Clear Sky Science · en
Evaluation of cytotoxicity, wound healing, and anti-inflammatory effects of netarsudil on human corneal epithelial cells
Why an Eye Drop’s “Sweet Spot” Matters
Glaucoma eye drops can save sight by lowering pressure inside the eye, but long-term use often leaves patients battling burning, redness, and dryness. This study looks closely at one modern glaucoma drug, netarsudil (sold as Rhopressa), and asks a simple but critical question: at what point does a helpful medicine start to hurt the clear surface of the eye? By testing different dilutions of the commercial eye drop on human corneal cells in the lab, the researchers uncover a narrow “sweet spot” where the drug appears to support healing and calm inflammation, and higher doses where it becomes toxic.
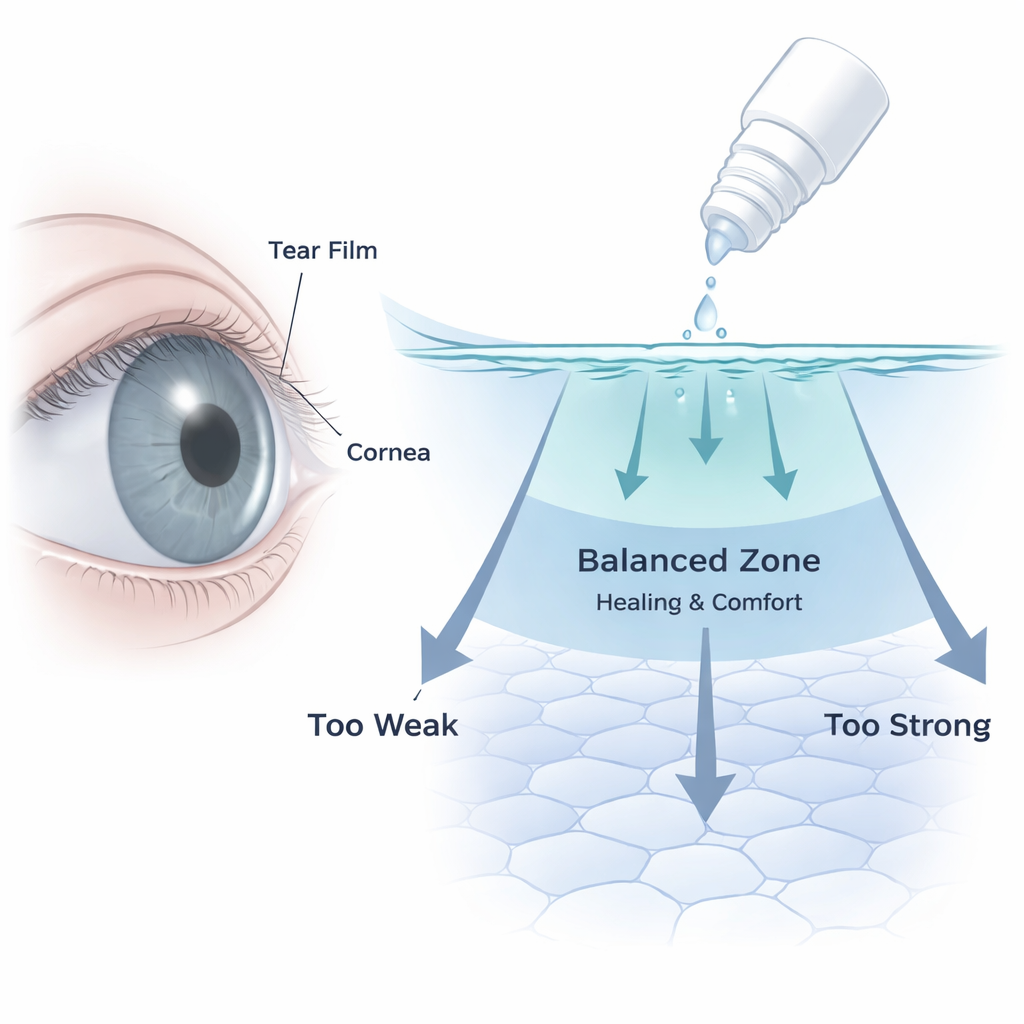
The Problem with Helpful but Harsh Eye Drops
Glaucoma slowly damages the optic nerve and is a leading cause of irreversible blindness worldwide. The standard way to slow it down is to lower eye pressure with daily eye drops. But these medications bathe the front of the eye for years, and many patients develop ocular surface disease: dry, irritated eyes with blurred or fluctuating vision. The causes are several—active drug molecules, preservatives such as benzalkonium chloride, and eye-drop formulas that are too acidic or salty compared with natural tears. Netarsudil belongs to a newer class of drugs called ROCK inhibitors, which work by relaxing internal drainage tissues rather than by changing fluid production. Because the same ROCK pathway also controls how corneal cells hold their shape, stick together, move, and respond to injury, there has been concern—and some clinical case reports—that netarsudil might disturb the corneal surface in ways we do not yet fully understand.
