Clear Sky Science · en
4-Nonylphenol regulates cell proliferation and apoptosis in cervical carcinoma through the MEG3/PI3K/AKT signaling pathway
Everyday Chemicals and Women’s Cancer Risk
We are surrounded by chemicals from plastics, detergents, and pesticides that can act like hormones in the body. This study explores whether one such compound, 4‑nonylphenol (4‑NP), might help drive cervical cancer, and how a natural protective molecule inside our cells, called MEG3, may fight back. Understanding this relationship could point to new ways to reduce risk from environmental pollution and to design smarter treatments for cervical cancer.
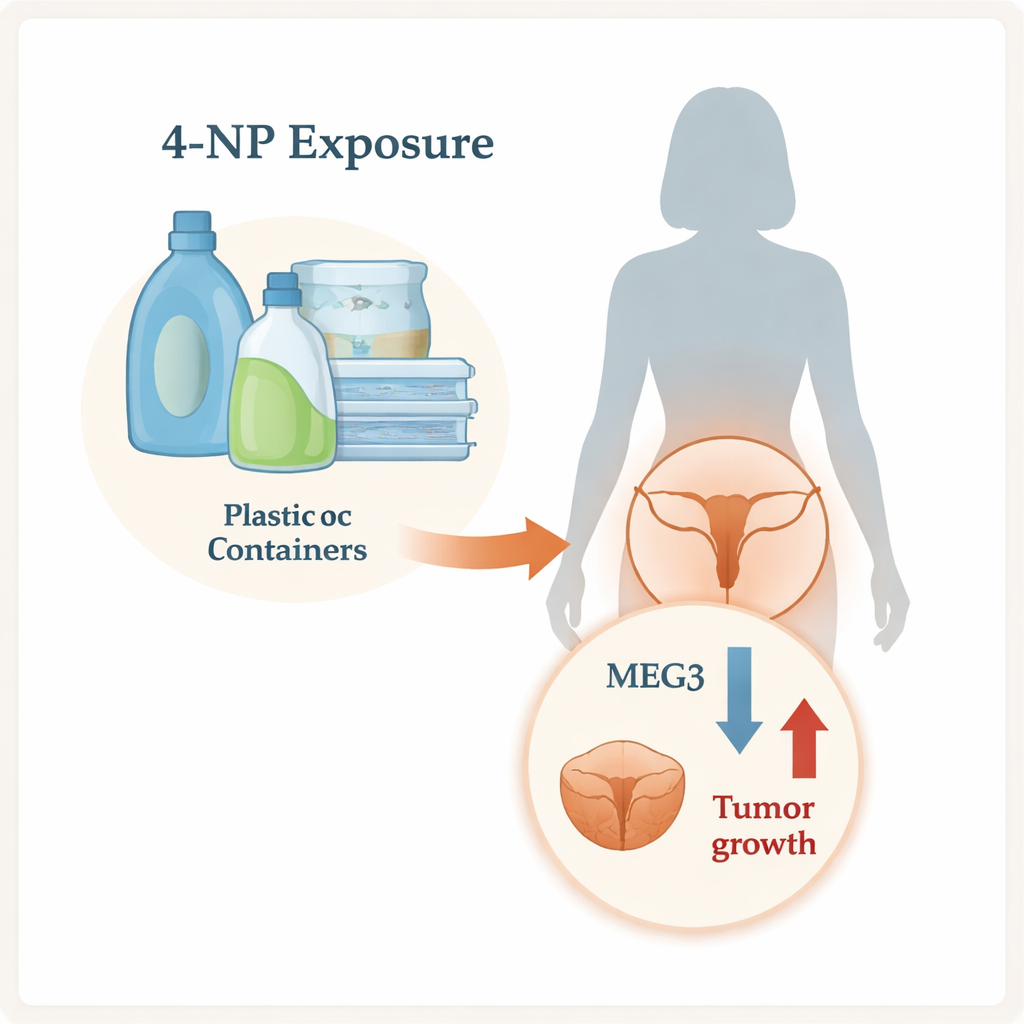
A Common Pollutant Under the Microscope
4‑NP is widely used in industrial products and can leak into water, soil, and ultimately the human body. It behaves a bit like estrogen, a key female hormone, and has been linked to problems in the reproductive system. The researchers first asked a simple question: do women with cervical cancer carry higher levels of this chemical inside their bodies? By measuring 4‑NP in urine from 35 women with cervical cancer and 35 healthy women, they found that patients had, on average, almost six times more 4‑NP. This does not prove that the chemical causes cancer, but it does show that women with cervical cancer tend to have much higher exposure.
The Body’s Built‑In Brake: MEG3
Inside our cells, long strands of RNA that do not code for proteins can still control how cells grow and die. One of these, called MEG3, normally acts as a brake on tumor growth. The team compared cervical cancer tissue with nearby healthy tissue from 31 patients and found that MEG3 levels were much lower in tumors. They saw the same pattern in cell experiments: cervical cancer cells (HeLa) had less MEG3 than normal cervical cells, and when the cells were exposed to 4‑NP, MEG3 dropped even further over time. This suggests that both cancer itself and environmental exposure to 4‑NP may work together to silence a natural tumor‑suppressing signal.
When the Brake Is Restored
Next, the scientists tested what would happen if they forced cancer cells to make more MEG3. Using a viral delivery system to boost MEG3 in HeLa cells, they watched the cells slow down. The cancer cells divided less, moved more sluggishly across a dish (a sign of lower ability to invade), and were more likely to undergo programmed cell death. Cell‑cycle tests showed that MEG3 pushed cells to pause before copying their DNA, another way of limiting growth. In mice, tumors grown from MEG3‑boosted HeLa cells were dramatically smaller and lighter than those from regular cancer cells, confirming that MEG3 can restrain tumor growth in a living organism.
