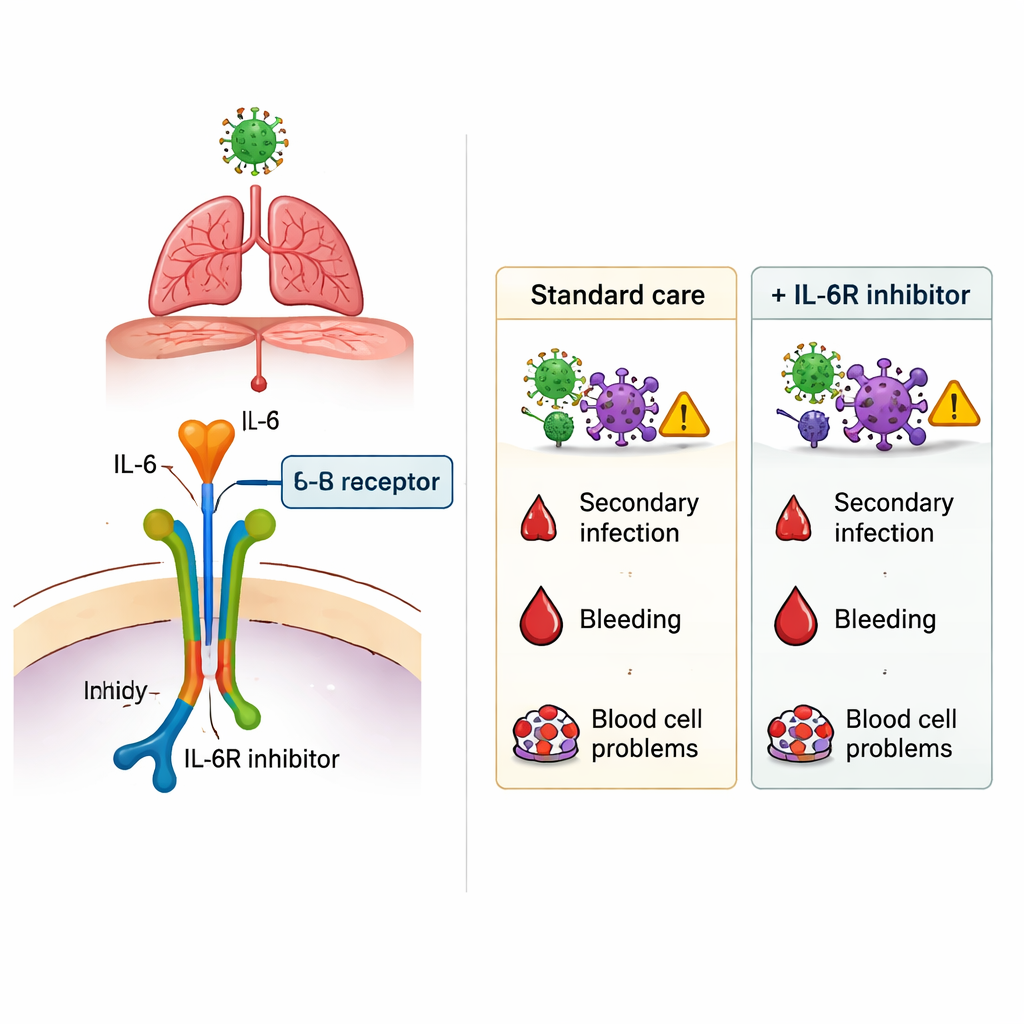
The key risk: more infections
The most striking finding emerged when the researchers looked at new infections that developed during the hospital stay, such as ventilator‑associated pneumonia, urinary tract infections, bloodstream infections, and other hospital‑acquired illnesses. Across the entire population, patients who received an IL‑6Ri had more secondary infections than those who did not. After carefully adjusting for age, weight, existing illnesses, and illness severity, the use of IL‑6Ri still remained linked to a higher infection risk—roughly a 40% to 100% relative increase, depending on the analysis method. This added risk was not limited to any single type of germ or site of infection. By contrast, serious bleeding in the stomach or intestines and blood cell complications occurred at similar rates with or without IL‑6Ri treatment, suggesting that these feared side effects were not noticeably amplified by the drugs in this setting.
What this means going forward
For lay readers, the bottom line is that drugs like tocilizumab and sarilumab can modestly change the course of severe COVID‑19, but they are not without cost. In this large French study, their use was tied to a higher chance of hospital‑acquired infections, while survival in the sickest ICU patients did not clearly improve and ward patients who received them died more often. The study does not prove that IL‑6 blockers directly caused these outcomes—doctors may have chosen them for those already at greatest risk—but it reinforces the need for careful case‑by‑case decisions. When considering IL‑6 blocking drugs, clinicians must weigh the potential benefit of slowing dangerous inflammation against the very real possibility of inviting new infections, especially in vulnerable patients.
Citation: Lefèvre, C., Funck-Brentano, T., Cachanado, M. et al. Benefit and risk associated with interleukin-6 receptor inhibitor administration during severe COVID-19: a retrospective multicentric study. Sci Rep 16, 5978 (2026). https://doi.org/10.1038/s41598-026-36864-w
Keywords: COVID-19, interleukin-6 inhibitor, tocilizumab, secondary infections, intensive care