Clear Sky Science · en
Irisin regulates lipid metabolism and ferroptosis in ovarian cancer cells by modulating the ALOX5-5-HETE-PD-L1 axis
Why this plant molecule matters for ovarian cancer
Ovarian cancer is one of the deadliest cancers in women because it is usually found late and often comes back after treatment. This study explores whether a natural compound called tectorigenin, found in the plant Belamcanda chinensis, can slow ovarian cancer by starving tumor cells of certain fats and forcing them into a special kind of iron‑driven cell death. It also looks at how this compound might weaken the “invisibility cloak” that tumors use to hide from the immune system.
A hidden fuel source inside tumor cells
Like many cancers, ovarian tumors rewire how they handle fats. Instead of simply using fats for energy, cancer cells stockpile them as building blocks for new membranes and as buffers against damage. In lab-grown ovarian cancer cells, adding extra monounsaturated fatty acids led to a buildup of free fatty acids, triglycerides, and cholesterol—key forms of stored fat. This fat surplus made the cells grow and invade more easily and helped them resist a destructive process called ferroptosis, where iron and damaged fats combine to rupture cell membranes. In other words, abnormal fat metabolism gave the cancer a growth advantage and a survival shield.
Forcing cancer cells toward self-destruction
The researchers then tested tectorigenin on both normal ovarian cells and several ovarian cancer cell lines. At doses up to 200 micrograms per liter, the compound did not harm normal cells but clearly slowed the growth of cancer cells, reduced their ability to invade through a membrane, and increased their rate of programmed cell death. When cancer cells were pretreated with a ferroptosis-blocking drug, they became more aggressive. Adding tectorigenin reversed these effects: fat stores fell, levels of iron- and oxidation-related damage markers rose, and more cells died. In mouse models bearing human ovarian tumors, injections of tectorigenin shrank tumors, reduced fat content in tumor tissues, and boosted chemical signs of ferroptosis, again suggesting the compound pushes cancer cells toward this iron‑dependent death route.
A key molecular switch linking fats and immune escape
To uncover how tectorigenin works at the molecular level, the team combined big-data mining with computer modeling of drug–protein interactions. They homed in on an enzyme called ALOX5, which converts a common fatty acid into a signaling molecule called 5‑HETE. In ovarian cancer cells and tumor samples, ALOX5 levels were much higher than in normal tissue. Computer docking and molecular dynamics simulations showed that tectorigenin can bind stably to ALOX5, acting like an internal brake. When the researchers artificially increased ALOX5 inside cancer cells, fat stores went up, ferroptosis markers went down, and the cells became more invasive. Tectorigenin treatment undid these changes. Knocking down ALOX5 had the opposite effect—less fat, more ferroptosis, and weaker growth—firmly placing this enzyme at the center of the compound’s action.
How tumors lose their shield against the immune system
The study also linked this fat pathway to an important immune checkpoint molecule called PD‑L1, which tumors use to switch off attacking immune cells. ALOX5’s product 5‑HETE boosted PD‑L1 levels, strengthening this shield. When ALOX5 was silenced, both 5‑HETE and PD‑L1 dropped; when 5‑HETE was reintroduced, PD‑L1 rose again and the cancer cells regained some of their protection from ferroptosis. Tectorigenin reduced ALOX5, lowered 5‑HETE, and in turn decreased PD‑L1 in cell cultures and in mouse tumors. This suggests that by targeting a single metabolic enzyme, the compound can both disrupt the cancer’s fat-based defenses and weaken its ability to hide from immune attack.
