Clear Sky Science · en
Effect of solid-state polymerization on fiber structure development in melt spinning of mechanical recycled PET
Turning Bottle Waste into Tough New Fibers
Plastic drink bottles are everywhere, and most are made from PET, a versatile but stubborn material that doesn’t break down in nature. Much of this plastic ends up in landfills or the environment. This study explores whether old PET bottles, after simple mechanical recycling, can be upgraded into strong, reliable industrial fibers—like those used in seat belts, geotextiles, and industrial fabrics—so that yesterday’s bottle can safely become tomorrow’s heavy‑duty yarn.
Why Recycled PET Usually Falls Short
When PET bottles are mechanically recycled, they are collected, cleaned, ground into flakes, and remelted into new pellets. This process is cheaper and simpler than chemical recycling, but the heat and moisture involved quietly damage the long PET chains, cutting them into shorter pieces. That damage lowers a key measure called intrinsic viscosity, which scientists use as a stand‑in for molecular weight and, ultimately, strength. As a result, ordinary mechanically recycled PET (mr‑PET) is usually fine for lower‑value uses, but it struggles to meet the demanding strength and durability needed for industrial fibers.
Boosting Polymer Chains with Gentle Heat
To repair those shortened chains, the researchers used a process called solid‑state polymerization (SSP). Instead of melting the plastic, they heated PET pellets to temperatures above their glass‑transition point but below their melting point, and held them there for several hours in a rotating, vacuum reactor. Under these conditions, the ends of the polymer chains slowly reconnect, increasing chain length without the severe breakdown that can occur in full‑melt processing. The team tested a range of temperatures (220, 230, and 240 °C) and times (6, 12, and 18 hours) for both virgin PET (v‑PET) and mr‑PET. They then measured how easily the material flowed when melted, how viscous its solutions were, and how its molecular weight changed, to track how well the chains were rebuilt.
