Clear Sky Science · en
YTHDC1 modulates the malignant phenotype of retinoblastoma via SQSTM1-mediated autophagy
Why this matters for children’s eye cancer
Retinoblastoma is the most common eye cancer in young children. Doctors can often cure it if caught early, but once the tumor starts invading tissues around the eye or spreading toward the brain, saving both the child’s life and vision becomes much harder. This study explores a molecular “brake” inside tumor cells—two molecules called YTHDC1 and SQSTM1—that seem to help keep retinoblastoma less aggressive. Understanding how this brake works could open new paths for gentler, more targeted treatments.
A closer look at childhood eye tumors
Retinoblastoma arises from the light-sensing layer of the eye, the retina, usually in children under five. Classically, the disease begins when both copies of a guardian gene, RB1, are lost. But RB1 loss alone does not fully explain why some tumors stay confined to the eye while others burrow into nearby tissues or spread along the optic nerve. Increasingly, researchers have realized that chemical tags placed on RNA—the temporary messages that tell cells which proteins to make—can strongly influence how cancers behave. One of the most common tags, called m6A, can change how stable these messages are and how long they last inside the cell.
Discovering a missing molecular brake
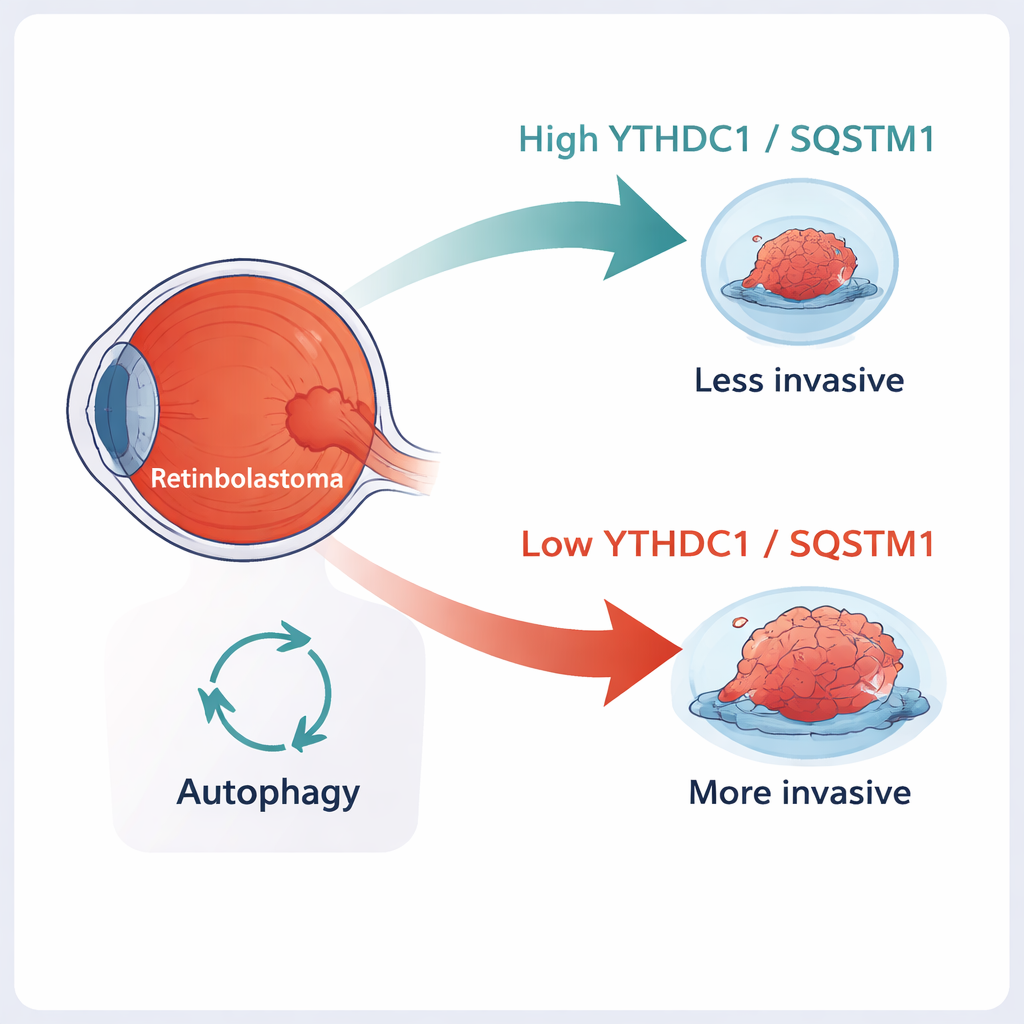
The authors began by comparing genetic activity in tumors that stayed inside the eye with those that had already become invasive. Using a public RNA sequencing dataset from retinoblastoma samples, they identified thousands of genes whose activity differed between the two groups and then focused on key m6A “regulators.” These include enzymes that add or remove m6A and “reader” proteins that