Clear Sky Science · en
Differential mechanisms of SARS-CoV-2 inactivation by anionic surfactants: a comparative study of fatty acid salts and synthetic surfactants
Why the Soap You Use Still Matters
The COVID‑19 pandemic made handwashing a household mantra, but not all soaps work the same way against the virus that causes the disease. This study looks under the microscope at common soap ingredients to see which ones are best at disarming SARS‑CoV‑2, and how they do it. The findings could guide better choices for everyday soaps and disinfectants, making a routine act like washing your hands an even more powerful tool for staying healthy.
How Soap Fights a Greasy Shield
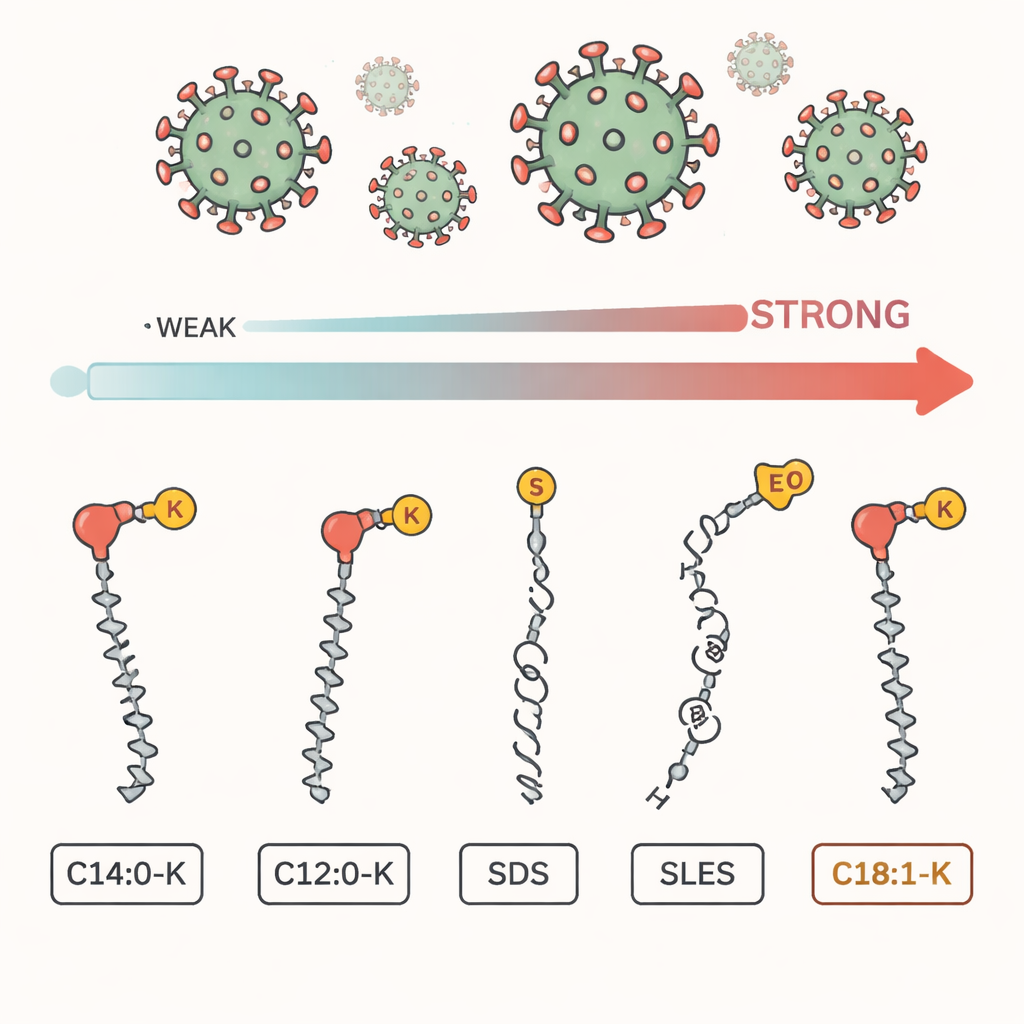
SARS‑CoV‑2, like many dangerous viruses, is wrapped in a fragile, oily shell made of fats. Soap molecules have two faces: one side loves water, the other loves oil. When we wash our hands, these molecules wedge themselves into that greasy shell and can tear it apart, making the virus unable to infect cells. But soaps are made from different ingredients, and this study compared five widely used ones: three natural fatty acid salts (including potassium oleate, called C18:1‑K) and two common synthetic surfactants, SDS and SLES, which are often found in liquid soaps and shampoos.
Which Soap Ingredients Hit the Virus Hardest
When the researchers mixed the virus with these surfactants in the lab, one ingredient stood out. C18:1‑K, a natural soap component with a long, slightly kinked tail, reduced the virus’s infectivity by more than 100,000‑fold at very low concentration. SDS, a harsher synthetic ingredient, only managed about a tenfold reduction at the same level, and SLES and a shorter‑tailed soap ingredient, C12:0‑K, barely inactivated the virus. Overall, the ranking was clear: C18:1‑K worked best, followed by SDS, then SLES, with the shorter fatty acids lagging behind. Longer, more oil‑loving chains made for much more powerful virus inactivation.
