Clear Sky Science · en
Biosynthesis and photocatalytic performance of Cumin extract-mediated ZnO nanoparticles with mechanistic investigation of degradation pathways
Spice Rack to Riverbank
Most of us know cumin as the warm, earthy spice in our kitchens, not as a tool for cleaning up industrial waste. This study shows how compounds extracted from humble cumin seeds can help build tiny particles that use light to scrub stubborn textile dyes from water. It is a glimpse of how everyday plants could play a role in tackling pollution from the clothes we wear.
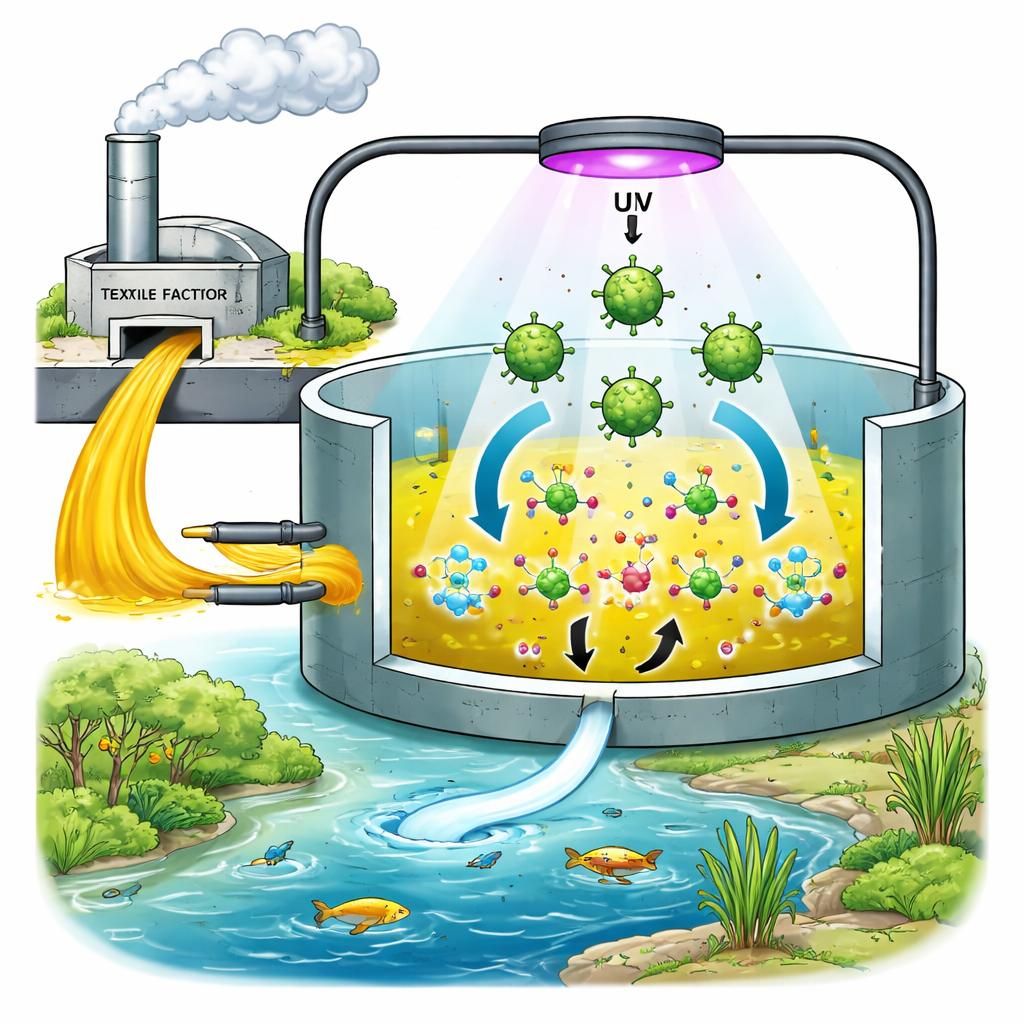
The Problem with Colorful Water
Modern textile factories use large amounts of synthetic dyes to give clothing bright, lasting colors. One such dye, Direct Yellow 86, is especially tough: it resists natural breakdown, blocks sunlight in rivers and lakes, and can form toxic byproducts that threaten fish, plants, and people. Many current clean-up methods either shift the pollution elsewhere, generate sludge that is hard to handle, or are too costly to use widely. Scientists are therefore searching for simple, affordable ways to actually break these dye molecules apart into harmless components.
Tiny Light-Driven Cleaners
The researchers focused on zinc oxide, a material already used in sunscreens and electronics, which can also act as a photocatalyst. When zinc oxide particles are exposed to ultraviolet light, they can create very reactive forms of oxygen that attack and break down organic molecules like dyes. The team produced zinc oxide in the form of nanoparticles—grains tens of thousands of times smaller than a grain of sand—because smaller, well-shaped particles have more surface area and can be more effective at cleaning. Instead of relying on harsh chemicals to make these particles, they used water extracts of cumin seeds, whose natural plant compounds both reduce zinc salts into zinc oxide and help keep the particles from clumping.
How the Cumin-Based Nanoparticles Work
Using several analytical techniques, the authors confirmed that they had successfully made pure, highly crystalline zinc oxide nanoparticles coated with cumin-derived molecules. Microscopy images showed mostly spherical particles around 60 nanometers across, evenly dispersed without large clumps. Chemical fingerprints indicated that plant-based groups were still attached to the surface, helping to stabilize the particles in water. When the nanoparticles were placed in a small reactor with solutions of the yellow dye and lit by a low-power UV lamp, the solution’s color faded rapidly, revealing that the particles were actively driving dye breakdown.
