Clear Sky Science · en
Adaptation of conventional water treatment technologies for organic component removal from liquid radioactive waste: sorption and coagulation mechanisms
Why Cleaning Radioactive Water Matters to Everyone
Nuclear power plants, research centers, and hospitals all create liquid radioactive waste that often contains stubborn organic chemicals such as oils, detergents, and solvents. These organics make radioactive waste harder and more expensive to clean, and they can interfere with safely locking radioactive materials into solid form. This study explores whether the same straightforward methods used to clean ordinary drinking water and wastewater could also help strip out these organic pollutants from radioactive liquids, offering a cheaper and more practical option—especially important for countries like Ukraine facing war-related strains on water and energy supplies.
Old Tools for a New Kind of Waste
Modern nuclear facilities often rely on advanced technologies such as membranes, plasma, or powerful oxidizing chemicals to treat liquid radioactive waste. While effective in the laboratory, these methods tend to be energy-intensive, technically complex, and not yet widely available as ready-to-use industrial equipment. Meanwhile, familiar drinking water treatments—like adsorption onto activated carbon, coagulation with metal salts, and simple filtration—are well tested, relatively inexpensive, and easy to operate. The central question of this research was whether these established techniques, already common in municipal water plants, could be adapted to remove the organic portion of liquid radioactive waste and make the final waste easier to solidify and store safely.
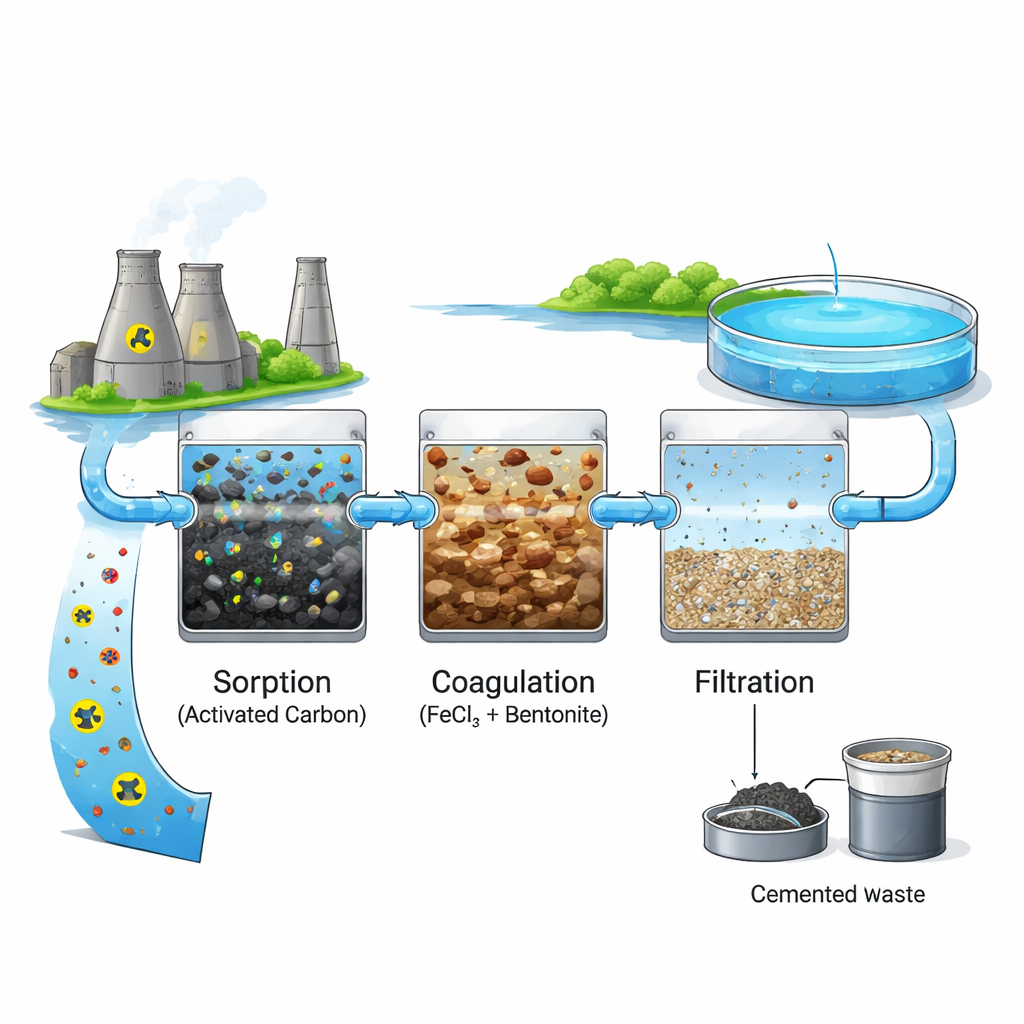
How the Cleaning Steps Work Together