Peeking at Invisible Battles
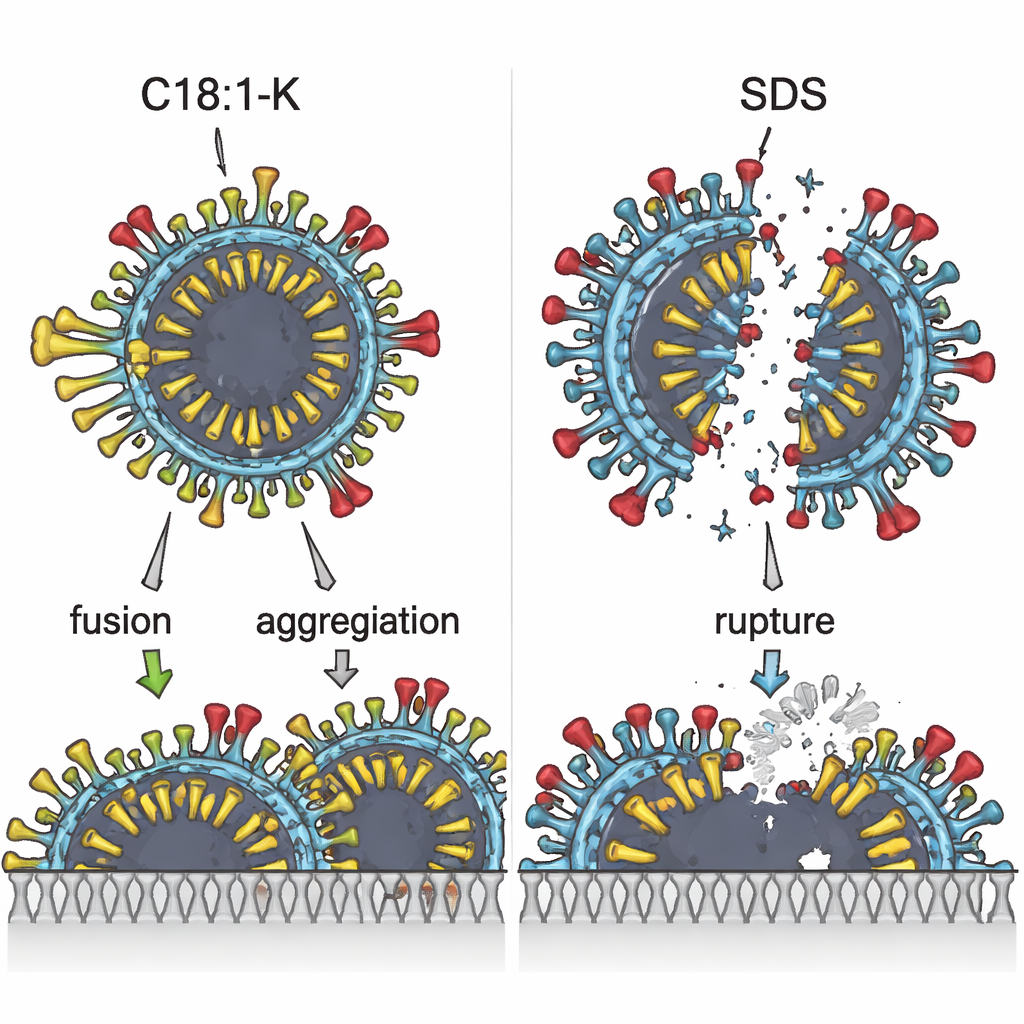
To understand why these differences were so large, the team used a heat‑measuring technique to track how surfactants interact with the virus. C18:1‑K produced a heat pattern showing it mainly burrows its oily tail into the virus’s fat shell, a process driven by hydrophobic (oil‑loving) forces. SDS and C12:0‑K, by contrast, showed the opposite kind of heat change, signaling that they cling more to charged protein parts on the virus surface rather than deeply disturbing the fatty shell. SLES appeared to sit in between, with its oil and water‑loving tendencies partly cancelling each other out. These energetic fingerprints revealed that not just strength, but the mode of attack, differed between ingredients.
What the Microscope Revealed
Electron microscopy gave a visual counterpart to these invisible energy shifts. When the virus was treated with surfactants that acted mainly through electric attraction to proteins, like SDS or C12:0‑K, many particles looked torn open or ruptured. With C18:1‑K, however, the virus particles more often appeared fused or clumped together, without obvious bursting. All surfactants caused some degree of virus aggregation at higher concentrations, but only C18:1‑K produced extensive “membrane‑fused” clusters, which are unlikely to remain infectious. The ingredient that best inactivated the virus, C18:1‑K, also had the lowest critical micelle concentration—a sign that its oily tails pack together easily and can strongly disturb the viral envelope once a certain threshold is reached.
What This Means for Everyday Protection
For non‑specialists, the take‑home message is straightforward: hand soap really does chemically damage SARS‑CoV‑2, and some naturally derived ingredients are unusually good at it. Soaps rich in long‑chain fatty acid salts like potassium oleate can attack the virus’s oily shell through strong oil‑seeking interactions and encourage virus particles to fuse and clump, making them harmless. While all surfactants tested can contribute to inactivation, those designed or chosen for strong hydrophobic action may offer superior protection. These insights can help chemists design more effective, skin‑friendly soaps and disinfectants not only against SARS‑CoV‑2, but also against other envelope‑wrapped viruses that rely on the same kind of fragile fatty coat.
Citation: Yamamoto, A., Iseki, Y., Elsayed, A.M.A. et al. Differential mechanisms of SARS-CoV-2 inactivation by anionic surfactants: a comparative study of fatty acid salts and synthetic surfactants. Sci Rep 16, 6394 (2026). https://doi.org/10.1038/s41598-026-36858-8
Keywords: handwashing, SARS-CoV-2, soap surfactants, viral envelope, disinfectants