interpret the tag. Among ten major regulators, they found that one reader, YTHDC1, was consistently lower in invasive tumors. When they examined samples from 50 children and performed laboratory tests on tumor tissue, they confirmed that YTHDC1 levels—both its RNA and protein—were reduced in more aggressive cancers.
Putting YTHDC1 to the test
To see what YTHDC1 actually does, the team manipulated its levels in two human retinoblastoma cell lines. When they lowered YTHDC1 using genetic tools, the cells multiplied faster and were better able to move through artificial membranes that mimic tissue barriers—signs of a more invasive tumor. In mice, cells lacking YTHDC1 formed larger, heavier tumors. The opposite was also true: forcing cells to make extra YTHDC1 slowed their growth and reduced their ability to migrate and invade. These experiments suggest that YTHDC1 acts as a tumor suppressor, helping to restrain retinoblastoma’s most dangerous behaviors.
How YTHDC1 controls the cell’s recycling system
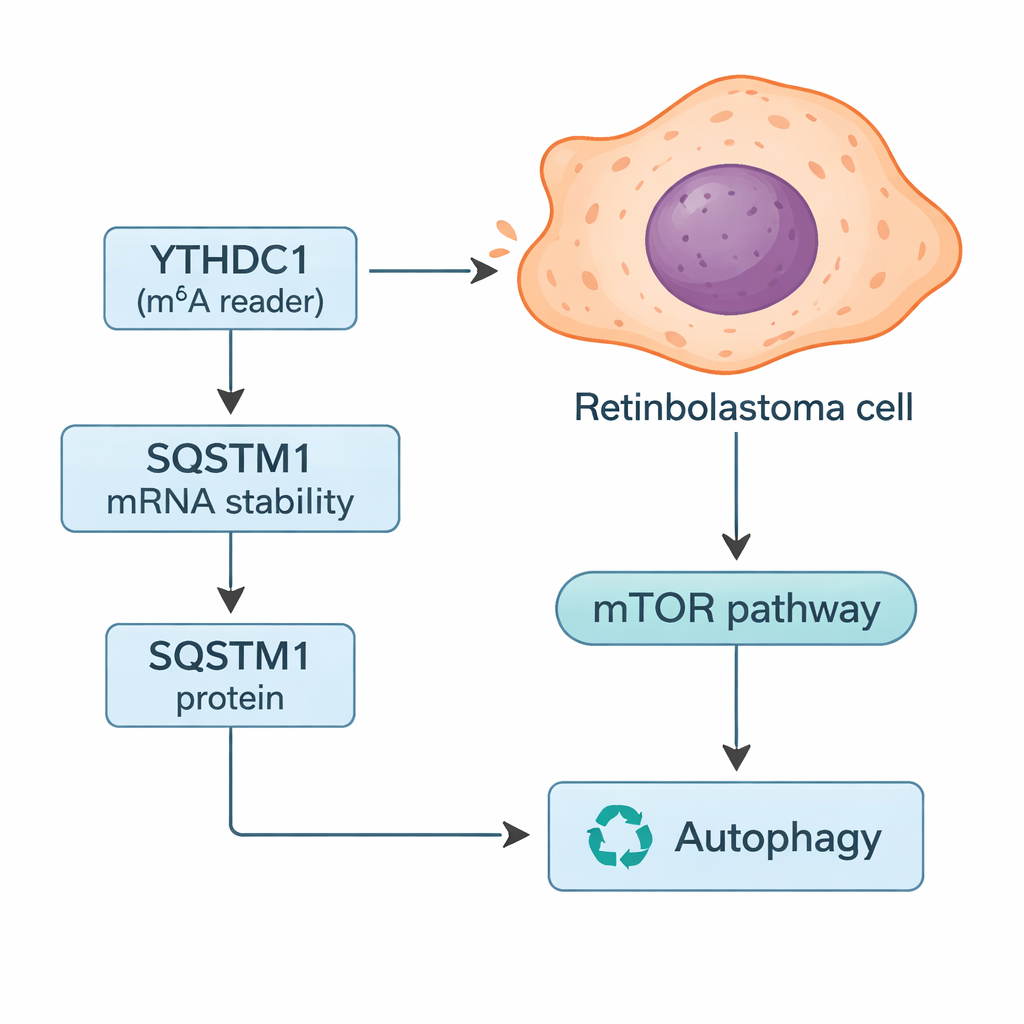
Diving deeper, the researchers looked for specific genes whose messages might be controlled by YTHDC1. By combining database searches with their tumor data, they homed in on SQSTM1, a protein better known as p62. SQSTM1 helps manage autophagy, the cell’s recycling system that breaks down damaged components and can provide fuel when resources are scarce. The study showed that YTHDC1 physically binds to SQSTM1’s RNA and stabilizes it, allowing more SQSTM1 protein to be made. When YTHDC1 was reduced, SQSTM1 levels dropped and its RNA decayed more quickly. Silencing SQSTM1 alone made retinoblastoma cells grow and invade more, and it partially erased the protective effects seen when YTHDC1 was overproduced, placing SQSTM1 as a key middle player in this pathway.