The researchers built a model liquid waste that mimicked the organic mix typically found in nuclear facilities, combining hydrazine, organic acids, detergents, and other common additives in water. They then applied a three-step treatment: first, finely powdered activated carbon was added and gently mixed to adsorb dissolved organic molecules onto its vast internal surface. Next, powdered bentonite clay from a large Ukrainian deposit was introduced as a turbidity agent, followed by a ferric chloride solution acting as a coagulant. In this stage, iron compounds helped bind suspended particles and bentonite into larger clumps, sweeping up additional organics as they formed. After a short settling period, the clarified water was passed through filter paper to capture the resulting sludge, leaving a much cleaner liquid behind.
What the Experiments Revealed
The team measured organic pollution using three standard indicators: total organic carbon (TOC) and two versions of chemical oxygen demand, COD(Mn) and COD(Cr), which reflect how much oxidizing power is needed to break down the organics. With optimized doses of activated carbon, bentonite, and ferric chloride, the process cut TOC by about 2.85 times, COD(Mn) by 2.63 times, and COD(Cr) by 4.19 times—corresponding to roughly 75% removal of dissolved organics. Statistical analysis showed that activated carbon and the iron-based coagulant were the main drivers of cleaning efficiency, while bentonite’s role was more subtle. When used in moderate amounts, bentonite sped up coagulation and sedimentation, but adding too much actually made the colloidal particles more stable and reduced how much organic matter could be removed.
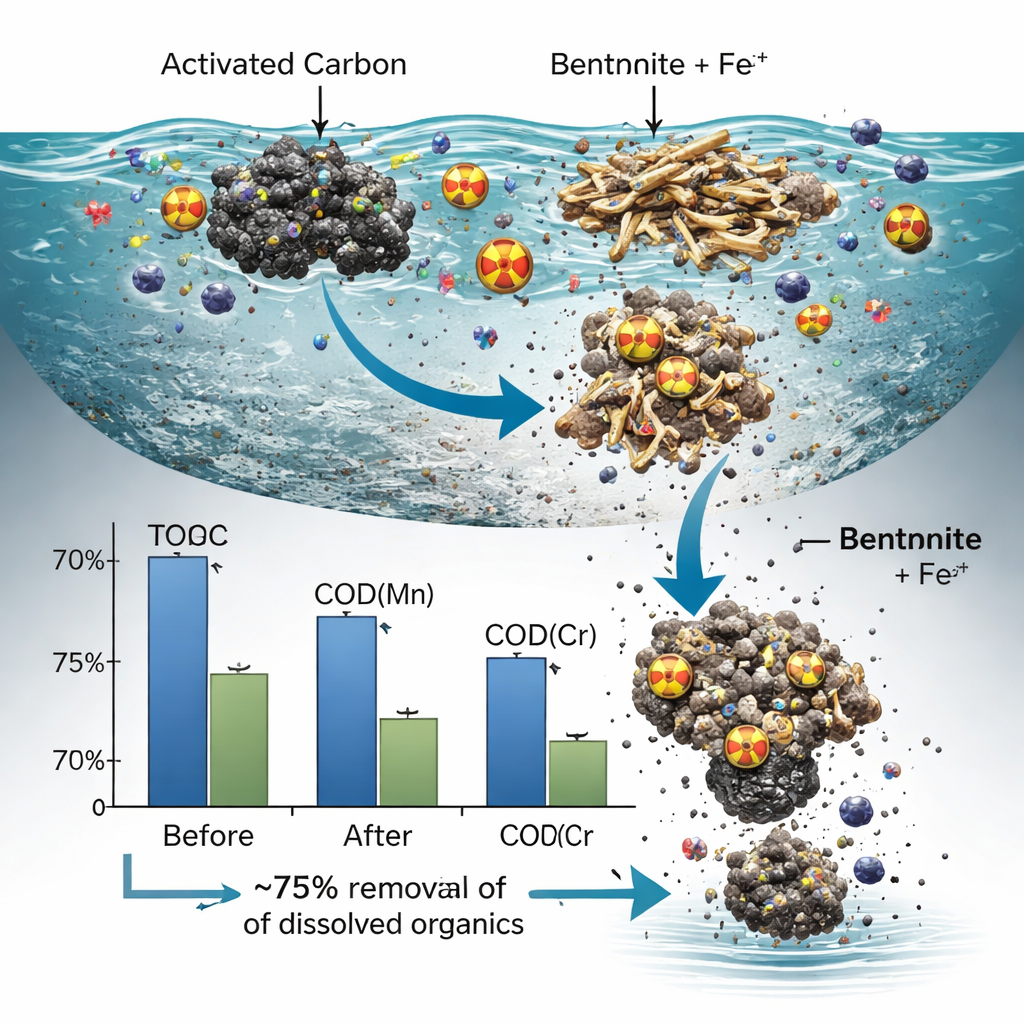
Making Sense of Different Pollution Tests
In real-world monitoring, laboratories may not always measure organic pollution the same way; some rely on COD(Cr), others on COD(Mn) or TOC. To bridge these gaps, the authors built mathematical “conversion” models that allow workers to estimate one indicator from another using simple equations. Within the range of their experiments, COD(Cr) values could be reliably translated into COD(Mn) or TOC, helping operators compare results, benchmark treatment performance, and make decisions even if only one type of test is available. This makes it easier to integrate the new method into existing plant control systems without overhauling laboratory routines.