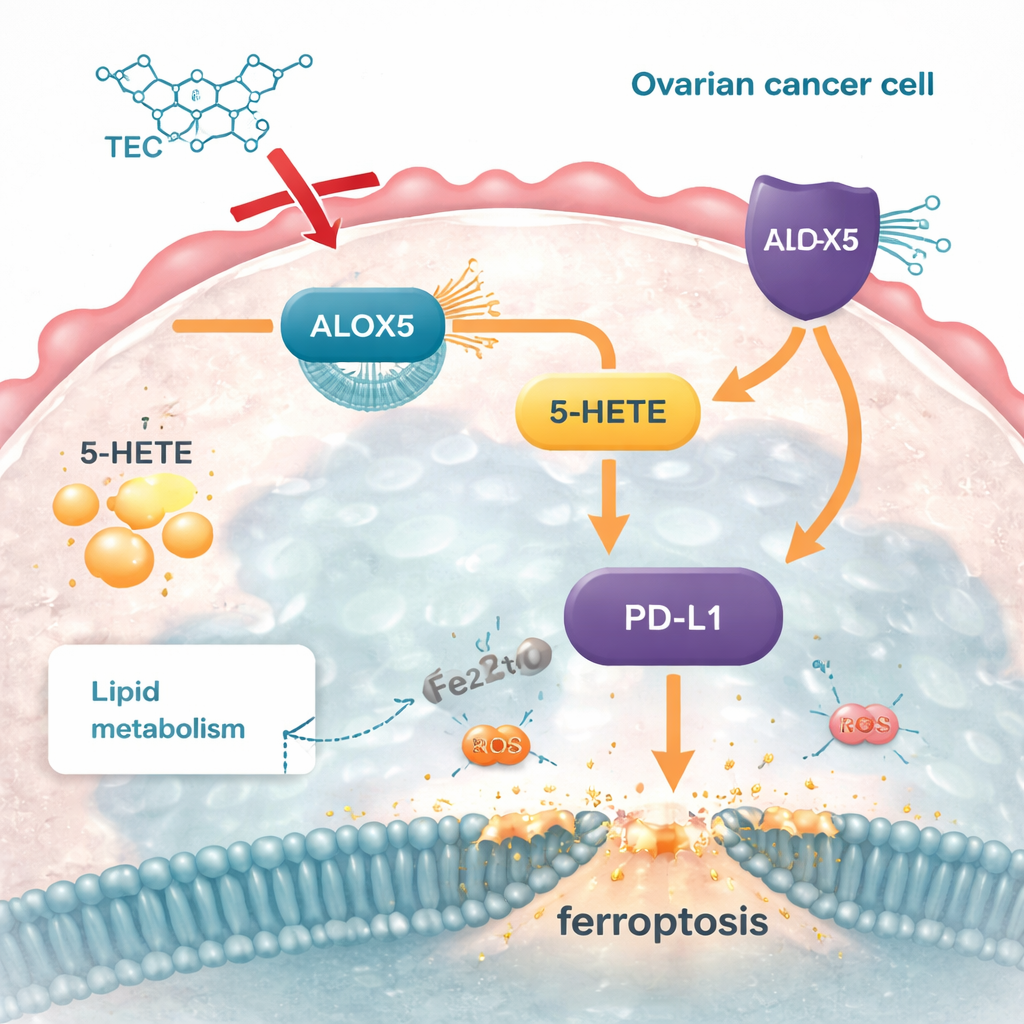
What this could mean for future treatments
Put simply, this work shows that tectorigenin acts like a precision wrench thrown into a cancer cell’s fat-handling machinery. By blocking ALOX5, it cuts off production of 5‑HETE, trims back excess fat stores, makes cells vulnerable to iron‑driven damage, and strips away part of their immune camouflage. Although these findings come from cell and mouse studies—and much remains to be tested in people—they point to a promising strategy: targeting the ALOX5–5‑HETE–PD‑L1 axis to both starve ovarian tumors and make them easier for the body’s own defenses, or future immunotherapies, to eliminate.
Citation: Cai, H., Huang, C. & Zhang, Z. Irisin regulates lipid metabolism and ferroptosis in ovarian cancer cells by modulating the ALOX5-5-HETE-PD-L1 axis. Sci Rep 16, 7223 (2026). https://doi.org/10.1038/s41598-026-36816-4
Keywords: ovarian cancer, lipid metabolism, ferroptosis, ALOX5, natural compound therapy