Clear Sky Science · en
Serum Mac-2 binding protein glycosylation isomer in predicting hepatocellular carcinoma occurrence among patients with direct-acting antiviral-induced HCV cure
Why this research matters to you
Modern medicines can now cure hepatitis C virus (HCV) in most people, but some former patients still go on to develop liver cancer years later. Doctors need better ways to tell who remains at high risk so that these individuals can be watched more closely. This study tested whether a blood marker called Mac‑2 binding protein glycosylation isomer (M2BPGi) can help predict the chance of developing hepatocellular carcinoma, the most common form of liver cancer, after a successful hepatitis C cure.
A lingering cancer risk after cure
Hepatitis C affects tens of millions of people worldwide and can silently scar the liver over many years. New direct‑acting antiviral drugs (DAAs) cure the infection in more than 95% of treated patients, greatly reducing liver damage. Yet even after the virus is cleared, people with past advanced scarring can still develop liver cancer. Current screening tools, such as ultrasound scans and alpha‑fetoprotein blood tests, miss a substantial fraction of early cancers. This leaves a pressing need for simple blood tests that can sort patients into low‑ and high‑risk groups and guide how intensively each person should be monitored.
A blood signal of liver damage
M2BPGi is a modified form of a naturally occurring protein that rises when the liver is inflamed and scarred. Unlike a biopsy, it can be measured from a routine blood sample. Earlier studies in Japan suggested that higher M2BPGi levels are linked to liver cancer in people with hepatitis C, but it was unclear whether the same cut‑off values would work in other populations and in patients treated with modern DAA drugs. To answer this, researchers in Taiwan followed 704 adults with chronic hepatitis C who were treated with DAAs and successfully cleared the virus. Everyone had M2BPGi measured just before treatment and again 12 weeks after finishing therapy, the standard time point used to confirm cure.
Following patients over the long haul
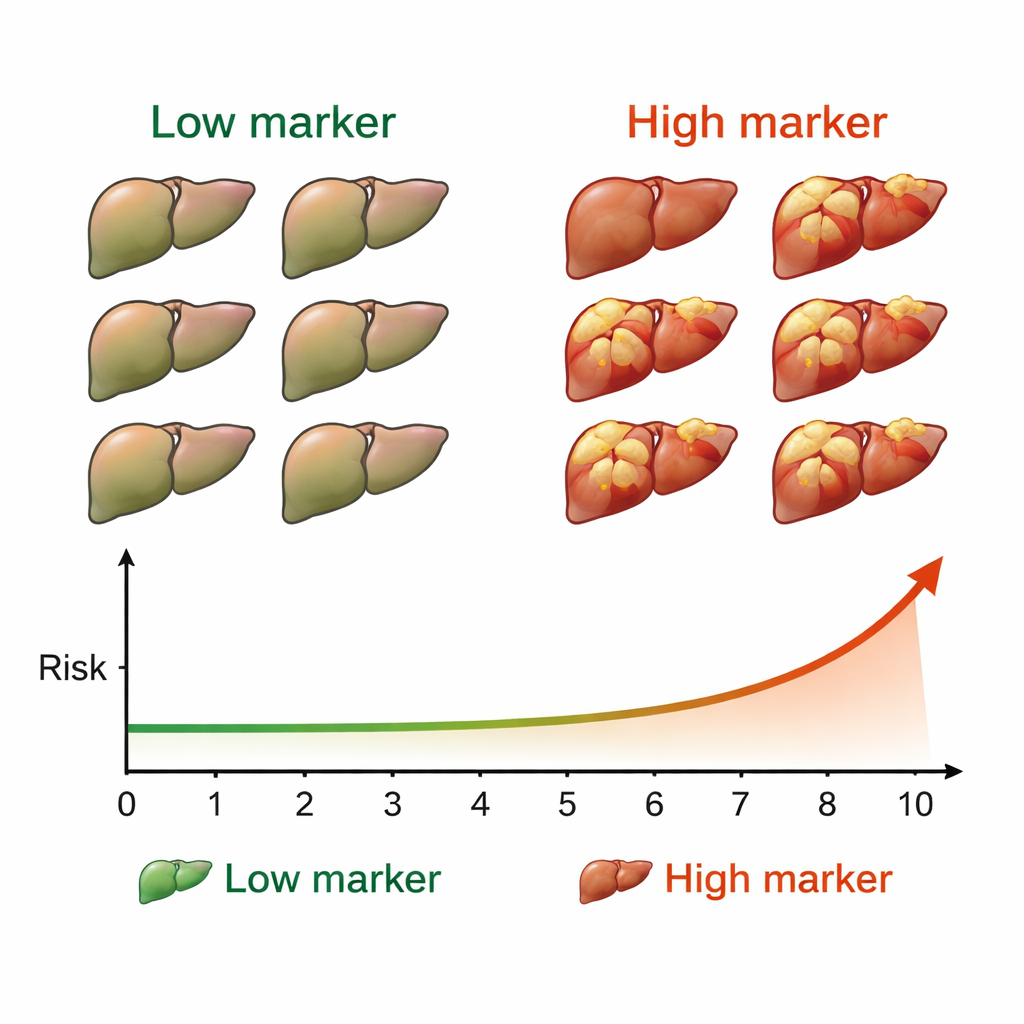
After cure, the patients were checked every six months with ultrasound and standard blood tests to look for new liver tumors. Over a median of four and a half years of follow‑up, 50 people—about 7% of the group—developed liver cancer. When the team compared cancer outcomes with M2BPGi levels, a clear pattern emerged. Patients whose M2BPGi was at least 4.0 units before treatment had far more cancers than those below that level. The difference was striking: their 10‑year cumulative cancer rate approached 53%, versus much lower rates in the low‑marker group. A second threshold, 2.0 units measured after cure, also separated patients into high‑ and low‑risk groups over the following decade.
Risk patterns over time
The researchers then combined both time points to see how changes in the marker related to risk. They divided patients into four categories based on whether M2BPGi was above or below 4.0 before treatment and above or below 2.0 after cure. People whose levels were low at both times had the lowest risk. Those whose levels were high both before and after treatment had by far the highest chance of developing liver cancer. Patients who moved from high to low, or from low to high, fell in between. Even after accounting for age, sex, ultrasound‑based measures of liver stiffness, liver enzyme tests, and alpha‑fetoprotein, M2BPGi remained an independent predictor of who would later develop cancer.
What this means for patients and doctors
For patients cured of hepatitis C, these findings suggest that a simple blood test could help clarify how worried they and their doctors should be about future liver cancer. High M2BPGi levels—especially if they stay high before and after treatment—signal that the liver remains at substantial risk, and that close, long‑term cancer surveillance is essential. Conversely, consistently low levels may reassure some patients that their risk is comparatively small, even though regular check‑ups are still advisable. In plain terms, M2BPGi acts like an early warning light for liver cancer after hepatitis C cure, offering a practical way to target the most intensive monitoring to those who need it most.
Citation: Chang, YP., Chen, YC., Su, TH. et al. Serum Mac-2 binding protein glycosylation isomer in predicting hepatocellular carcinoma occurrence among patients with direct-acting antiviral-induced HCV cure. Sci Rep 16, 5757 (2026). https://doi.org/10.1038/s41598-026-36809-3
Keywords: hepatitis C, liver cancer, blood biomarker, risk prediction, M2BPGi