Testing Netarsudil on the Eye’s Front-Line Cells
The team used an established human corneal epithelial cell line—the type of cells that form the eye’s clear outermost layer—and exposed them to diluted versions of the actual netarsudil bottle patients receive. They checked basic physical properties first: the drop solution had an acidic pH of 5.5 (tears are closer to neutral, around 7.0–7.5) and a slightly lower salt concentration than natural tears, but an overall saltiness (osmolarity) still near the physiological range. They then created a range of working solutions by mixing the commercial drop with growth medium to final strengths of 0.1%, 0.5%, 1%, and 2% by volume, and compared them with a drug-free control. Across these dilutions and over times from 30 minutes to 24 hours, they measured whether cells stayed alive, whether their membranes leaked, how their shape and fine structure changed under the microscope, and how quickly they could close an artificial “scratch” wound.
A Narrow Window Between Healing and Harm
The results showed a clear, two-phase pattern. Very low concentrations (0.1%) caused only mild stress, while a mid-level dilution (0.5%) emerged as a sublethal “optimal zone.” At this level, overall cell survival remained high, the cell layer stayed relatively intact, and scratch wounds in the cell sheet closed faster than in untreated controls, suggesting better repair. In contrast, stronger exposures at 1% and 2% sharply reduced cell survival and increased leakage of an enzyme (LDH) that signals membrane damage, especially after 12–24 hours. Under standard and electron microscopes, cells at these higher dilutions rounded up, lost their fine surface projections, developed large internal bubbles (vacuoles), and showed clumped DNA—hallmarks of cells on the path to death. In other words, as the effective concentration rose, the same drug that could support healing at one level became clearly harmful beyond a critical threshold.
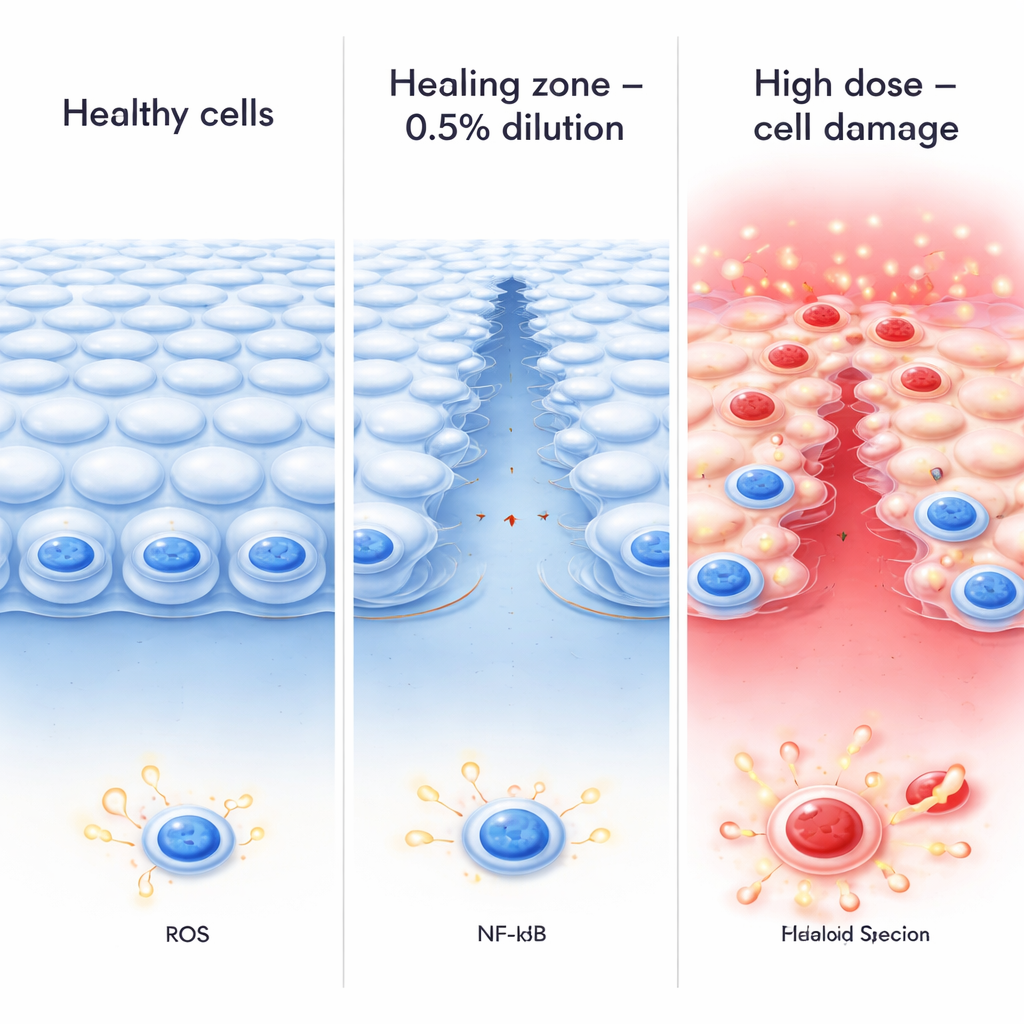
Calming Inflammation in a Stressed Eye Surface
