Clear Sky Science · en
Pharmacokinetics, pathology and efficacy of SARS-CoV-2 main protease inhibitor VPC285785 in a murine model of coronavirus infection
Why we still need better COVID pills
The first antiviral pills for COVID-19, such as Paxlovid, were a turning point in the pandemic. But they are not perfect: they must be taken very early, require multiple tablets a day, and can dangerously interfere with other medicines. This study explores new drug candidates designed to block the coronavirus in more than one way, work well as single pills taken by mouth, and cause fewer drug–drug problems, using a mouse model of coronavirus infection.
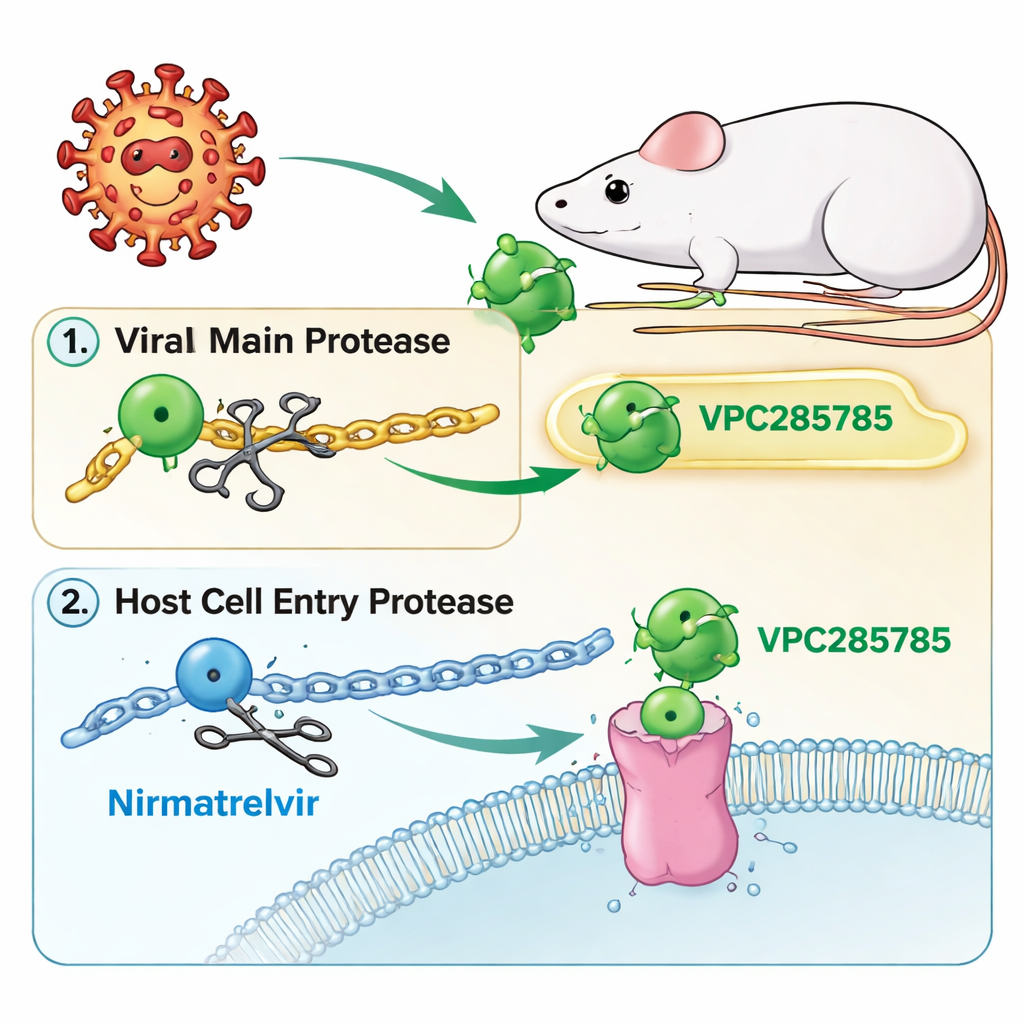
A new twist on the COVID-19 protease target
Like Paxlovid, the new compounds—called VPC285785 and VPC285786—aim at the virus’s “main protease,” a molecular scissors that cuts long viral proteins into working parts. If these scissors are jammed, the virus cannot finish building itself and stalls. The researchers also designed the molecules to hit a second protein in our own cells, cathepsin L, which helps some coronaviruses get inside. By aiming at both the viral scissors and a host entry helper, the team hoped to create a dual-action pill that might be harder for the virus to escape and would not need a booster drug.
Built for staying power, not for a booster
Paxlovid combines nirmatrelvir with another drug, ritonavir, whose job is not to fight the virus but to slow nirmatrelvir’s breakdown in the liver. Ritonavir, however, interferes with many enzymes that process common medications, leading to complex and sometimes dangerous interactions. VPC285785 and VPC285786 were chemically tuned to resist breakdown on their own, using features such as fluorine atoms and ring “stiffening” that make them less attractive to liver enzymes. In laboratory tests with human and mouse liver preparations, both new compounds proved at least as stable as nirmatrelvir, and VPC285786 was in some ways even sturdier, without any ritonavir present.
How the drugs behave and move in mice
The team then examined how the compounds travel through the body of mice, measuring how fast they disappear from blood and how well they are absorbed by mouth. After injection, VPC285785 and VPC285786 lingered in the bloodstream about as long as nirmatrelvir and reached higher overall exposure. When given orally, however, their paths diverged: VPC285785 achieved moderate but workable absorption (about 15% of the swallowed dose reaching the circulation), whereas VPC285786 barely entered the blood (around 3%). Because pills need to cross the gut wall to be practical treatments, only VPC285785 was taken forward into infection experiments.