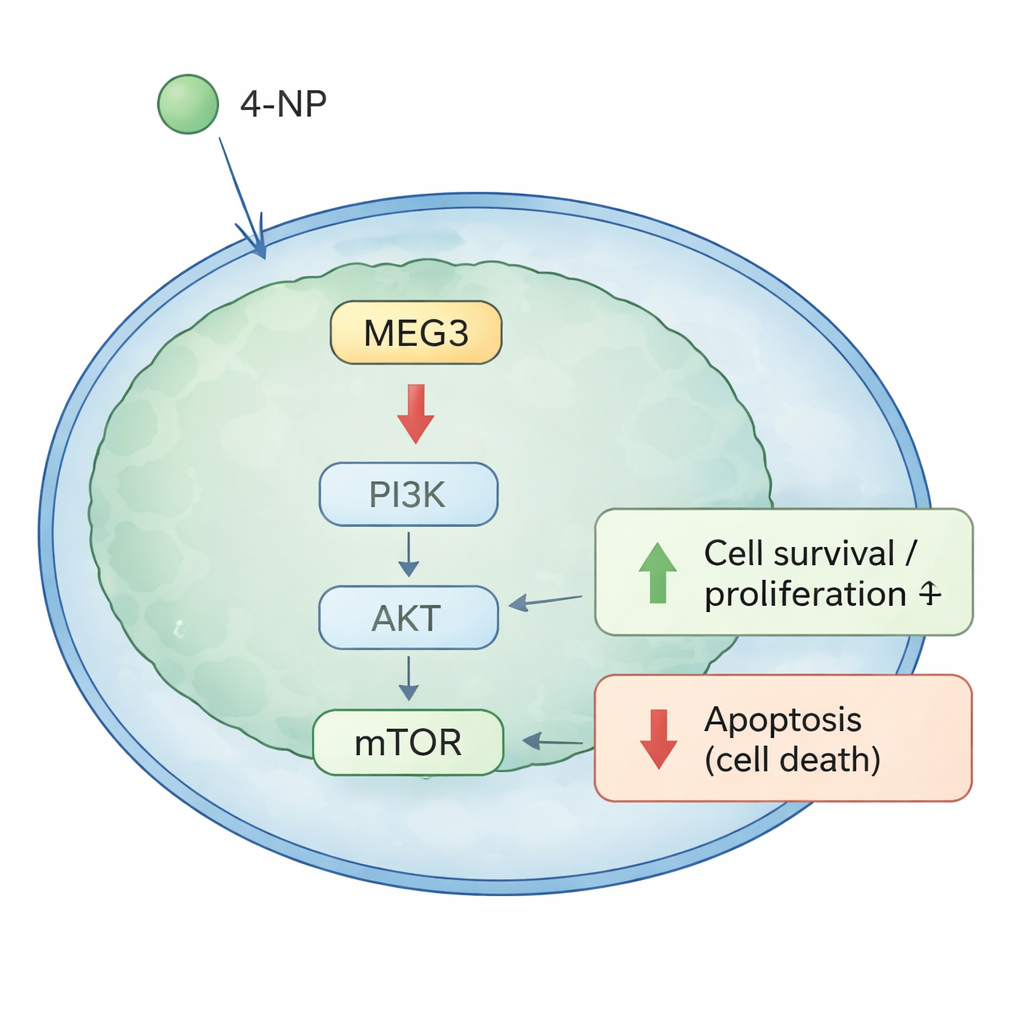
A Growth Switch Inside Cancer Cells
To understand how MEG3 exerts this control, the researchers focused on a well‑known growth switch in cells called the PI3K/AKT pathway. When this pathway is active, cells tend to grow and survive; when it is dampened, cells are more likely to die. In both cell cultures and mouse tumors, boosting MEG3 reduced the active (phosphorylated) forms of PI3K, AKT, and mTOR, as well as BCL‑2, a protein that protects cells from death, while favoring pro‑death signals. In contrast, exposing cells to 4‑NP had the opposite effect: it increased signals for growth and survival and reduced cell death. Importantly, when MEG3 was restored, it largely reversed the pro‑growth, anti‑death effects of 4‑NP, implying that this RNA acts as a key mediator between environmental exposure and cancer cell behavior.
What This Means for Patients and Prevention
For non‑specialists, the message is that a common environmental chemical appears linked to higher internal exposure in women with cervical cancer and can push cancer cells to grow faster and die less often in the lab. MEG3, a natural molecule in our cells, works like a safeguard against this process by dialing down a powerful growth pathway. While the study cannot yet prove that 4‑NP causes cervical cancer, and it was limited in size and models, it highlights a new chain of events: 4‑NP lowers MEG3, which releases a growth switch and helps tumors thrive. In the future, monitoring both 4‑NP levels and MEG3 activity could aid in assessing environmental risk, and therapies that restore MEG3 or block the same growth pathway may offer new options for women facing cervical cancer.
Citation: Wu, W., Ren, X., Chen, Y. et al. 4-Nonylphenol regulates cell proliferation and apoptosis in cervical carcinoma through the MEG3/PI3K/AKT signaling pathway. Sci Rep 16, 6067 (2026). https://doi.org/10.1038/s41598-026-36863-x
Keywords: cervical cancer, environmental pollutants, 4-nonylphenol, long non-coding RNA MEG3, PI3K AKT pathway