Clear Sky Science · en
Fecal carriage and molecular characterization of ESBL-producing Enterobacteriaceae among farmers in Mid-Western Uganda
Why germs in farmers’ guts matter to all of us
Antibiotic-resistant bacteria are often thought of as a problem confined to hospitals, but this study shows they are quietly spreading in everyday life, including on small farms in rural Uganda. The researchers looked at bacteria living in the intestines of farmers and asked: how common are highly drug-resistant strains, what makes them resistant, and what everyday conditions help them spread? Their findings shed light on how resistance can move between people, animals, and the environment—and why that matters for anyone who may one day need antibiotics to work.
Checking the health of gut bacteria on small farms
The team worked in Kibimba Parish, a largely agricultural area in Mid-Western Uganda where many families keep livestock and poultry. They recruited 250 adult farmers who volunteered a small stool sample. In the laboratory, scientists grew bacteria from these samples and focused on a family of microbes called Enterobacteriaceae, which includes familiar species like Escherichia coli. Using standard culture methods and biochemical tests, they identified which species were present. They then tested how these bacteria responded to a panel of commonly used antibiotics and used a special method to spot those capable of breaking down powerful drugs known as third-generation cephalosporins.
Hunting for resistance genes inside the microbes
To understand what made some bacteria so hard to treat, the researchers looked for three well-known resistance genes: blaCTX-M, blaTEM, and blaSHV. These genes code for enzymes called extended-spectrum beta-lactamases (ESBLs), which allow bacteria to inactivate many important antibiotics. Using a technique called PCR, they checked whether these genes were present in the ESBL-positive strains. They also examined whether single bacteria carried more than one resistance gene at the same time, which would suggest that resistance can be shared and built up through mobile genetic elements that move between microbes.
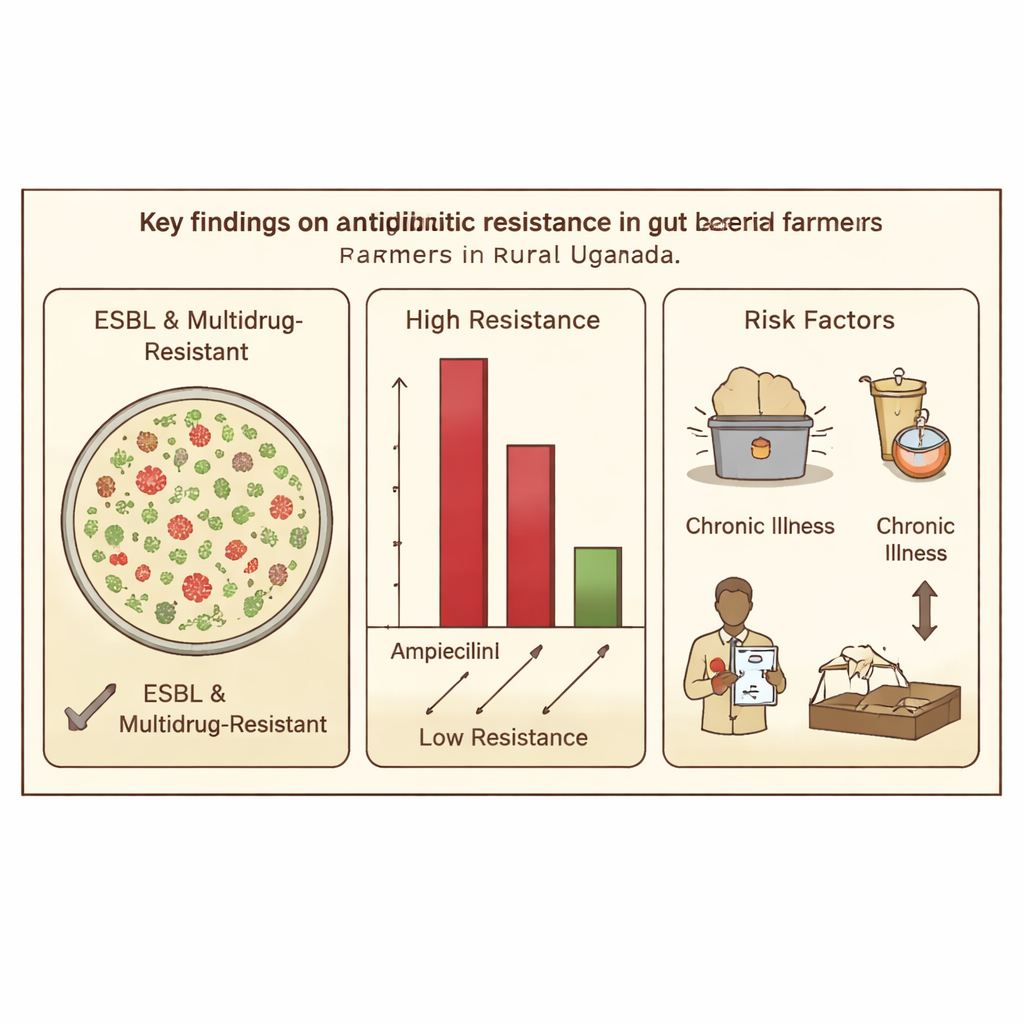
What the scientists found in farmers’ stools
Out of 250 farmers, more than one in three (36.4%) carried ESBL-producing bacteria in their intestines, showing that these hard-to-treat microbes are common even in people who are not in hospital. From 312 bacterial isolates grown from the samples, Escherichia coli dominated, making up about 70% of all strains and of the ESBL producers in particular. Many isolates were resistant to several different classes of drugs at once, a pattern known as multidrug resistance. Resistance was especially high to older, commonly used antibiotics such as ampicillin and piperacillin, while a last-resort drug, imipenem, remained largely effective, likely because it is expensive and less frequently used in rural settings.
Genes that power resistance and the role of daily life
Among the confirmed ESBL-producing bacteria, about four out of five carried at least one of the three key resistance genes. The most common was blaCTX-M, followed by blaTEM and blaSHV. Many bacteria carried more than one gene at once, and some harbored all three, indicating that these microbes have accumulated multiple tools for defeating antibiotics. The researchers also linked bacterial carriage to everyday conditions. Farmers who relied on shallow well water were more likely to carry ESBL-producing bacteria, suggesting that untreated water can act as a reservoir. Having chronic conditions such as hypertension or HIV/AIDS was also associated with higher odds of carrying resistant strains, possibly because of more frequent contact with healthcare and antibiotics. Interestingly, goat farming appeared to be linked with lower risk, hinting that different animals and husbandry styles may influence how resistance circulates.
What this means for farmers and the wider world
Taken together, the study paints a clear picture: in this rural Ugandan community, many farmers quietly carry gut bacteria that can withstand multiple antibiotics, powered by potent resistance genes. For a layperson, this means that infections which might once have been easily cured could become much harder to treat, even far from big-city hospitals. The findings support a “One Health” view, in which human health is tied to animals and the environment. Improving access to safe water, promoting better hygiene, using antibiotics more carefully in both people and livestock, and expanding simple lab tests in rural clinics could all help slow the spread of these dangerous microbes before they become even more entrenched.
Citation: Wilson, G., Micheal, K., Catherine, A. et al. Fecal carriage and molecular characterization of ESBL-producing Enterobacteriaceae among farmers in Mid-Western Uganda. Sci Rep 16, 6249 (2026). https://doi.org/10.1038/s41598-026-36817-3
Keywords: antibiotic resistance, ESBL bacteria, rural farming, One Health, Uganda