From Sludge to Solid Safety
Beyond cleaning the water, the study highlights what happens to the captured contaminants. The combined sorption–coagulation process concentrates organic matter and radionuclides into a sludge that can be mixed into special alkaline concretes, known as geoconcretes. These materials are resistant to leaching and do not require high-temperature processing, offering a durable way to lock radionuclides into a solid form while returning treated water safely to the environment. For Ukraine, where nuclear power is crucial, fresh water is limited, and infrastructure is under wartime pressure, such low-cost, low-energy, and robust methods could significantly reduce risks from stored radioactive liquids.
What This Means in Everyday Terms
Put simply, the researchers showed that you do not always need cutting-edge, power-hungry technology to make radioactive wastewater safer. By smartly combining well-known steps—letting activated carbon grab organic pollutants, using clay and iron salts to clump and settle them, and then filtering the mixture—they achieved about three- to fourfold reductions in organic contamination. This makes the remaining radioactive waste easier to turn into a stable solid and cuts the volume of dangerous liquid that must be stored. For the public, this work points toward more affordable and deployable ways to keep nuclear energy’s liquid by-products under control, even in regions with tight budgets and strained infrastructure.
Citation: Charnyi, D., Zabulonov, Y., Lukianova, V. et al. Adaptation of conventional water treatment technologies for organic component removal from liquid radioactive waste: sorption and coagulation mechanisms. Sci Rep 16, 2626 (2026). https://doi.org/10.1038/s41598-026-36799-2
Keywords: radioactive wastewater, activated carbon, bentonite clay, coagulation and filtration, nuclear waste treatment