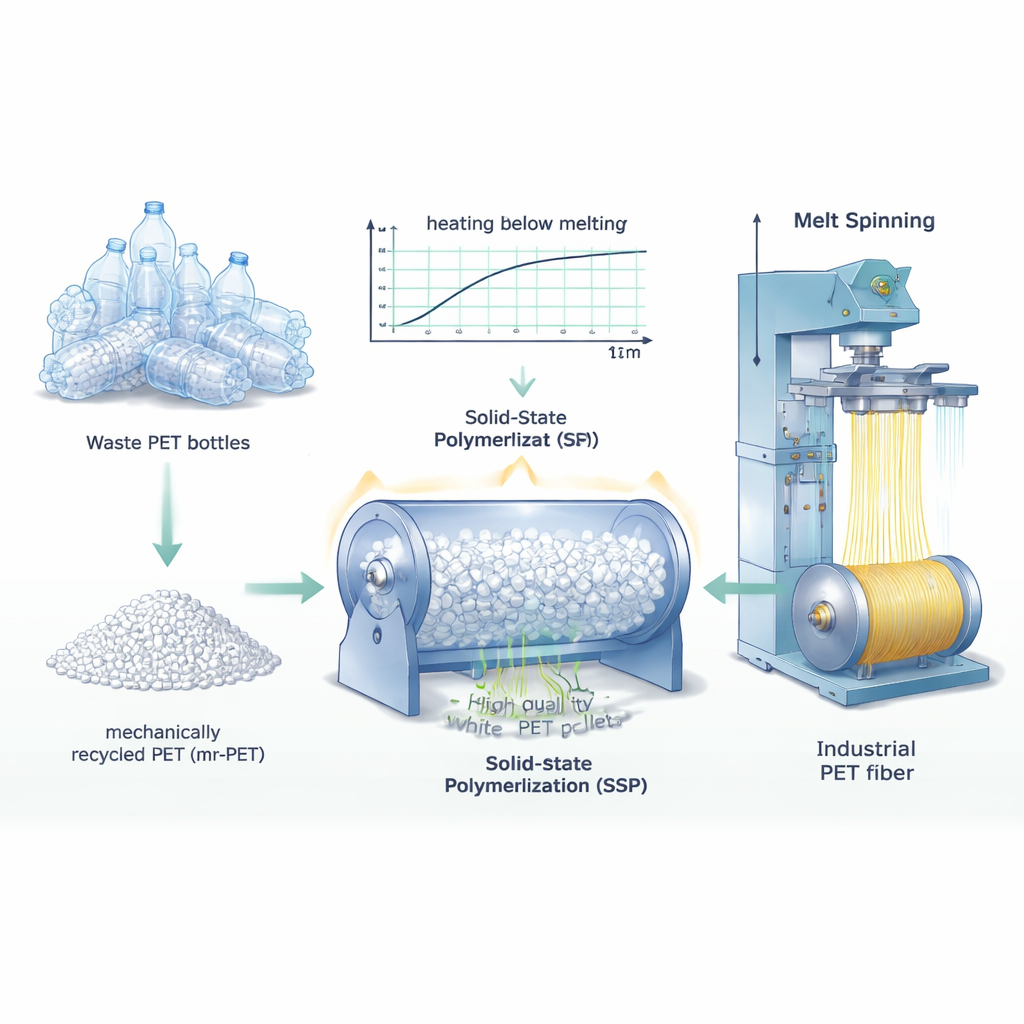
Finding the Sweet Spot for Recycling
The analyses showed that both the chain length and crystallinity—how orderly the polymer structure becomes—increased as SSP temperature and time rose. However, longer and hotter treatments also meant more energy use and slower production. The researchers identified 230 °C for 6 hours as a practical sweet spot: under this condition, mr‑PET reached an intrinsic viscosity of about 1.1 dL/g, the level often targeted for high‑strength industrial fibers, while keeping process time reasonable. At this setting, the recycled PET’s average molecular weight closely matched that of virgin PET treated the same way, even though the recycled material still contained traces of impurities from its previous life as bottles.
Spinning Fast to Build Structure
Next, the team melted both the treated and untreated PET pellets and extruded them through tiny holes to form filaments, which were pulled away at high speed—a method known as melt spinning. By changing the take‑up velocity from 1000 to 4000 meters per minute, they could control how much the molten threads were stretched as they cooled. Using thermal analysis and X‑ray diffraction, they found that higher spinning speeds encouraged the PET chains to align and crystallize along the fiber axis, which in turn raised the melting point and internal order of the fibers. Interestingly, fibers made from SSP‑treated PET began to show clear crystalline structure at lower speeds than untreated PET, meaning the repaired, longer chains were more ready to organize into strong, ordered regions during spinning.
