Clear Sky Science · en
Experimental characterization of proton minibeam therapy delivery under FLASH dose-rate conditions
Faster, Gentler Radiation Beams
Radiation therapy is a mainstay of cancer treatment, but it can harm healthy tissue along with tumors. This study explores a new way to deliver proton radiation that aims to hit tumors hard while softening the blow to normal organs. By combining two emerging ideas—very finely spaced “minibeams” and ultra-fast FLASH dose rates—the researchers show that it is technically possible to deliver highly precise treatments in just a few seconds using equipment similar to what already exists in modern hospitals.
Why Make Radiation into Tiny Beams?
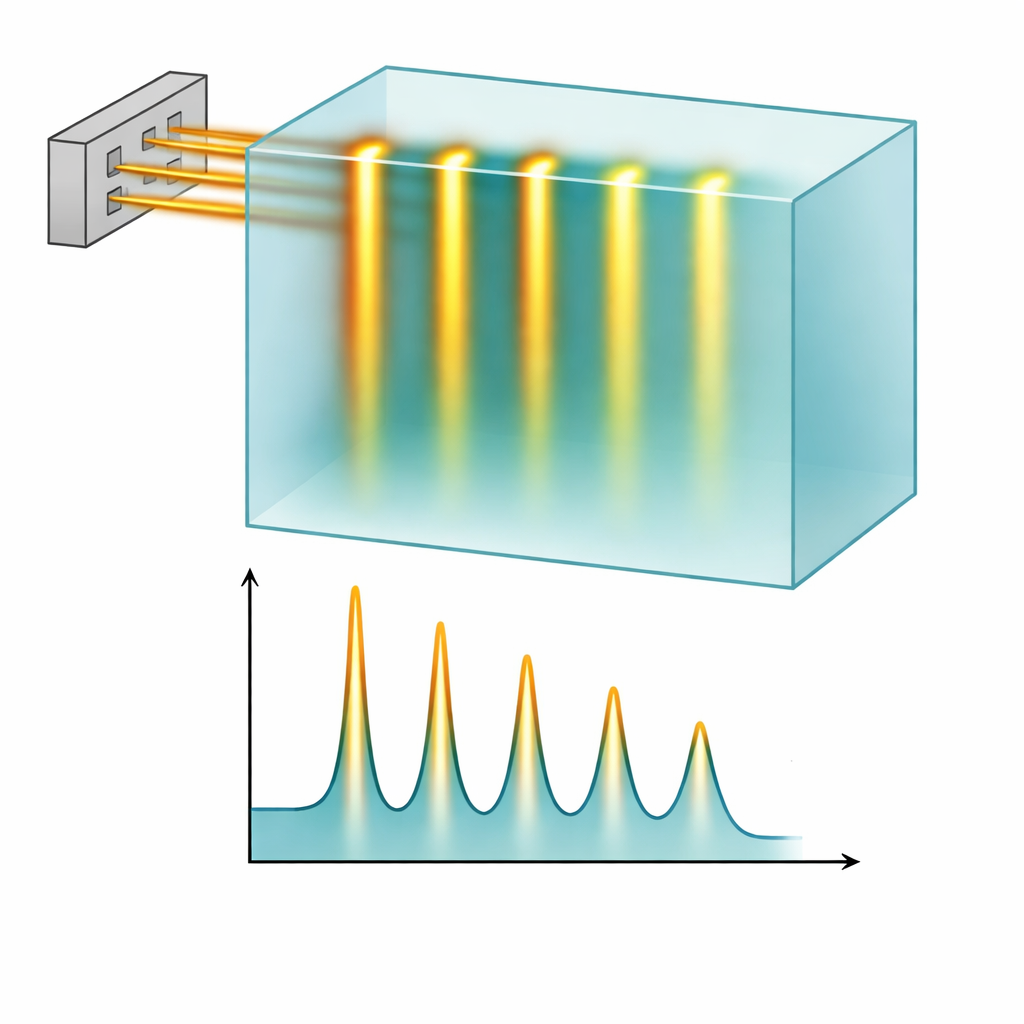
Traditional radiation fields look smooth: the dose is spread fairly evenly across the treated region. Spatially fractionated radiation therapy breaks that rule on purpose. In proton minibeam radiation therapy, the radiation is split into many thin, submillimeter beams separated by gaps. Near the surface, this creates a pattern of high-dose “peaks” and low-dose “valleys.” Healthy tissue between the peaks has a better chance to repair itself, while deeper in the body the beams spread out and overlap, giving the tumor a more uniform and effective dose. Early laboratory and animal studies suggest that this pattern can reduce side effects and may even trigger helpful immune responses against cancer.