Autophagy, energy, and tumor aggressiveness
Because SQSTM1 is central to autophagy, the team tested how altering YTHDC1 and SQSTM1 changed this recycling process. Using a combination of fluorescent markers and protein measurements, they found that lowering either YTHDC1 or SQSTM1 boosted autophagic “flux”—more cellular material was being shuttled into and through the recycling system. They also observed changes in the mTOR pathway, a major nutrient-sensing switch that normally keeps autophagy in check. With less SQSTM1, mTOR activity fell, which is consistent with turning autophagy up. In invasive retinoblastoma, the authors propose that reduced YTHDC1 leads to weaker SQSTM1 signals, dampened mTOR activity, and heightened autophagy that helps tumor cells survive stress and spread.
What this means for future treatments
To a non-specialist, the take-home message is that this study identifies a chain of events inside retinoblastoma cells—YTHDC1 controlling SQSTM1, which in turn shapes the cell’s recycling machinery—that influences how aggressive the cancer becomes. When this chain is weakened, tumors grow faster and invade more. While much work remains before these findings could reach the clinic, the pathway offers new ideas for therapy: restoring YTHDC1 function, stabilizing SQSTM1, or fine-tuning autophagy and mTOR activity might one day complement existing chemotherapies. For children facing this eye cancer, such targeted strategies could help limit tumor spread while preserving more vision and reducing treatment side effects.
Citation: Ding, J., Sun, J., Wang, J. et al. YTHDC1 modulates the malignant phenotype of retinoblastoma via SQSTM1-mediated autophagy. Sci Rep 16, 6292 (2026). https://doi.org/10.1038/s41598-026-36833-3
Keywords: retinoblastoma, YTHDC1, SQSTM1, autophagy, mTOR pathway