The researchers also asked how netarsudil behaves when the eye’s surface is already inflamed. They mimicked bacterial injury by adding lipopolysaccharide (LPS), a molecule that triggers an immune response, and watched how corneal cells fared with or without the 0.5% dilution. LPS alone lowered cell survival, boosted the production of reactive oxygen species (damaging oxygen-based chemicals), and drove a key inflammatory regulator called NF-κB into the cell nucleus, where it switches on inflammatory genes. When cells were co-treated with LPS and the 0.5% netarsudil dilution, they survived better, produced less reactive oxygen, and showed reduced nuclear NF-κB. This suggests that, at least in this mid-range zone, netarsudil can dampen inflammatory signaling rather than amplify it.
What This Means for People Using Glaucoma Drops
To a lay reader, the takeaway is that the same netarsudil eye drop can push corneal cells toward healing or toward damage, depending on how much effectively reaches them and for how long. In a controlled lab dish, a 0.5% dilution of the commercial formulation sped up wound closure and softened inflammatory responses, while higher dilutions of the same product killed cells and disrupted their structure. Real eyes are more complex: blinking, tear turnover, and natural defenses dilute and clear drops quickly, and the product includes preservatives and other ingredients that may also play a role. Still, these findings give eye doctors and drug designers a mechanistic clue: there is a narrow therapeutic window where ROCK inhibition may support a healthier corneal surface, and pushing beyond it risks tipping the balance toward toxicity. Future work that links these lab-based thresholds to real-world dosing and tear-film dynamics could help refine formulations and schedules that protect both vision and day-to-day comfort for people living with glaucoma.
Citation: Han, K.E., Ahn, J.H., Kim, SJ. et al. Evaluation of cytotoxicity, wound healing, and anti-inflammatory effects of netarsudil on human corneal epithelial cells. Sci Rep 16, 6164 (2026). https://doi.org/10.1038/s41598-026-36860-0
Keywords: glaucoma eye drops, netarsudil, corneal epithelium, ocular surface disease, ROCK inhibitor