What Is FLASH, and Why Does Speed Matter?
FLASH therapy is a new way of thinking about time in radiation delivery. Instead of slowly giving a dose over tens of seconds or minutes, FLASH delivers the same dose in a fraction of a second at ultra-high dose rates—tens to hundreds of gray per second. Surprisingly, many experiments have shown that normal tissues can be better preserved under these extreme conditions, while tumors remain just as vulnerable. For proton minibeams, however, there has been a practical obstacle: the multi-slit metal blocks used to carve the beam into thin rays waste a lot of the incoming particles, so treatments can take several minutes. If minibeams could be delivered in FLASH mode, the lost time might be regained and the biological benefits of both methods could be combined.
Putting Minibeams and FLASH on a Clinical Machine
The team worked with a compact clinical proton system that usually treats patients with a smooth, scanning proton beam. They attached custom brass multi-slit collimators—each containing five narrow slits only 1 millimeter wide and spaced 2.8 millimeters apart—to the end of the treatment nozzle. By increasing the beam current and carefully tuning the beam optics, they operated the system in an ultra-high-dose-rate mode at 228 MeV, while still being able to switch back to ordinary clinical settings. They then measured how the dose was distributed in water-like plastic blocks using special radio-sensitive films and compared those measurements to detailed computer simulations that track individual proton interactions.
How Well Did the New Beam Perform?
Under FLASH conditions, the proton beam maintained a clear minibeam pattern: sharp peaks along the slit paths and deep valleys in between. The measurements and simulations agreed closely on how this pattern evolved with depth, and on how quickly it blurred out as the beams scattered. A thicker 10 cm collimator produced cleaner separation between peaks and valleys than a 6.5 cm version, especially near the surface, meaning it better protected the “valley” regions. Crucially, running the machine in FLASH mode cut the delivery time for a typical 3 by 3 centimeter minibeam field from about 3 minutes to just 2.5 seconds. Calculations based on the machine’s timing logs showed that local dose rates in the peak regions easily exceeded the commonly used FLASH thresholds, all while preserving the fine spatial structure of the minibeams.
What This Could Mean for Future Cancer Care
This work is an early but important proof of principle. It shows that a standard-type clinical proton therapy system can be adapted to deliver proton minibeams at FLASH dose rates without sacrificing precision or beam quality. Patients are not being treated this way yet: the experiments were done in test setups, and only a single high-energy beam was used, rather than the layered energy patterns that real tumors require. The biological effects of combining spatial minibeam patterns with ultra-rapid FLASH delivery also remain to be proven in animals and, eventually, people. Still, the study lays the technical groundwork for future treatments that might control tumors as effectively as today’s best methods while dramatically reducing damage to healthy organs by exploiting both where and how fast radiation is given.
Citation: Lin, Y., Wu, W., Setianegara, J. et al. Experimental characterization of proton minibeam therapy delivery under FLASH dose-rate conditions. Sci Rep 16, 7803 (2026). https://doi.org/10.1038/s41598-026-36739-0
Keywords: proton therapy, FLASH radiotherapy, minibeam radiation, cancer treatment, radiation dose rate