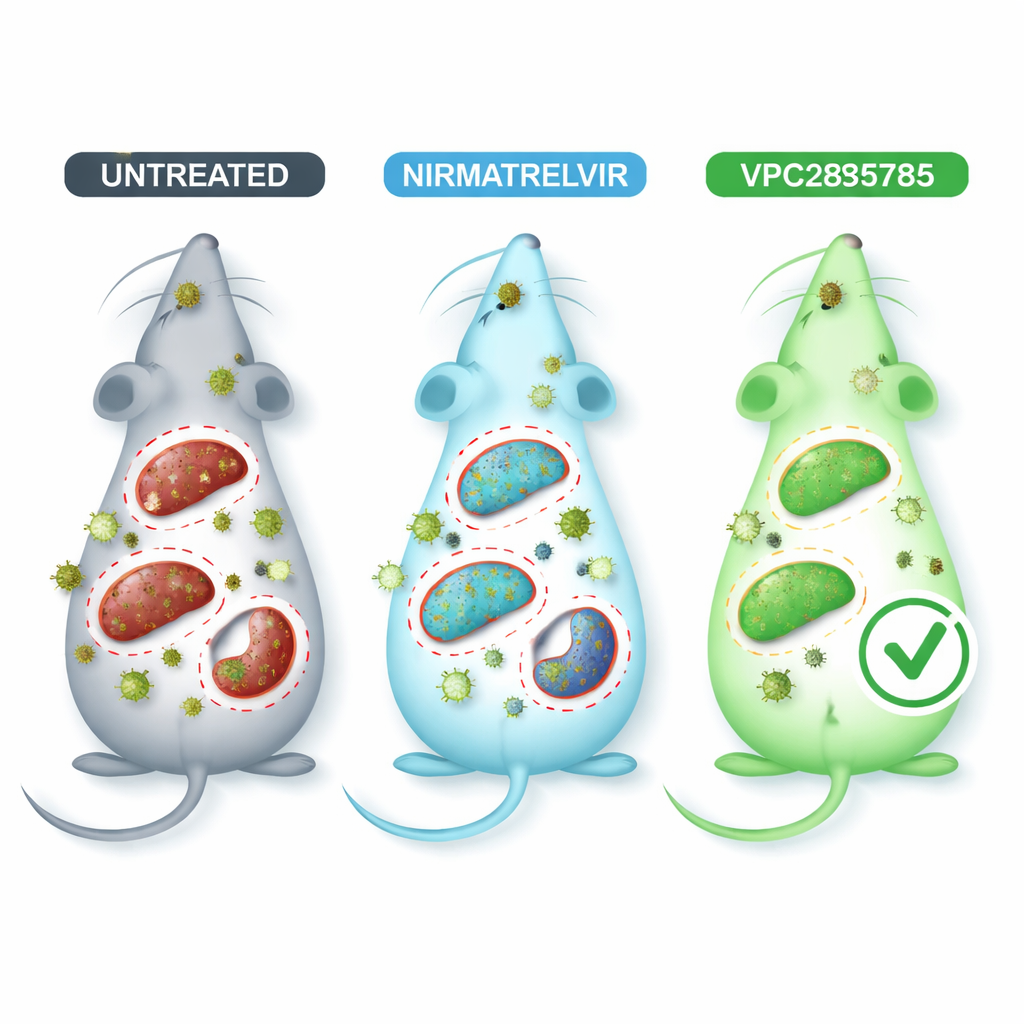
Putting the new pill to the test in infected mice
To mimic coronavirus disease in a safer setting, the researchers used a mouse coronavirus called MHV-A59, which invades multiple organs and causes lung and liver damage similar in many ways to human infection. Infected mice were treated by mouth with VPC285785, with nirmatrelvir as a comparison, or with an inactive vehicle. Blood tests showed that infection strained the liver and kidneys, but animals given VPC285785 or nirmatrelvir tended to have healthier liver enzyme and kidney markers than untreated mice. Most strikingly, VPC285785 sharply lowered the amount of viral genetic material in the liver, brain, and spleen, while nirmatrelvir produced a clear drop only in the brain. In the lungs, heart, and kidneys, neither treatment made a strong difference under the conditions tested.
What this could mean for future COVID treatments
Although VPC285785 is less potent against the viral protease in a test tube than nirmatrelvir, it performs well in living animals, significantly reducing virus levels in several key organs while preserving organ function, all without a ritonavir booster. Its sister compound, VPC285786, shows that even stronger dual targeting of the viral protease and cathepsin L is possible, but its poor absorption must be fixed. Together, these results suggest that next‑generation COVID pills could be designed as single, orally available drugs that hit both viral and host targets, offer fewer dangerous interactions with other medications, and still deliver strong protection in vital tissues.
Citation: Smith, J.R., Toro, A., Sabater, A. et al. Pharmacokinetics, pathology and efficacy of SARS-CoV-2 main protease inhibitor VPC285785 in a murine model of coronavirus infection. Sci Rep 16, 6905 (2026). https://doi.org/10.1038/s41598-026-36842-2
Keywords: SARS-CoV-2 antivirals, main protease inhibitors, Paxlovid alternatives, murine coronavirus model, oral COVID treatment