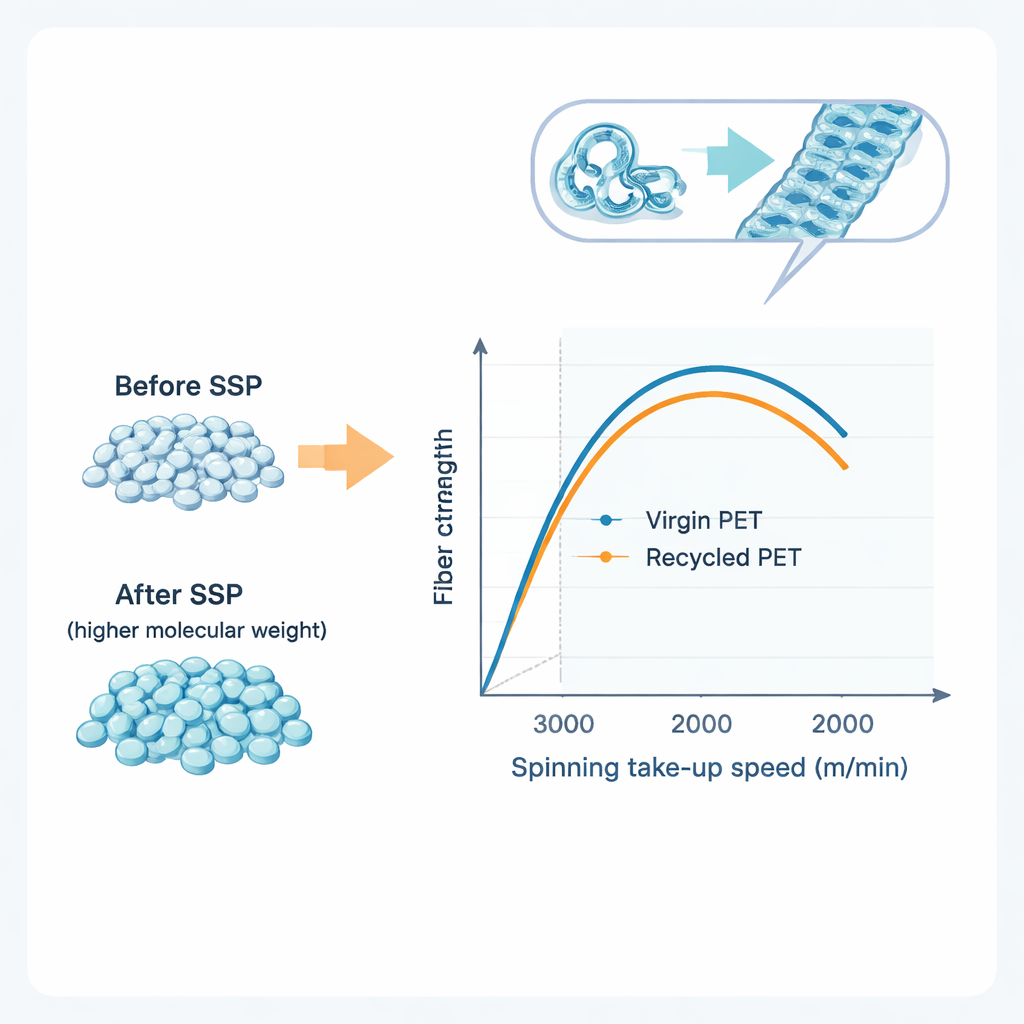
Strength That Rivals Virgin Plastic
Mechanical tests of the resulting filaments confirmed what the structural measurements suggested. As the spinning speed increased, all fibers became stronger (higher tenacity) but stretched less before breaking, a hallmark of more oriented, crystalline material. After SSP, both virgin and recycled PET showed better performance overall. Most notably, when mr‑PET that had undergone SSP at 230 °C for 6 hours was melt‑spun at 3000 m/min, its tenacity was essentially the same as that of similarly processed virgin PET fibers, around 4.4 grams per denier. In other words, despite carrying a history of use, collection, and reprocessing, the recycled material could be engineered to match the strength of “new” PET in industrial‑grade yarns.
What This Means for Everyday Products
For non‑specialists, the takeaway is straightforward: with carefully tuned heat treatment and spinning conditions, plastic bottles can be turned into high‑performance fibers suitable for demanding industrial uses, not just low‑end products. By using SSP to rebuild polymer chains and optimizing spinning speed to align them, this study shows that mechanically recycled PET can overcome its usual weaknesses and stand shoulder to shoulder with virgin material. That opens the door to more circular use of PET, where hard‑working technical textiles—think car parts, construction fabrics, and heavy‑duty ropes—can be made from the same bottles we once tossed into the recycling bin.
Citation: Kim, H., Bae, J.H., Hahm, WG. et al. Effect of solid-state polymerization on fiber structure development in melt spinning of mechanical recycled PET. Sci Rep 16, 6752 (2026). https://doi.org/10.1038/s41598-026-36850-2
Keywords: recycled PET fibers, solid-state polymerization, melt spinning, plastic bottle recycling, industrial polyester yarn