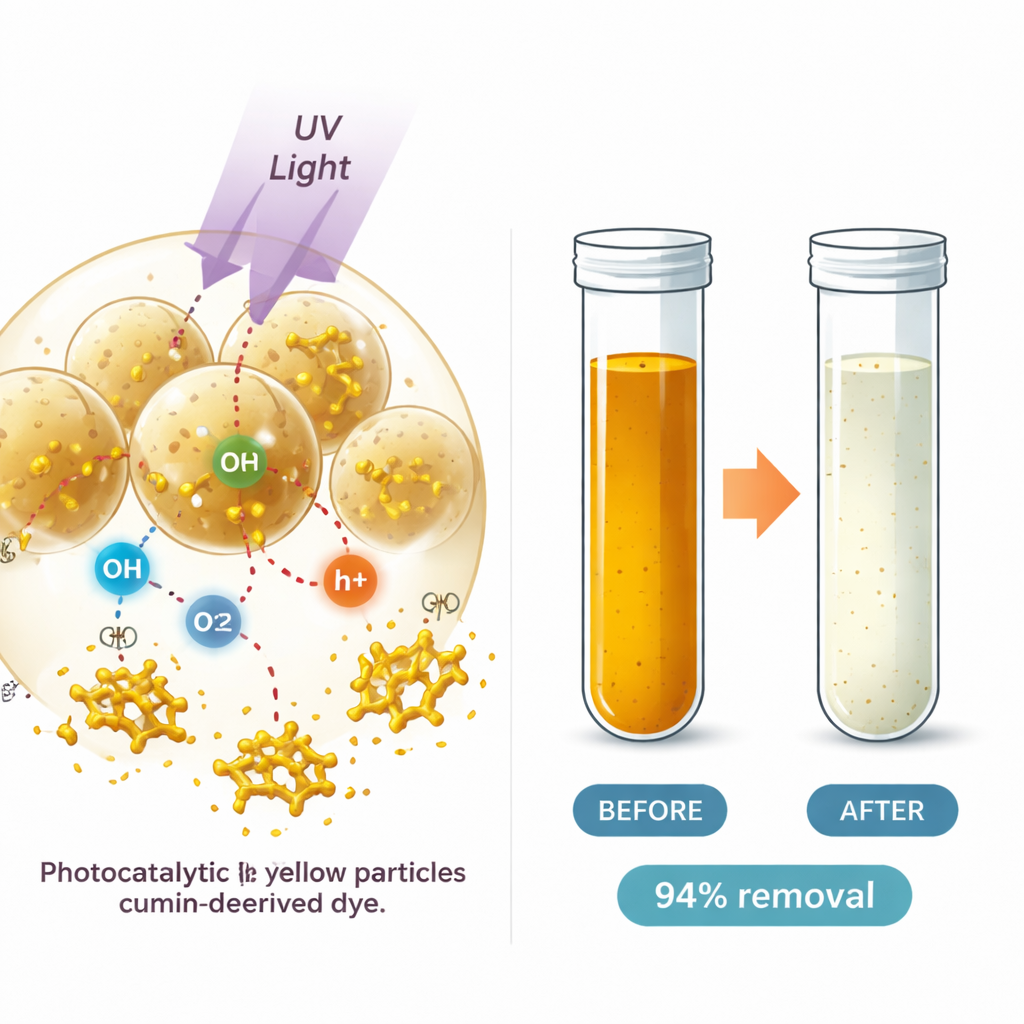
Finding the Sweet Spot for Clean-Up
To make the process practical, the team systematically varied four key conditions: the acidity of the water, how much dye it contained, how much catalyst they added, and how long they shone the UV light. Using a statistical approach called response surface design, they ran 29 different combinations and built a mathematical model that predicts how efficiently the dye can be removed. They found that performance was most sensitive to pH: strongly alkaline water gave the best results, because it encouraged the formation of more reactive oxygen species. There was also an optimal catalyst amount—too little gave few active sites, while too much scattered the light and reduced effectiveness. Under the best conditions, the system removed about 94 percent of the dye in just 30 minutes.
Following the Dye as It Falls Apart
To move beyond simple color measurements, the researchers tracked the actual dye molecules and their fragments using liquid chromatography–mass spectrometry, a technique that separates and weighs molecules with great precision. Before treatment, the yellow solution showed strong signals corresponding to the intact dye and related aromatic structures. After exposure to the cumin-based nanoparticles and UV light, those signals almost disappeared, replaced by many new peaks that matched smaller, more oxidized fragments. This pattern fits a stepwise breakdown path in which the most energetic bonds in the dye molecule are attacked first, followed by gradual cutting and oxidation of the rings until only small, less harmful pieces remain.
From Lab Bench to Cleaner Streams
In plain terms, this work demonstrates that nanoparticles grown with help from cumin seeds can use simple UV light to tear apart a particularly stubborn textile dye, turning bright yellow wastewater into nearly clear liquid within half an hour. While more studies are needed to scale up the process and confirm long-term safety, the findings point toward a future in which plant-assisted materials help clean industry’s dirtiest streams in a cheaper and more environmentally friendly way.
Citation: Mehralian, M., Shahrokhi, A.M., Mohammadi, F. et al. Biosynthesis and photocatalytic performance of Cumin extract-mediated ZnO nanoparticles with mechanistic investigation of degradation pathways. Sci Rep 16, 6198 (2026). https://doi.org/10.1038/s41598-026-36801-x
Keywords: textile wastewater, photocatalysis, zinc oxide nanoparticles, green synthesis, azo dye removal